

Periodic Trends
Presentation
•
Chemistry
•
10th Grade
•
Medium
Oyerohunke oyediran
Used 6+ times
FREE Resource
16 Slides • 29 Questions
1

The Periodic Table:
Periodic Trends
2


• Trends are patterns seen in the periodic table.
• Trends are seen in mass, size, and reactivity.
Trends
3
Lets Review the Parts of The Periodic Table
You can do it!
4
Multiple Choice
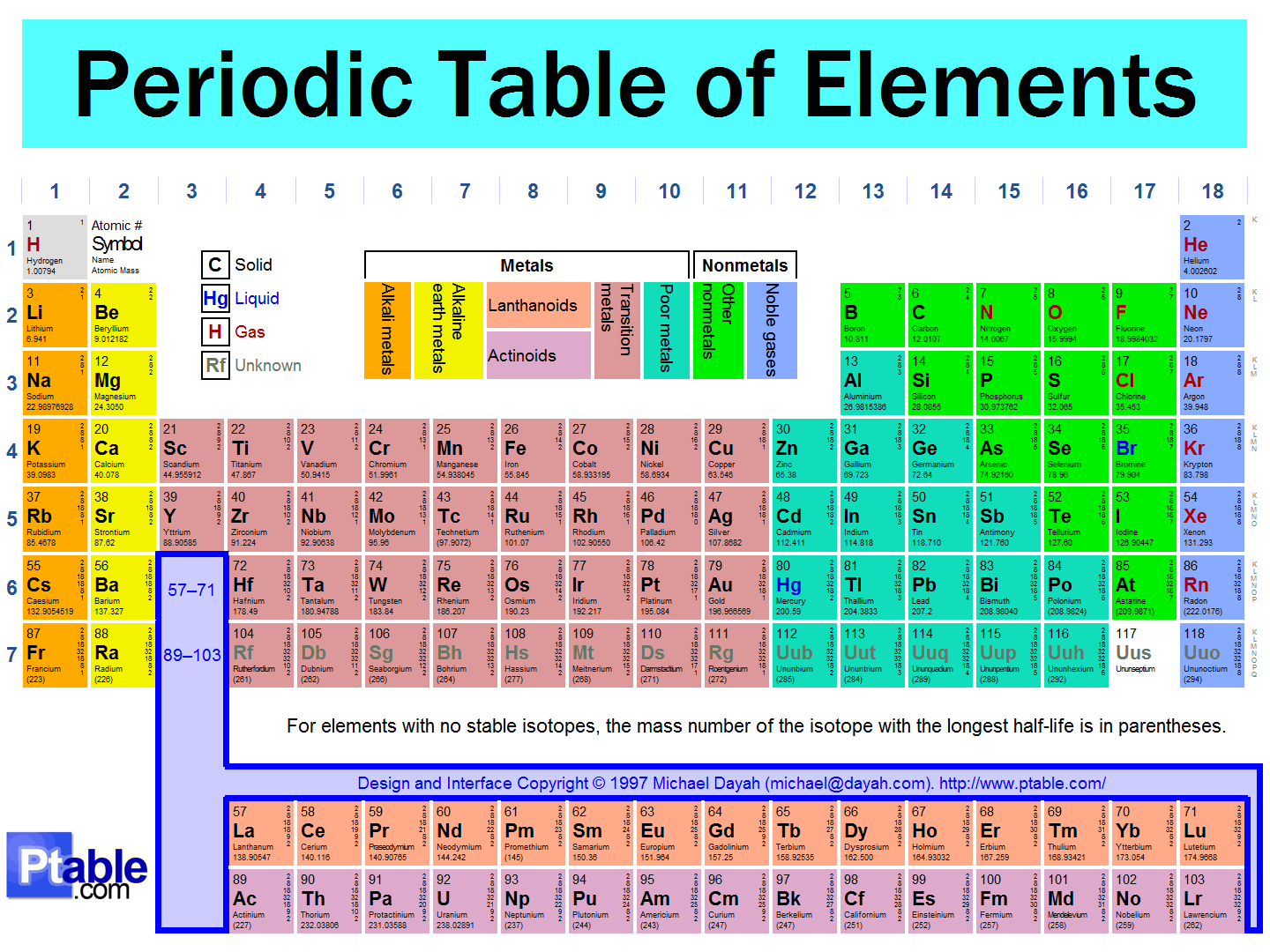
The vertical (up and down) columns in the Periodic Table are called
groups
towers
periods
atomic numbers
5
Multiple Choice
6
Multiple Choice

What group is element A in?
Alkali Metals
Alkali Earth Metals
Transition Metals
Halogens
7
Multiple Choice

What group is element B in?
Alkali Metals
Alkali Earth Metals
Transition Metals
Halogens
8
Multiple Choice

What group is element C in?
Alkali Metals
Alkali Earth Metals
Transition Metals
Halogens
9
Multiple Choice

What group is element D in?
Alkali Metals
Alkali Earth Metals
Transition Metals
Halogens
10
Multiple Choice

What group is element E in?
Halogens
Alkali Earth Metals
Transition Metals
Noble Gases
11
Multiple Choice
Most of the periodic table is mode of _______.
Metals
Non-metals
Semi-metals
12
Multiple Choice

13


Atomic Mass
The atomic mass of elements increases moving
down each group and right across each period.
Increasing Atomic Mass
14

We know that mass increases as the atomic number increases.....
Because there are more protons, it gets heavier.
Has nothing to do with the size.
Atomic mass depends on Protons and Neutrons.
15




• The atomic radius is the distance from
the valence electrons to the nucleus.
• Simply put, it describes the size of an atom.
Atomic Radius
Increasing Atomic Radius
Lithium
Sodium
Potassium
16



Atomic Radius
The atomic radius of elements increases moving
down each group and left across each period.
Increasing Atomic Radius
17
Multiple Choice
18
Multiple Choice

19
Multiple Choice
Of the halogens, which has the smallest radius?
F
Br
He
At
20
Multiple Choice
21
Multiple Choice

What property is being measured in this diagram?
Density
Ionization Energy
Atomic Radius
Atomic Mass
22
Multiple Choice

Which of these elements in group 1A has the largest atomic radius?
cesium
rubidium
potassium
sodium
23
Multiple Choice
24


• Ionization energy is the energy required to
remove an electron from an atom.
• The more tightly electrons are held by the
nucleus, the harder they are to remove.
Ionization Energy
Energy Needed
to Remove Electron
-
-
+
+
The smaller the size of the atom the more
25


Ionization Energy
The ionization energy of elements increases
moving up each group and right across a period.
Increasing Ionization Energy
Opposite of atomic radius
26
Multiple Choice
27
Multiple Choice

Which group of elements has the lowest ionization energies?
Alkali Metals (Group 1)
Alkaline Earth Metals (Group 2)
Halogens (Group 17)
Noble Gases (Group 18)
28
Multiple Choice

What is the amount of energy required to remove an electron from an atom?
atomic energy
ionization energy
ionic energy
electron energy
29
Multiple Choice
30
Multiple Choice
31
Multiple Choice
When across a period, the atomic size decreases so the first IE ___________.
increases
decreases
constant
i am not sure
32


• Electronegativity is a measure of how
strongly an atom is attracted to electrons.
• In bonds, electrons will be more attracted to
the atom with greater electronegativity.
Electronegativity
Attraction of electrons
to the nucleus
-
-
+
+
33


Electronegativity
The electronegativity of elements increases
moving up each group and right across a period.
Increasing Electronegativity
Same as
Ionization Energy
34
Multiple Choice
Which of the following will have a higher electronegativity than arsenic (As)?
Carbon (C)
Neon (Ne)
Antimony (Sb)
Germanium (Ge)
35
Multiple Choice

Which of the following halogens has the greatest electronegativity?
Chlorine (Cl)
Bromine (Br)
Iodine (I)
Astatine (At)
36
Multiple Choice

What is a measure of the tendency of an atom to attract a bonding pair of electrons when the atom is in a compound?
ionization energy
electropositivity
electronegativity
electron energy
37
Multiple Choice

Which element in period 4 has the highest electronegativity?
potassium
calcium
copper
bromine
38
Multiple Choice

What is a measure of the tendency of an atom to attract a bonding pair of electrons when the atom is in a compound?
ionization energy
electropositivity
electronegativity
electron energy
39


• Reactivity is the tendency of an atom or
molecule to undergo a chemical reaction.
• Certain elements react very easily with just
water. Other elements do not react at all.
• Note: Noble Gas elements (Group 18)
are not reactive. They are excluded.
Reactivity
40


Reactivity of Metals
The reactivity of metals increases moving
down each group and left across each period. Because it gets easier to remove electrons
Increasing Reactivity of Metals
Same as
atomic radius
41


Reactivity of Non-Metals
Increasing Reactivity of Non-Metals
The reactivity of non-metals increases moving
up each group and right across each period.
Because it is easier to gain electrons.
42


Reactivity of Non-Metals
• Fluorine is the most reactive non-metal.
• Francium is the most reactive metal.
F
Fr
9
F
Fluorine
18.99
87
Fr
Francium
223
43

44
Multiple Choice
What metal is most reactive
lithium
sodium
potassium
rubidium
45
Multiple Choice
What non-metal is the most reactive
iodine
sulfur
oxygen
fluorine

The Periodic Table:
Periodic Trends
Show answer
Auto Play
Slide 1 / 45
SLIDE
Similar Resources on Wayground

42 questions
Y4 Revision for EOY
Presentation
•
10th Grade

41 questions
Sentence Construction Errors
Presentation
•
10th Grade

39 questions
Only Americans use feets while world use meters.. SI
Presentation
•
10th Grade

42 questions
Density
Presentation
•
10th Grade

42 questions
10/14/20 Periodic Trends
Presentation
•
10th Grade

38 questions
Nuclear Chemistry Day 1
Presentation
•
10th Grade

39 questions
Naming Acids and Bases
Presentation
•
10th Grade

42 questions
Circle Vocabulary Part 2
Presentation
•
10th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade