

KMT and Phase Change
Presentation
•
Science
•
10th Grade
•
Hard
+3
Standards-aligned
John Oglesby
FREE Resource
17 Slides • 17 Questions
1
Kinetic Molecular Theory

Review
2

The states of matter : All matter is composed of tiny particles that are always in motion.
The Kinetic Molecular Theory
3
Multiple Choice
The kinetic theory states
Particles won't move if you don't apply energy to it.
Particles only move in liquids and gases.
Particles are always in motion.
At the same temperature the object that has the most mass heats quicker.
4

Definition Shape
Definite Volume
Little movement
Example:
ICE
Solids
5
Multiple Choice
definite shape and indefinite volume
indefinite shape and indefinite volume
definite shape and definite volume
indefinite shape and definite volume
6

NO Definition Shape
Takes the shape of the container.
Definite Volume
Molecules have a "FLOW"
Medium Kinetic Energy
Example:
Water
Liquids
7
Multiple Choice
shape, volume
mass, area
smell, taste
volume, shape
8
Multiple Choice
mass
density
kinetic energy
potential energy
9

NO definition Shape
Takes the shape of the container.
NO definite Volume
Fills the volume of the container
Molecules move FAST and Random
High Kinetic Energy
Example:
Steam
Gas
10
Change the TEMPERATURE!
How do we move from one state to the next?

High Kinetic Energy
Low Kinetic Energy
11
Multiple Choice
Kinetic Theory
Kinetic energy
Potential energy
Electric energy
12
Multiple Choice
Gas
Liquid
Solid
Plasma
13
Multiple Choice
Gas
Liquid
Solid
Plasma
14
Multiple Choice
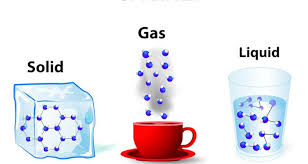
Solid
Liquid
Gas
15
Multiple Choice
As the _________________ of a gas increases, the kinetic energy of the particles increases. The particles move farther apart, and volume increases.
speed
height
temperature
area
16
Multiple Choice
Gases assume the volume and shape of their containers
TRUE
FALSE
17
Multiple Choice

State of matter that can fill containers with no fixed shape or volume.
Solid state
Liquid state
Gas state
Plasma state
18
Multiple Choice

State of matter in which materials have a definite shape and volume.
Solid state
Liquid state
Gas state
Plasma state
19
Energy & Phase Changes
Phase Change: the transformation from one state of matter to another
Adding energy to a material increases the temperature of that material
At certain points, the temperature stops increasing while the substance changes state
Energy is used to either break or form intermolecular bonds
Experience Chemistry | Lesson 4.2

20
Types of Phase Changes
Some types of phase changes we experience on a daily basis
Solid → Liquid= Melting
Liquid → Solid= Freezing
Liquid → Gas= Vaporization
Gas → Liquid= Condensation
Other types are less common in our everyday lives
Solid →Gas= Sublimation
Gas → Solid= Deposition
The properties of a material can change at different states even if the particles are the same
Experience Chemistry | Lesson 4.2
21
Heating a Liquid
In the liquid state, particles have vibrational movement and rotational movement
still held together by intermolecular forces
Heating a liquid adds energy to the particles, allowing them to break those forces holding them together and become a gas
Boiling Point: the temperature at which intermolecular forces break in a liquid
Can be predicted by looking at intermolecular forces
Stronger forces have higher boiling points
Experience Chemistry | Lesson 4.2
22
Evaporation
A liquid doesn't have to boil for vaporization to occur
Below the boiling point, most particles do not have enough kinetic energy to vaporize into a gas, but a few do
Every Particle has different energy levels
Evaporation: The process by which particles at the surface of a liquid have enough energy to escape intermolecular forces and enter the gas phase
Increasing the temperature can increase the evaporation rate, even if it is not raised to the boiling point
Experience Chemistry | Lesson 4.2
23
Condensation
In the process of condensation, some particles have a lower energy level, that allows them to enter the liquid phase
Cooling the temperature reduces the energy or particles, which allows particles to return to a liquid state
Experience Chemistry | Lesson 4.2

24
Vapor Pressure
When Liquid is in a container, some particles will evaporate
When those particles collide with the wall of the container, it produces pressure on the container
Vapor Pressure: the pressure of gas above a liquid
Vapor pressure leads to a an equilibrium, where some particles evaporate while others condenses
Experience Chemistry | Lesson 4.2

25
Vapor Pressure and Boiling
Boiling point is the temperature of where the vapor pressure is equal to the external pressure on a fluid
In a system of variable vapor pressure, different factors affect the boiling point of the liquid
Pressure: At sea level, warmer temperatures are needed to reach the boiling point
At higher altitudes with lower pressure, liquids boils at a lower temperature
At higher pressures, liquids boil at higher temperatures
Experience Chemistry | Lesson 4.2
26
Heating a Solid
particles in a solid gain vibrational energy
With enough energy, the particles can no longer contain it, and break free from each other
As they break free, they change to a liquid phase
Melting Point: the temperature at which a solid becomes a liquid
Experience Chemistry | Lesson 4.2

27
Sublimation
Solids generally melt into a liquid at a gradual temperature change
In some cases, solids change to the vapor state without passing through the liquid phase
Sublimation: The change from a solid to a gas state
Occurs at STP with substances that have extremely weak intermolecular bonds
Gives some solids vapor pressure
Experience Chemistry | Lesson 4.2

28
Match
Match the phase change to it's name
Condensation
Vaporization
Melting
Freezing
Gas → liquid
Liquid → Gas
Solid → Liquid
Liquid → Solid
Gas → liquid
Liquid → Gas
Solid → Liquid
Liquid → Solid
29

30
Multiple Choice

What state of matter would this substance be between points A & B?
Solid
Liquid
Gas
31
Multiple Choice

What state of matter would this substance be between points C & D?
Solid
Liquid
Gas
32
Multiple Choice
When does condensation occur?
When liquid water changes into water vapour
When water vapour changes into liquid water
33
Multiple Choice
melting
boiling
Schaffer
pressure
34
Phase Change Lab
Some text here about the topic of discussion
Kinetic Molecular Theory

Review
Show answer
Auto Play
Slide 1 / 34
SLIDE
Similar Resources on Wayground

30 questions
Unit 5 Concept 1 Balancing Chemical Equations
Presentation
•
10th Grade

24 questions
Concave and Convex Mirrors
Presentation
•
10th Grade

29 questions
Unusual sports from around the world
Presentation
•
10th Grade

27 questions
World War 1
Presentation
•
10th Grade

27 questions
Human reproductive systems
Presentation
•
10th Grade

28 questions
Cells Theory, Microscopes, and Types of Cells
Presentation
•
10th Grade

27 questions
Groundwater
Presentation
•
9th - 10th Grade

28 questions
Section 2 Lesson 5-Empirical Formulas
Presentation
•
10th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade

49 questions
AP Environmental Science Final Exam Review
Quiz
•
10th Grade - University

10 questions
Exploring Chemical and Physical Changes
Interactive video
•
6th - 10th Grade

10 questions
Exploring Animal Adaptations: Physical, Behavioral, and Life Cycle Changes
Interactive video
•
6th - 10th Grade