

Molecular Polarity
Presentation
•
Science
•
12th Grade
•
Practice Problem
•
Easy
+2
Standards-aligned
Salvador Magalong
Used 27+ times
FREE Resource
34 Slides • 26 Questions
1
Molecular Polarity
Synthesis Session

2
Poll
Self- Assessment:
• I can define what chemical bonding is and explain why it happens.
CRYSTAL CLEAR
CLEAR ENOUGH
BLURRY
3
Poll
Self- Assessment:
• I can differentiate the different types of chemical bonds from one another.
CRYSTAL CLEAR
CLEAR ENOUGH
BLURRY
4
Poll
Self- Assessment:
• I can identify the type of chemical bond present between two atoms.
CRYSTAL CLEAR
CLEAR ENOUGH
BLURRY
5
Poll
Self- Assessment:
• I can determine verbally if a molecule is polar or non-polar given its structure.
CRYSTAL CLEAR
CLEAR ENOUGH
BLURRY
6
Poll
Self- Assessment:
• I can relate the polarity of a molecule to its properties.
CRYSTAL CLEAR
CLEAR ENOUGH
BLURRY
7
REVIEW of the
LESSON PLAYLIST RESPONSES and FEEDBACK
8
Open Ended
Synthesis Question 1:
What is a chemical bond?
9
Synthesis Question 1:
What is a chemical bond?
an electrical attraction between the nuclei and valence electrons of an atom and which binds atoms together.

10
Open Ended
Synthesis Question 2:
Differentiate the three (3) types of chemical bonds that may exist in substances: ionic, covalent, and metallic.
11
Types of Chemical Bonds
a. ionic – transfer of electrons
(metal and nonmetal )(cation and anion)

12
Types of Chemical Bonds
b. covalent – sharing of electrons (among nonmetals)

13
Types of Chemical Bonds
c. metallic – existing among metals

14
Open Ended
Synthesis Question 3:
How will you differentiate POLAR from NON-POLAR covalent bond?
15
Non-polar covalent bond
• electrons are equally shared by the bonded atoms.
• balanced distribution of electrical charge
• electrons spend the same length of time in the locality of each atom

16
Polar covalent bond
• bonded atoms have an unequal sharing of electrons (partial electron transfer)

17
Multiple Choice
Concept Check 1:
In a polar covalent bond, the electron density (length of time of the electrons in the locality of each atom) is the same around the nuclei of the atoms involved.
True
False
18
Multiple Choice
Concept Check 2:
In a polar covalent bond, the electrons are closer to the more electronegative atom.
True
False
19
DIPOLE MOMENT
20
Dipole Moment
• the two partially charged regions or poles of a molecule are referred to as a dipole.
• symbolized by an arrowhead to the more electronegative atom (negative pole), and a crossed tail located at the less negative atom (positive pole)

21
Open Ended
Synthesis Question 4:
What is electronegativity?
22
Electronegativity
A measure of the tendency of an atom to attract electrons toward itself.

23
HOW TO DISTINGUISH A NONPOLAR FROM A POLAR COVALENT BOND?

24
Concept Check 3
Predict first if the bond between each pair of atoms is ionic or covalent. Further, classify the covalent bond as either polar or nonpolar using the concept of electronegativity. Determine the more electronegative atom in each pair.
25

For 5 minutes, answer the items above using your notebook and get ready with your answers in the next Quizizz Fill-in-the-blanks questions.
26
Fill in the Blanks
Type answer...
27
Multiple Choice
Pair 1: Chlorine and Potassium
TYPE of BOND
Ionic
Polar
Non-polar
28
Multiple Choice
Pair 1: Chlorine and Potassium
MORE ELECTRONEGATIVE ATOM
Chlorine
Potassium
29
Fill in the Blanks
Type answer...
30
Multiple Choice
Pair 2: Chlorine and Carbon
TYPE of BOND
Ionic
Polar
Non-polar
31
Multiple Choice
Pair 2: Chlorine and Carbon
MORE ELECTRONEGATIVE ATOM
Chlorine
Potassium
32
Fill in the Blanks
Type answer...
33
Multiple Choice
Pair 3: Two atoms of Chlorine
TYPE of BOND
Ionic
Polar
Non-polar
34
Multiple Choice
Pair 3: Two atoms of Chlorine
MORE ELECTRONEGATIVE ATOM
Cannot be determined
Same
35
MOLECULAR GEOMETRY and POLARITY
A molecule whose bonds are all nonpolar in character is likely to be nonpolar overall.
However, NOT ALL POLAR COVALENT BONDS result to POLAR MOLECULES!!!
Molecule polarity is also affected by the molecular geometry, which in turn is influenced by the electrons in the molecule.
36
MOLECULAR GEOMETRY and POLARITY
• The polarity of an atom-atom bond may be different from the polarity of the whole molecule itself.

37
MOLECULAR GEOMETRY and POLARITY
•The difference in the electron density of each atom makes EACH Be-H bond POLAR.
• However, since the molecular geometry of BeH2 is LINEAR and the two poles are on the OPPOSITE SIDES, the polarities “CANCEL OUT”, which makes the whole molecule NONPOLAR. (NO DIPOLE MOMENT!)

38
NOTES on MOLECULAR GEOMETRY and POLARITY
When determining the polarity of a molecule, it is all about symmetry.
Asymmetric molecules tend to be polar.
Symmetric molecules are always non-polar.
39
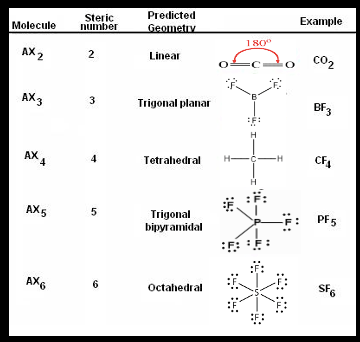
COMMON MOLECULAR GEOMETRIES
Polar or non-polar?

40
Key
1. Nonpolar
2. Nonpolar
3. Nonpolar
4. Nonpolar
5. Nonpolar
41
Key
6. Polar
7. Polar
8. Polar

42
EXERCISES
Given the Lewis structure, predict if each substance is polar or non-polar.
43
Multiple Choice

1. N2
Polar
Non-polar
44
Multiple Choice

2. SO2
Polar
Non-polar
45
Multiple Choice

3. BrF3
Polar
Non-polar
46
Multiple Choice

4. HCN
Polar
Non-polar
47
Multiple Choice

5. CH2Cl2
Polar
Non-polar
48
Open Ended
GIve at least two (2) insights you had from our discussions.
49

50

51

52

53

54

55

56

57

58

59

60

Molecular Polarity
Synthesis Session

Show answer
Auto Play
Slide 1 / 60
SLIDE
Similar Resources on Wayground

51 questions
Average Atomic Mass
Presentation
•
11th Grade

53 questions
Folding of Amino Acids
Presentation
•
11th - 12th Grade

49 questions
CH 2 Test Review
Presentation
•
11th - 12th Grade

49 questions
Big Bang
Presentation
•
11th - 12th Grade

49 questions
Big Bang Theory Lesson
Presentation
•
11th - 12th Grade

49 questions
Big Bang Theory
Presentation
•
11th - 12th Grade

56 questions
Science Form 4 KSSM (Revision c9-12)
Presentation
•
12th Grade

53 questions
8.6 Review (Slides and questions)
Presentation
•
11th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade

49 questions
AP Environmental Science Final Exam Review
Quiz
•
10th Grade - University

16 questions
Taxonomy and Classification
Quiz
•
6th - 12th Grade

50 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

28 questions
Greenhouse effect
Quiz
•
9th - 12th Grade