

Law of Conservation of Mass
Presentation
•
Science
•
8th Grade
•
Medium
+9
Standards-aligned
Garfield Baker
Used 36+ times
FREE Resource
23 Slides • 30 Questions
1
Multiple Choice
2
Multiple Choice
What does the number 4 represent in 4NH3?
Subscript
Superscript
Coefficient
3
Multiple Choice
2H2 + O2 --> 2H2O
4
Multiple Choice
2H2 + O2 --> 2H2O
5
Multiple Choice
6
Multiple Choice
The coefficient in 3H2O is
7
Multiple Choice
Which are the reactants in the following reaction?
2Mg + O2 ---> 2MgO
8
Multiple Choice

If I burn this piece of paper what type of change is taking place
Physical
Chemical
9
What do you believe happen to the different things that made up the Hindenburg when it was burned ?
10

What do you believe happen to the different things that made up the Hindenburg when it was burned ?

11
Would the mass of remaining Ash from a burnt paper be the same as that of the paper burnt ?


12
Poll
Do you believe the mass that is not accounted for was destroyed by the fire ?
yes
no
13
During a chemical reaction matter is neither created or destroyed even though it might appear that way . Matter is changed into another form affecting the mass of the original reactants
14
Law of Conservation of Mass
by Garfield Baker
15
This is called the Law of Conservation of Mass. The Law of Conservation of Mass states that during a chemical Reaction mass is neither created or destroyed

16
In a chemical reaction the starting mass of the reactants must be the same as that of the products .

17
Open Ended
Why is it that mass of the product APPEARS to have being destroyed ?
18
The reason was that the water vapor and CO2 escaped in the air . It is not destroyed but just changed the state that it was in .
This happens in an OPENED SYSTEM
19
In an OPEN SYSTEM matter can enter or leave increasing or decreasing the mass of the final product . For example when baking soda and vinegar is mixed and the gas escaped that is an OPEN system

20
In a CLOSE SYSTEM no matter is able to enter in the reaction as a result the mass is not affected . For example if Vinegar and Baking Soda is mixed together and Covered no matter ( mass) will be added or lost ,because nothing will be able to enter or leave

21
Multiple Choice
22
Multiple Choice
23
Multiple Choice
24
Multiple Choice
What do we call the chemicals that result from a chemical reaction?
reactants
products
subscripts
coefficients
25
Multiple Choice
According to the Law of Conservation of Mass, what happens to the wood and oxygen when it is burned?
The wood disappears.
It stays the same chemically.
The atoms in the wood and oxygen rearrange and form smoke and ash.
Nothing.
26
Multiple Choice
The Law of Conservation of Mass States
Energy cannot be created nor destroyed, it can only change form.
Mass cannot be created nor destroyed, it can only change form.
Mass can be created or destroyed, it cannot change form.
27
Multiple Choice
28
Multiple Choice

29
Multiple Choice

30
Does the equation below that represent a chemical reaction shows the Law of Conservation of mass to be true ?

31
Balancing Equations
Because we know that the reactants should be the same as that of the products but is not the case in an OPEN system , Chemical Equations must be balanced
Balancing Equation satisfies the LAW of CONSERVATION OF MASS
To do this Coefficient are added before the elements or compounds
Coefficient are used to multiply the subscript (number of atoms ) of each elements until both sides are equal
32
Chose different elements and add keep adding coefficient until both sides are balanced (same). If there is a subscript when a Coefficient is added they will multiply together . Ex. there were 2 O atoms and a Coefficient of 11 was added making it 22

33
Make a Table to show how many atoms of each elements are present on either sides

This Equation is not balanced . There are 7 Carbon (C) atoms on left while there is 1 on the right. There are 16 Hydrogen(H) on the left while 2 are on the right . There are 2 Oxygen on the on the left while 3 on the right
34

Is this Equation Balanced ?
35
2 Al2O3-------- 4Al + 3O2
Reactant Product
Al = 4 Al = 4
O = 6 O= 6
36
Multiple Choice
Is the following equation balanced or unbalanced ?
N2 +3 H2 --> 2NH3
Balanced
Unbalanced
37
Multiple Choice
Al + O2 --> Al2O3
38
Multiple Choice
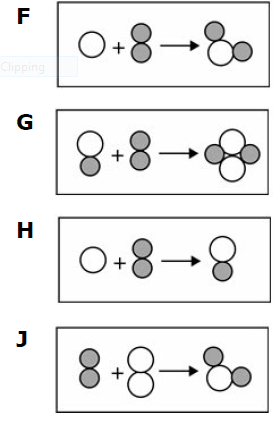
Which of the following models best demonstrates a balanced chemical equation?
F
G
H
J
39
Multiple Choice

40
Multiple Choice

41
Multiple Choice
42
How to balance a Chemical Equation ?

43

Balancing Chemical Equation - Google Slides
You can open this webpage in a new tab.
44

45

Reactants Product
Al - 2 Al - 1
O - 3 O - 2
46
Balance this equation
Li + Cl2 -> LiCl
47
Balance this Equation
MgCl2 --> Mg4+ Cl2
48
Complete 5 balancing equation problems
49
Multiple Choice
_CF4 + _Br2 -- _CBr4 + _F2
50
Multiple Choice
_N2 + _H2 --> _NH3
51
Multiple Choice
Balance this equation:
MgCl2 --> Mg4+ Cl2
It's already balanced.
4MgCl2 --> Mg4+ 4Cl2
4MgCl2 --> Mg4+ 5Cl2
52
Multiple Choice
Balance this equation:
2Li + Cl2 -> LiCl
2Li + Cl2 -> 4LiCl2
2Li + Cl2 -> LiCl2
2Li + Cl2 -> 2LiCl
53
Multiple Choice
Which chemical equation best represents the Law of Conservation of Mass?
H + O → H2O
H2 + O2 → H2O
2H2 + O2 → 2H2O
H2 + O2 → 2H2O2
Show answer
Auto Play
Slide 1 / 53
MULTIPLE CHOICE
Similar Resources on Wayground

44 questions
Interaction of Light Waves
Presentation
•
8th Grade

45 questions
PMA 1 Review: 8th Grade
Presentation
•
8th Grade

47 questions
EMS- 5.1 Sun's Intensity
Presentation
•
8th Grade

50 questions
Pure Substances and Mixtures, Pre-Summative Review, 2022
Presentation
•
8th Grade

50 questions
Intro to the Rock Cycle Lesson
Presentation
•
8th Grade

50 questions
Pure substances
Presentation
•
8th Grade

48 questions
Renewable and Nonrenewable Energy Sources
Presentation
•
8th Grade

47 questions
LT 5 HR Diagram Synchronous
Presentation
•
8th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

30 questions
GMAS Physical Science Review
Quiz
•
8th Grade

10 questions
Exploring the Rock Cycle
Interactive video
•
6th - 8th Grade

18 questions
Carbon Cycle
Quiz
•
8th Grade

10 questions
Exploring Earth's Seasons and Their Causes
Interactive video
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

10 questions
Evidence of Evolution
Presentation
•
6th - 8th Grade

20 questions
8th Grade Science NC EOG Review
Quiz
•
8th Grade