

The Periodic Table and Bonding
Presentation
•
Chemistry
•
6th - 8th Grade
•
Practice Problem
•
Medium
Standards-aligned
Michael Stange
Used 104+ times
FREE Resource
8 Slides • 8 Questions
1
The Periodic Table and Bonding

2

3

4
Bonding
When elements chemically combine to form compounds, they either lose, share, or gain electrons.
Metals LOSE electrons (+ ions)
Metalloids (and elements near them on the periodic table) typically SHARE electrons
Nonmetals GAIN electrons (_ ions)
5
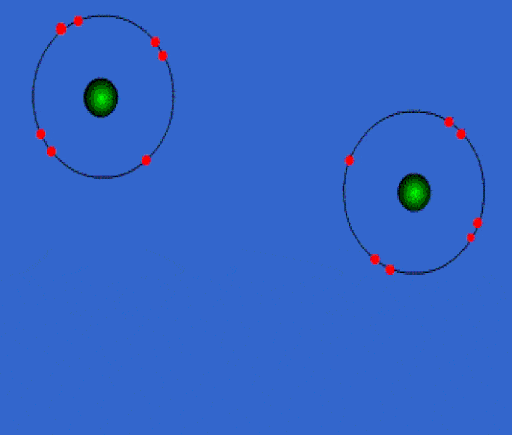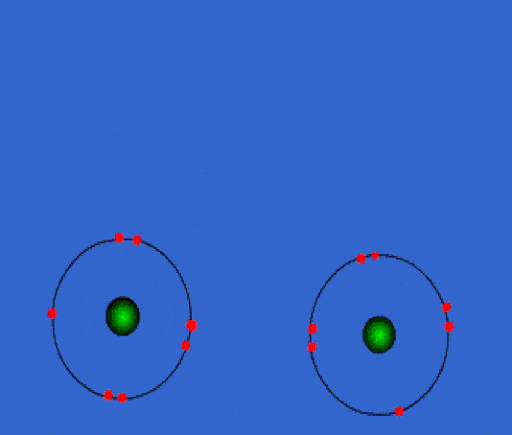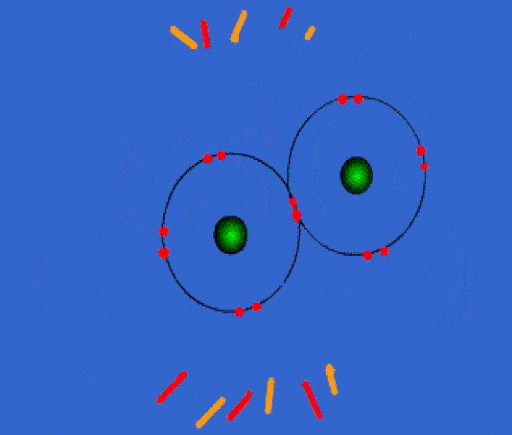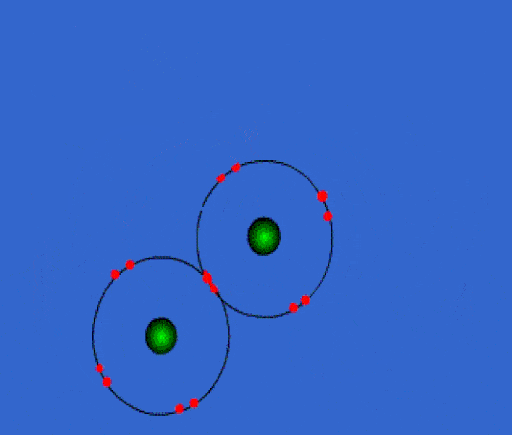
Bonding
Whenever you have an electron pair from 2 separate elements, it creates an attraction that bonds the atoms together to form a compound.

6
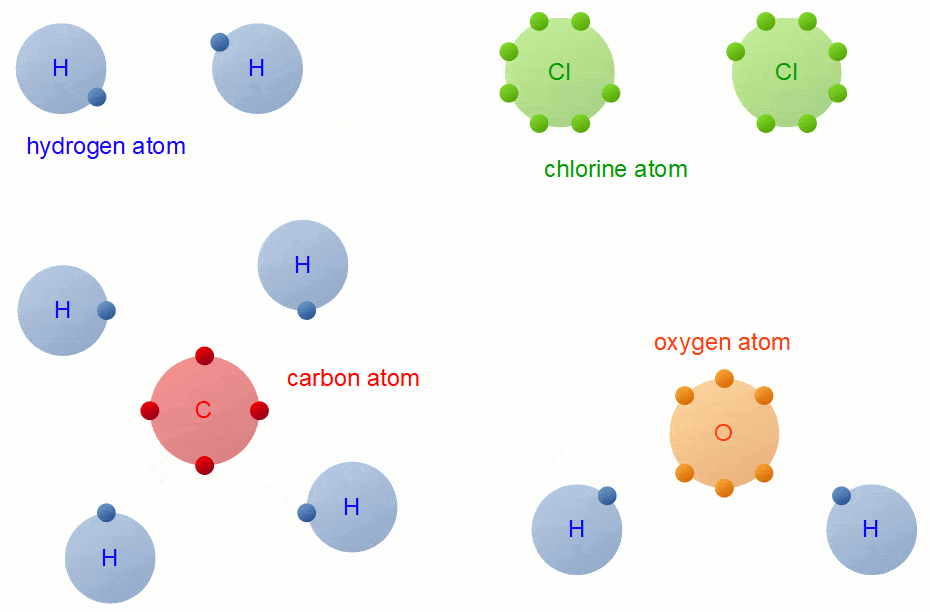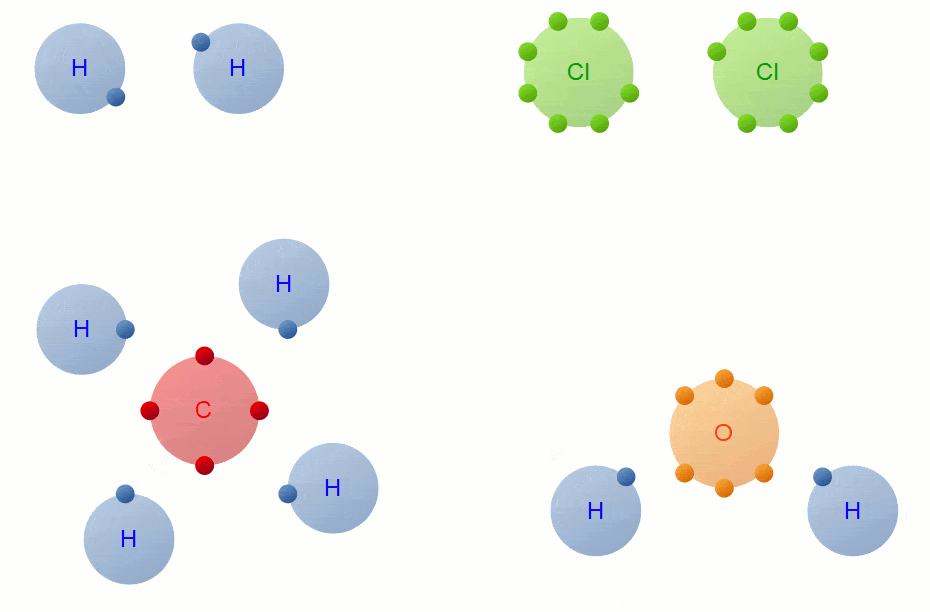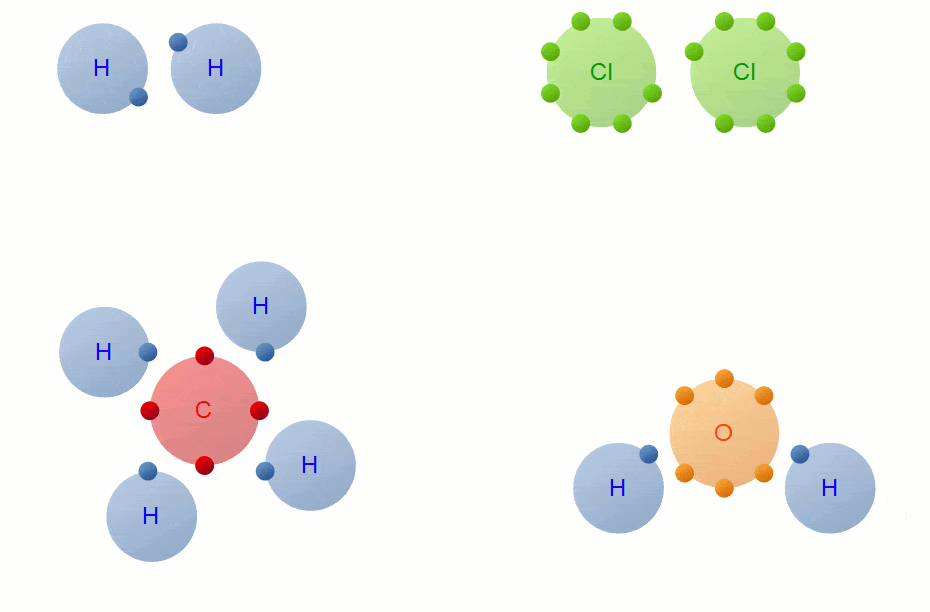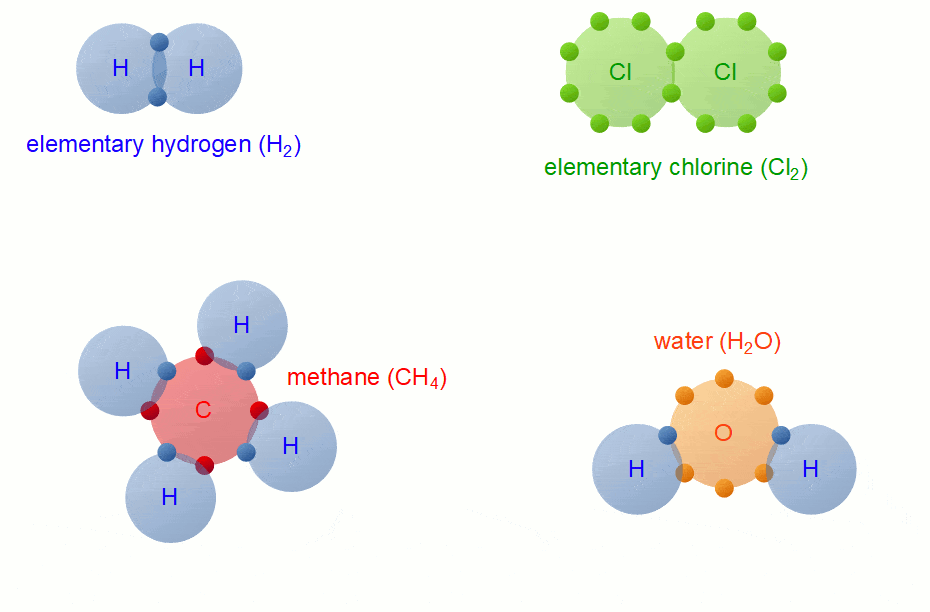
7

8

9
Multiple Choice

In the periodic table, the number of valence electrons in each element DECREASES from left to right across each period.
True
False
10
Multiple Choice

Atoms tend to be stable and nonreactive if they have ______ valence electrons.
2
4
6
8
11
Multiple Choice

Which group of elements on the periodic table is composed gases that easily react with metals?
alkali metals
alkaline earth metals
halogens
noble gases
12
Multiple Choice

Which group of elements on the periodic table is composed of super reactive metals?
alkali metals
alkaline earth metals
halogens
noble gases
13
Multiple Choice

The reactivity of a NONMETAL depends on how easily it _______ valence electrons.
loses
gains
shares
14
Multiple Choice

The reactivity of a METAL depends on how easily it _______ its valence electrons.
loses
gains
shares
15
Multiple Choice

When an atom loses an electron, it becomes a __________, and when an atom gains an electron, it becomes a __________.
positive ion, negative ion
negative ion, positive ion
neutral ion, positive ion
neutral atom, negative ion
16
Multiple Choice

A(n) ____________ ______________ is the force of attraction that holds atoms together based on the arrangement of electrons between them.
electron cloud
chemical bond
positive charge
negative charge
The Periodic Table and Bonding

Show answer
Auto Play
Slide 1 / 16
SLIDE
Similar Resources on Wayground

10 questions
Movement
Presentation
•
6th - 9th Grade

11 questions
HTML coding
Presentation
•
7th Grade

11 questions
Balancing Equations: Practice
Presentation
•
6th - 8th Grade

11 questions
5.4.18
Presentation
•
6th - 8th Grade

12 questions
Greetings and Introductions Pt. 2 8B
Presentation
•
6th - 8th Grade

11 questions
PHYSICAL AND CHEMICAL PROPERTIES
Presentation
•
6th - 8th Grade

14 questions
Heat Transfer
Presentation
•
6th - 8th Grade

12 questions
Element Compound Lesson
Presentation
•
6th - 8th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

13 questions
Bill Nye - Energy
Interactive video
•
6th Grade

20 questions
Counting Atoms Practice
Quiz
•
8th Grade

20 questions
Counting Atoms
Quiz
•
8th Grade

18 questions
Experimental Design
Presentation
•
8th Grade

10 questions
Balancing Chemical Equations Challenge
Interactive video
•
6th - 10th Grade

41 questions
Atomic Structure and Periodic Table Unit Review
Quiz
•
8th Grade