

Chapter 4. Atoms and Elements
Presentation
•
Chemistry
•
University
•
Medium
+5
Standards-aligned

Luis Bello
Used 16+ times
FREE Resource
35 Slides • 43 Questions
1
Chapter 4. Atoms and Elements
Presenter: Dr. Luis Bello

2
Previous... Chapter 3 Matter and Energy
•3.3 Classifying Matter According to Its State: Solid, Liquid, and Gas
•3.4 Classifying Matter According to Its Composition: Elements, Compounds, and Mixtures
•3.5 Differences in Matter: Physical and Chemical Properties
•3.6 Changes in Matter: Physical and Chemical Changes
•3.7 Conservation of Mass: There Is No New Matter
•3.8 Energy
•3.9 Energy and Chemical and Physical Change
•3.1Temperature: Random Motion of Molecules and Atoms
•3.1Temperature Changes: Heat Capacity
•3.1Energy and Heat Capacity Calculation
3
Checking Comprehension
4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
The specific heat of aluminum is 0.9025 J/g°C. How much heat(Q) is released when a 10.0 g piece of aluminum foil is taken out of the oven and cools from 100.0° to 50.0°?
451 J
45.1 J
400 J
7
Multiple Choice
How many Hydrogen atoms are in 4H2O?
6
8
2
4
8
Multiple Choice
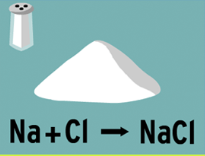
When sodium and chlorine combine to form sodium chloride, sodium chloride is the:
Originator
Reactant
Product
Produce
9
Multiple Choice
10
Multiple Choice

11
Multiple Choice
12
Multiple Choice
13
Today: Chapter 4. Atoms and Elements
4.2 Indivisible: The Atomic Theory
4.3 The Nuclear Atom
4.4 The Properties of Protons, Neutrons, and Electrons
4.5 Elements: Defined by Their Numbers of Protons
4.6 Looking for Patterns: The Periodic Law and the Periodic Table
4.7 Ions: Losing and Gaining Electrons
4.8 Isotopes: When the Number of Neutrons Varies
4.9 Atomic Mass: The Average Mass of an Element’s Atoms
14
Early Ideas about Atoms
15

16
17
Dalton’s experiments
Dalton’s experiments with gases led to some of the earliest measurements of atomic masses and a concept of atomic structure and reactivity.
Dalton’s atomic theory contained the following ideas:
All atoms of a given element are identical.
The atoms of different elements vary in mass and size. Atoms are indestructible.
Chemical reactions may result in their rearrangement, but not their creation or destruction.
18
The first part to be discovered was the electron, a tiny subatomic particle with a negative charge. It is often represented as e−, with the right superscript showing the negative charge.
Later, two larger particles were discovered. The proton is a more massive (but still tiny) subatomic particle with a positive charge, represented as p+.
The neutron is a subatomic particle with about the same mass as a proton but no charge. It is represented as either n or n0. We now know that all atoms of all elements are composed of electrons, protons, and (with one exception) neutrons.
19

20
The Properties of Protons, Neutrons, and Electrons
21

22

23

24
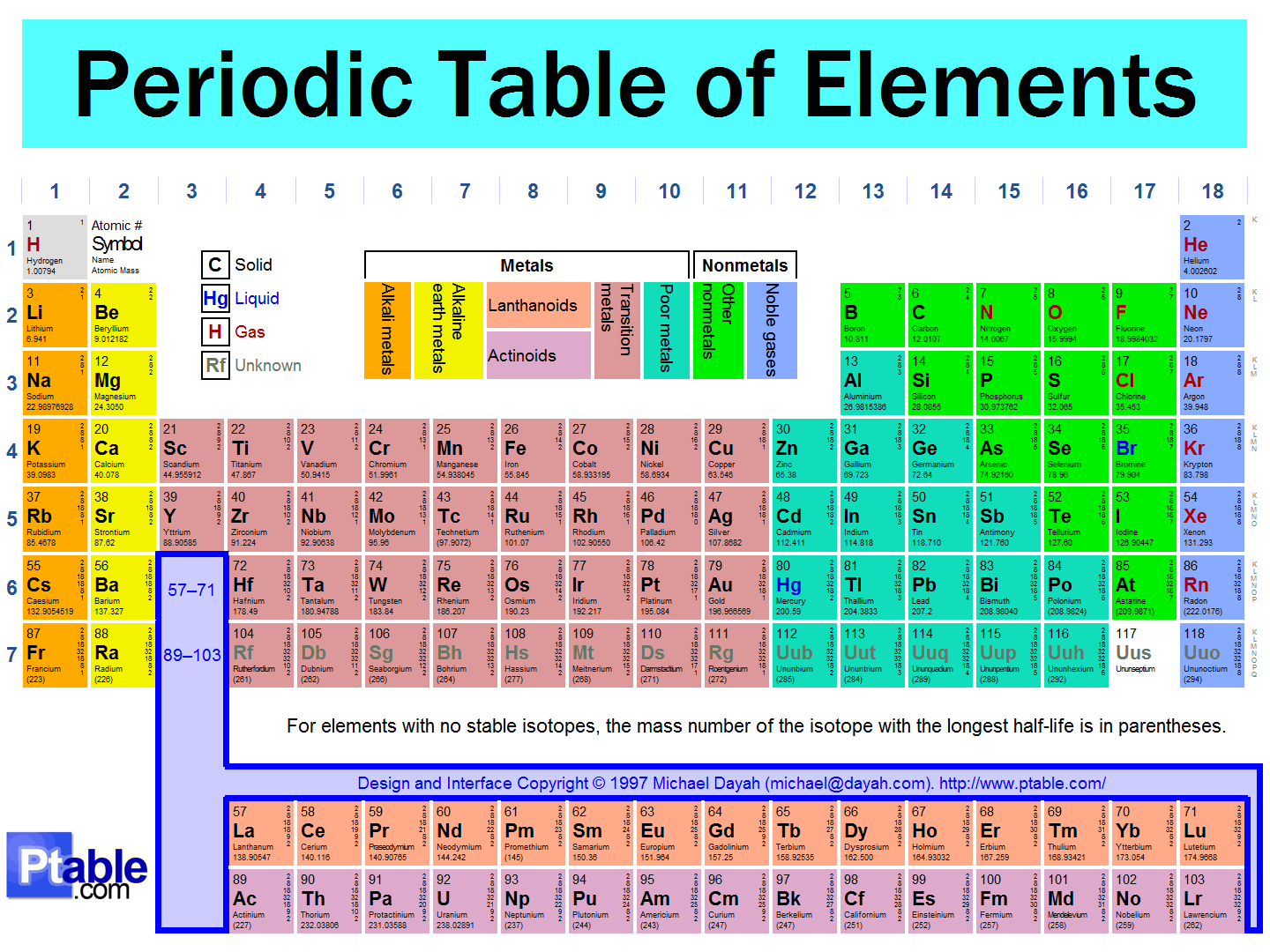
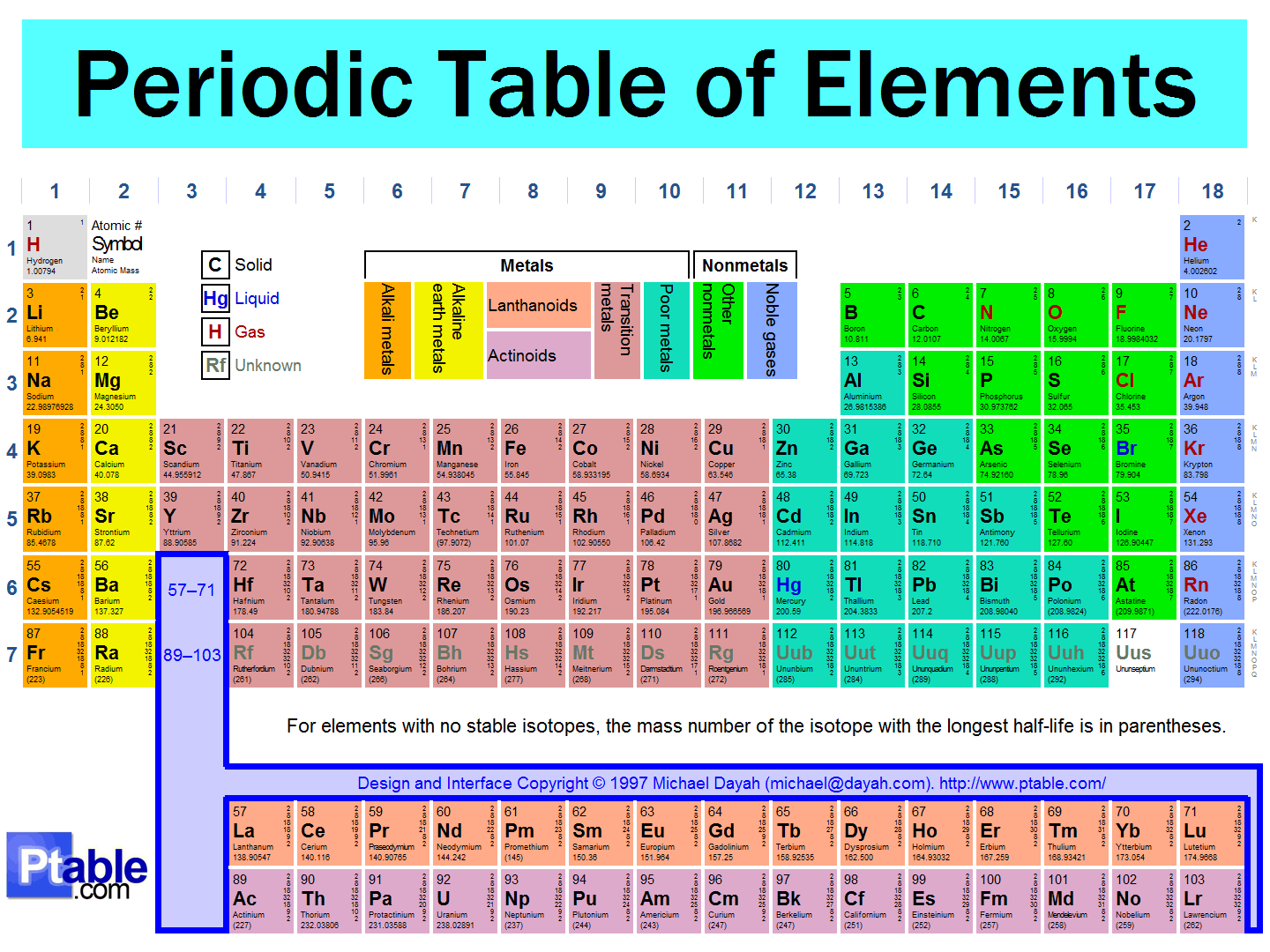
The Periodic Law and the Periodic Table
25
The periodic table lists all the elements, with information about their atomic weights, chemical symbols, and atomic numbers.
The arrangement of the periodic table leads us to visualize certain trends among the atoms.
The vertical columns (groups) of the periodic table are arranged such that all its elements have the same number of valence electrons.
All elements within a certain group thus share similar properties.
26

27

Periodic Table – Royal Society of Chemistry
You can open this webpage in a new tab.
28
Ions: Losing and Gaining Electrons
29
So far, we have discussed elements and compounds that are electrically neutral. They have the same number of electrons as protons, so the negative charges of the electrons are balanced by the positive charges of the protons.
However, this is not always the case. Electrons can move from one atom to another; when they do, species with overall electric charges are formed. Such species are called ions.
30
Species with overall positive charges are termed cations, while species with overall negative charges are called anions.
Remember that ions are formed only when electrons move from one atom to another; a proton never moves from one atom to another. Compounds formed from positive and negative ions are called ionic compounds.
31

32

33
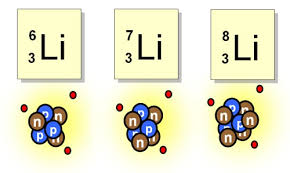
Isotopes
34
Isotopes are atoms of the same element that contain an identical number of protons, but a different number of neutrons.
Despite having different numbers of neutrons, isotopes of the same element have very similar physical properties. Some isotopes are unstable and will undergo radioactive decay to become other elements.
The predictable half-life of different decaying isotopes allows scientists to date material based on its isotopic composition, such as with Carbon-14 dating.
35

36

37
Atomic Mass: The Average Mass of an Element’s Atoms
38
An element can have differing numbers of neutrons in its nucleus, but it always has the same number of protons.
The versions of an element with different neutrons have different masses and are called isotopes.
The average atomic mass for an element is calculated by summing the masses of the element’s isotopes, each multiplied by its natural abundance on Earth.
When doing any mass calculations involving elements or compounds, always use average atomic mass, which can be found on the periodic table.
39
Calculating Average Atomic Mass
40
The average atomic mass of an element is the sum of the masses of its isotopes, each multiplied by its natural abundance (the decimal associated with the percent of atoms of that element that are of a given isotope).
Average atomic mass = f1M1 + f2M2 + … + fnMn where f is the fraction representing the natural abundance of the isotope and M is the mass number (weight) of the isotope.
41

42

43

44
Multiple Choice
45
Multiple Choice
Atom 2 has Mass= 14 Electrons=6
Are these atoms isotopes of each other or different elements?
46
Multiple Choice
Aluminum-27
An atom with 14 protons and 13 neutrons
47
Multiple Choice
48
Multiple Choice
49
Multiple Choice
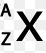
50
Multiple Choice

51
Multiple Choice

52
Multiple Choice

53
Multiple Choice
If an atom loses electrons, the charge will be positive.
true
false
54
Multiple Choice
What is the ion formed from Sulfur?
S+2
S-2
S+6
S-6
55
Multiple Choice
Which types of elements from anions?
metals
nonmetals
metalloids
noble gases
56
Multiple Choice
Which types of elements from cations?
metals
nonmetals
metalloids
noble gases
57
Multiple Choice
What is a negative ion called?
anion
cation
covalent
isotope
58
Multiple Choice
What is a positive ion called?
anion
cation
isotope
covalent
59
Multiple Choice

60
Multiple Choice
61
Multiple Choice
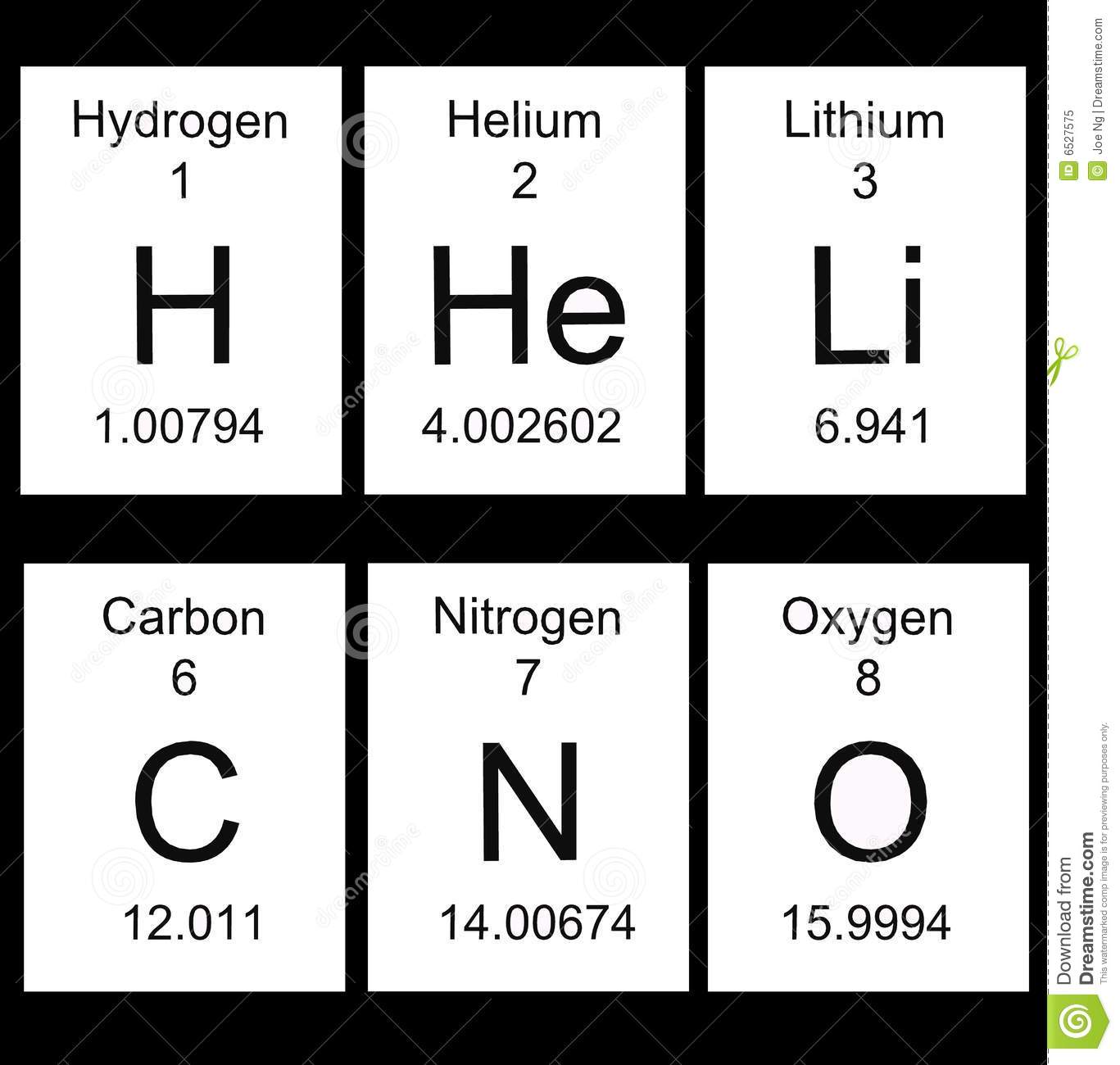
62
Multiple Choice
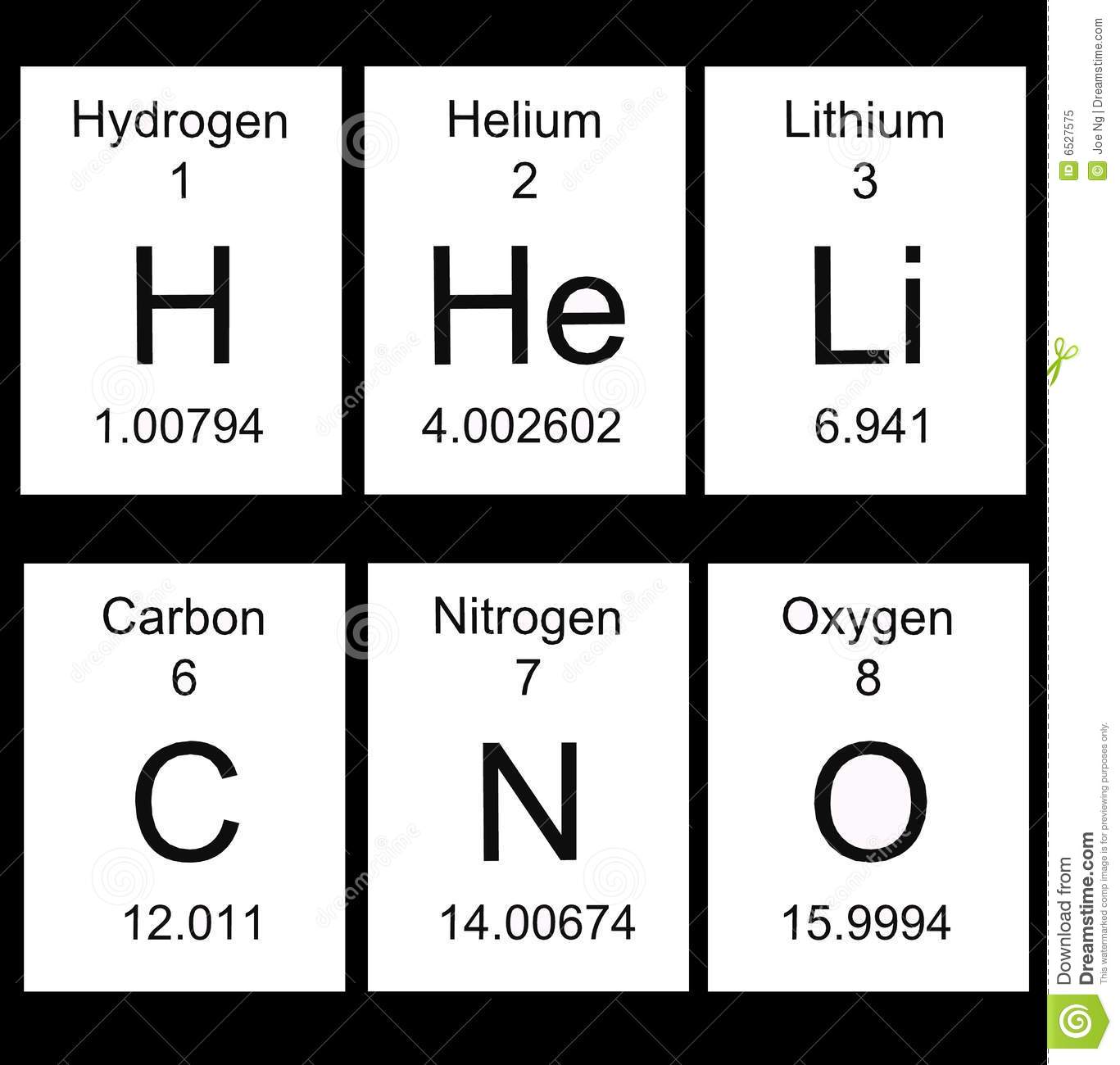
63
Multiple Choice
64
Multiple Choice
65
Multiple Choice
The atomic # tells us the # of ____________________ in an atom.
protons
electrons
neutrons
Cassietrons
66
Multiple Choice
What charge is the neutron?
no charge (neutral)
positive
negative
Katlyn-charged
67
Multiple Choice
What charge is the proton?
positive
no charge (neutral)
negative
Senaida-charged
68
Multiple Choice
Where are electrons found in an atom?
in the nuclues
electrons don't exist o_O
orbiting the nucleus
Only Josie knows where they are.
69
Multiple Choice
Where is the neutron found in the atom?
protons don't exist o_O
in the nucleus
orbiting the nucleus
Carleah has it!
70
Multiple Choice
Where is the proton found in an atom?
nucleus
orbiting around the nucleus
protons don't exist o_O
Justmir is hiding it.
71
Multiple Choice
72
Multiple Choice

73
Multiple Choice

74
Multiple Choice

75
Multiple Choice

76
Multiple Choice

How did Rutherford discover the proton?
Cathode Ray Tube
Gold Foil Experiment
Planetary Model
Plum Pudding Model
77
Multiple Choice

How did Rutherford discover the proton?
Cathode Ray Tube
Gold Foil Experiment
Planetary Model
Plum Pudding Model
78

Chapter 4. Atoms and Elements
Presenter: Dr. Luis Bello

Show answer
Auto Play
Slide 1 / 78
SLIDE
Similar Resources on Wayground

72 questions
At the airport
Presentation
•
University

72 questions
BASIC 01 REVIEW FINAL TEST CIL-LAEL
Presentation
•
University

74 questions
FRESHDESK QUIZ
Presentation
•
KG - University

74 questions
Cloning
Presentation
•
University

75 questions
Unit 5 lesson 3
Presentation
•
University

73 questions
First Conditional
Presentation
•
Professional Development

75 questions
Bahasa Inggris 1 - Week 08 and 09
Presentation
•
University
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Chemistry

20 questions
Guess The App
Quiz
•
KG - Professional Dev...

11 questions
NFL Football logos
Quiz
•
KG - Professional Dev...

19 questions
Minecraft
Quiz
•
6th Grade - Professio...

40 questions
8th Grade Math Review
Quiz
•
8th Grade - University

20 questions
Block Buster Movies
Quiz
•
10th Grade - Professi...

10 questions
Would you rather...
Quiz
•
KG - University

40 questions
Flags of the World
Quiz
•
KG - Professional Dev...

14 questions
Superhero
Quiz
•
1st Grade - University