

Counting Atoms in Formulas
Presentation
•
Science
•
8th Grade
•
Medium
+5
Standards-aligned
Deborah Miller
Used 29+ times
FREE Resource
8 Slides • 25 Questions
1
Counting Atoms in Formulas
Define the law and counting atoms in formulas.
2
Law of Conservation of Mass states that in a chemical reaction mass is not created or destroyed.
This means that the mass you start with (reactants) is the same as the mass you end with (products).
This law holds true whether it is a chemical or a physical change.
Make sure you copy the slide into your journal
Title for Notes: Counting Atoms in Formulas
3
Elements, Molecules and Compounds
Element: Any substance that cannot be broken down into a simpler substance, its a pure substance made up of only one type of atom.
Examples: H, Ne, F, B, He, O
Molecule: A particle made of two or more atoms bonded together.
Examples: O2, H2O, CO2, CH4, Cl2, H2, O3
Compound: A substance formed by two or more elements bonded together in a set ratio.
Examples: H2O, CO2, CH4, H2O2, C6H12O6
4
Multiple Choice
The Law of Conservation of Mass says that matter can neither be __________ or __________.
created; destroyed
recycled; renewed
subtracted; added
joined; separated
5
Multiple Choice

Table Salt (NaCl)
element
compound
mixture
6
Multiple Choice
many types of atoms
the periodic table
one type of atom
molecules
7
Multiple Choice

YES
NO
8
Multiple Choice
Which of the following cannot be broken down into something simpler?
a compound
an element
a chemical formula
a mixture
9
10


11
Subscripts
Subscripts are numbers that are written below and to the right of a chemical symbol. Subscripts tell you the number of atoms of an element. If an element does not have a subscript, it is understood that it is 1.
Formula | N |
Elements | # of Atoms |
|---|---|
N | 2 |
Total Atoms | 2 |
Elements | 1 |
Molecules | 1 |
Formula | C |
Elements | # of Atoms |
|---|---|
C | 6 |
H | 12 |
O | 6 |
Total Atoms | 24 |
Elements | 3 |
Compounds | 1 |
Formula | H |
Elements | # of Atoms |
|---|---|
H | 2 |
O | 1 |
Total Atoms | 3 |
Elements | 2 |
Compounds | 1 |
12
Coefficients
Coefficients are whole numbers that appear in front of a formula that tell how many compounds or elements you have. The distributive property is used - the coefficient is multiplied by the subscript of each element.
Example:
Formula | 4H |
Elements | #of Atoms |
|---|---|
H: 4x2 | 8 |
O: 4x1 | 4 |
Total Atoms | 12 |
Elements | 2 |
Compounds | 4 |
Formula | 3HCO |
Elements | #of Atoms |
|---|---|
H: 3x1 | 3 |
C: 3x1 | 3 |
O: 3x3 | 9 |
Total Atoms | 15 |
Elements | 3 |
Compounds | 3 |
Formula | 5H |
Elements | #of Atoms |
|---|---|
H: 5x2 | 10 |
P: 5x1 | 5 |
O: 5x4 | 20 |
Total Atoms | 35 |
Elements | 3 |
Compounds | 5 |
13
Multiple Choice
How many elements are in the compound Fe2O3?
0
1
2
3
14
Multiple Choice
How many atoms are present if there is no subscript following the element symbol?
0
2
only 1
1 million
15
Multiple Choice

How many atoms of Iron are in this compound?
1
2
3
4
16
Multiple Choice
How many different elements are present in H2COCH2
5
4
3
2
17
Multiple Choice
How many atoms are there TOTAL in H2SO4 ?
6
5
7
3
18
Multiple Choice
How many Magnesium atoms are in 10MgCl2?
10
5
20
19
Multiple Choice

How many total POTASSIUM atoms are in this formula?
8
2
4
12
20
Multiple Choice
three hydrogen (H)
one sulfur (S)
four oxygen (O)
H3SO4
HSO4
H4S3O
H2O
21
Multiple Choice
What does the subscript in a chemical formula tell you?
How many molecules are present
How many atoms are present
How many elements are present
How many neutrons are present
22
Multiple Choice
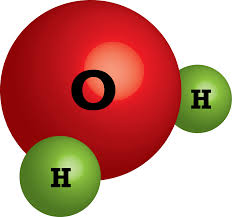
How many atoms of Hydrogen are in this compound?
1
2
3
4
23
Multiple Choice

subscripts
coefficients
I don't know, and don't want to try
None of the answers are correct
24
Multiple Choice

I don't know, and don't want to try
coefficients
subscripts
none of the answers are correct
25
Multiple Choice
How many elements are in this formula?
Li2O
1
2
3
4
26
Multiple Choice
Which of the following shows the elements with correct numbers of ATOMS for this molecule
H2SO4
H = 2, S = 4, O = 4
H = 2, S = 1, O = 4
H = 8, S = 4, O = 4
H = 2, SO = 4
27
Parenthesis in a formula are used to show that atoms in a particular structure are repeated in the molecule.
Parenthesis in a formula:
Formula | Al |
Elements | #of Atoms |
|---|---|
Al: 1x2 | 2 |
S: 1x1x3 | 3 |
O: 1x4x3 | 12 |
Total Atoms | 17 |
Elements | 3 |
Compounds | 1 |
Formula | 5(NH |
Elements | #of Atoms |
|---|---|
N: 5x1x2 | 10 |
H: 5x4x2 | 40 |
O: 5x1 | 5 |
Total Atoms | 55 |
Elements | 3 |
Compounds | 5 |
Formula | 2Ca |
Elements | #of Atoms |
|---|---|
Ca: 2x3 | 6 |
P: 2x1x2 | 4 |
O: 2x4x2 | 16 |
Total Atoms | 26 |
Elements | 3 |
Compounds | 2 |
28
Multiple Choice
C3H5(NO3)3
C = 3, H = 5, N = 3, O = 9
C = 3, H = 5, N = 1, O = 9
C = 9, H = 15, N = 3, O = 9
C = 9, H = 15, N = 1, O = 3
29
Multiple Choice
4
12
7
24
30
Multiple Choice
How many atoms of oxygen(O) are in
3Fe2O3
6
9
1
3
31
Multiple Choice
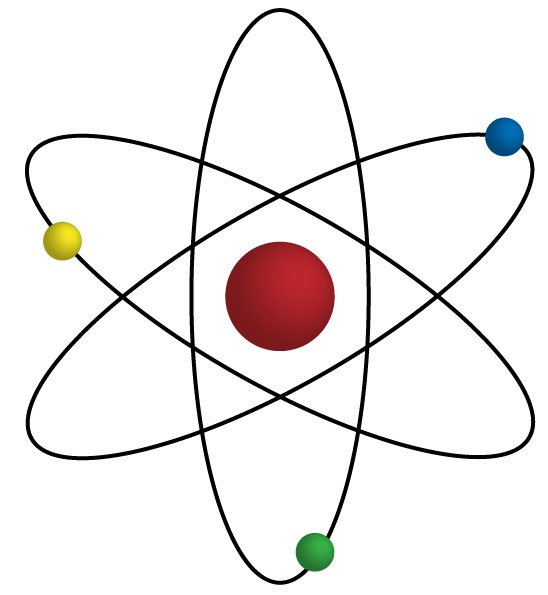
protons, neutrons, and isotopes
neutrons, isotopes, and electrons
positives, negatives, and electrons
protons, neutrons, and electrons
32
Multiple Choice
C3
O2
CH3CHO
H2
33
Multiple Choice
A ______ is a number written in front of a chemical formula.
Coefficient
Subscript
Exponent
Coordinator
Counting Atoms in Formulas
Define the law and counting atoms in formulas.
Show answer
Auto Play
Slide 1 / 33
SLIDE
Similar Resources on Wayground

27 questions
Plate Tectonics
Presentation
•
8th Grade

22 questions
Metals, Nonmetals and Metalloids
Presentation
•
8th Grade

26 questions
Ionic Bonds
Presentation
•
9th Grade

27 questions
Factoring Perfect Square Trinomials
Presentation
•
8th Grade

26 questions
STAAR 8TH SCIENCE REVIEW
Presentation
•
8th Grade

27 questions
Forces
Presentation
•
8th Grade

25 questions
Elements, Compounds, Mixtures
Presentation
•
8th Grade

25 questions
States of Matter Review
Presentation
•
8th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade

48 questions
8th Grade NC Science EOG Review 2
Quiz
•
8th Grade

19 questions
Renewable and Nonrenewable Resources (CK)
Quiz
•
8th Grade

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade

10 questions
Exploring Weather Influences and Map Reading
Interactive video
•
6th - 10th Grade

43 questions
Amplify Earth's Changing Climate Unit Review
Quiz
•
6th - 8th Grade