
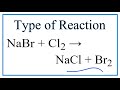
Chemical Reactions and Oxidation States
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Lucas Foster
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What type of reaction occurs between NaBr and Cl2?
Synthesis
Double replacement
Single replacement
Decomposition
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In a single replacement reaction involving NaBr and Cl2, which element is replaced?
Sodium
Chlorine
Bromine
Hydrogen
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the final product when NaBr reacts with Cl2?
Sodium chloride
Bromine gas
Sodium bromide
Chlorine gas
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is chlorine able to replace bromine in the reaction with NaBr?
Chlorine is less reactive than bromine
Chlorine is more reactive than bromine
Chlorine and bromine have the same reactivity
Bromine is more reactive than chlorine
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the purpose of balancing a chemical equation?
To alter the reactants
To change the products
To ensure mass conservation
To increase the reaction rate
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the oxidation state change for bromine in the reaction?
From 0 to +1
From -1 to 0
From 0 to -1
From +1 to 0
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to chlorine's oxidation state in the reaction?
It remains the same
It increases
It decreases
It fluctuates
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
