

1a States of Matter
Presentation
•
Science
•
8th Grade
•
Medium
Michelle Zou
Used 19+ times
FREE Resource
17 Slides • 28 Questions
1
1a States of Matter
by Ms Michelle Zou
2

3
Match
solid
liquid
gas
solid
liquid
gas
4
Drag and Drop
5
Drag and Drop
6
Drag and Drop
7

8
Multiple Choice
What is the process called when something goes directly from a solid to a gas?
sublimation
condensation
melting
deposition
9
Multiple Choice
What is the phase change of a solid to a liquid?
freezing
melting
boiling
condensation
10
Multiple Choice

evaporation
precipitation
condensation
sublimation
11

12
Multiple Choice

In a Kool-Aid solution, what is the solute?
The powder
The water
The mix of both of them
13
Multiple Choice

In a Kool-Aid solution, what is the solvent?
The powder
The water
The mix of both of them
14
Multiple Choice
the ability to catch on fire
the ability of a substance to dissolve
the ability to fly.
the ability to get on Mrs. D's nerves.
15


16

17

18
Multiple Choice
melting point
boiling point
19
Multiple Choice
A substance has a melting point of 25 oC and a boiling point of 235 oC. What state is it at 5oC?
Solid
Liquid
Gas
20
Multiple Choice
100 degrees Celsius
0 degrees Celsius
0 degrees Fahrenheit
21

22

23

24
Multiple Choice

If the boiling point of water is 100 degrees celsius, what is the state of matter of water at 115 degrees celsius?
Solid
Liquid
Gas
Plasma
25
What is Diffusion?
Diffusion is the movement of a substance from an area of high concentration to an area of lower concentration .

26
Diffusion occurs in liquids and gases when their particles collide randomly and spread out. Diffusion is an important process for living things - it is how substances move in and out of cells.

27

Diffusion in everyday life

28
Factors affecting diffusion
The state of matter
Temperature
Consentration gradient
29
Multiple Choice
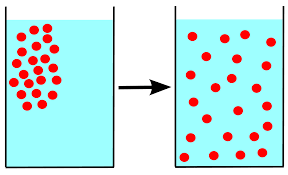
The picture below is an example of _________________.
osmosis
isotonic
diffusion
active transport
30
Multiple Choice
What is DIFFUSION?
31
Multiple Choice
Diffusion occurs only in solids
True
False
32
Multiple Choice
Diffusion is the free movement of particles from an area of __________ concentration to an area of ________ concentration.
High to high
High to low
Low to low
Low to high
33
Multiple Choice

Two gas jars are set up as shown.
The lid is removed and the gas jars are left to stand. After some time the contents of both gas jars are brown. Which process causes this to happen?
condensation
diffusion
evaporation
filtration
34
Multiple Choice

Hydrogen chloride gas, HCl, reacts with ammonia gas, NH3, to form solid ammonium chloride. The apparatus is set up as shown. After a few minutes, solid ammonium chloride forms where the two gases meet.
A
B
C
D
35
Multiple Choice

Ammonia gas is reacted with hydrogen chloride gas using the apparatus shown. Solid ammonium chloride is produced
Ammonia solution is a base and hydrogen chloride solution is an acid.
Ammonia molecules diffuse more slowly than hydrogen chloride molecules.
Hydrogen chloride has a greater molecular mass than ammonia.
Hydrogen chloride moves by Brownian motion.
36

37
Multiple Choice
Solutions are composed of __________ .
salts and solutes
solvents and salts
solutes and alloys
solutes and solvents
38

39
Multiple Choice

When 42 grams of potassium chloride, is dissolved in 100 grams of water at 50 ºC, the solution can be correctly described as:
supersaturated
saturated
unsaturated
40
Multiple Choice

When 20 grams of potassium chlorate, KClO3, is dissolved in 100 grams of water at 80 ºC, the solution can be correctly described as:
supersaturated
saturated
unsaturated
41
Multiple Choice

Which solute is the most soluble at 10 ⁰C?
KI
KClO3
NH4Cl
NH3
42
Multiple Choice

Which solute is the least soluble at 90 ⁰C?
SO2
KClO3
KI
HCl
43
Multiple Choice

How many grams of K2Cr2O7, are soluble in 100 g of water at 90 ºC?
85 grams
70 grams
40 grams
15 grams
44
Multiple Choice

25 grams of NaCl are dissolved in 100 mL of water at 60 C. How many more grams need to be dissolved for the solution to be saturated?
13 grams
25 grams
6 grams
38 grams
45

1a States of Matter
by Ms Michelle Zou
Show answer
Auto Play
Slide 1 / 45
SLIDE
Similar Resources on Wayground

39 questions
Roller Coaster Engineering
Presentation
•
8th Grade

38 questions
sound waves
Presentation
•
7th - 9th Grade

39 questions
Intro to Robotics
Presentation
•
8th Grade

42 questions
Food Chains, Food Webs & Energy Pyramids
Presentation
•
8th Grade

37 questions
State of Matter/Phase Changes
Presentation
•
8th Grade

36 questions
Ecology
Presentation
•
8th Grade

37 questions
Force and Motion Review
Presentation
•
7th - 8th Grade

38 questions
Ecosystem Interdependence Notes
Presentation
•
8th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

30 questions
GMAS Physical Science Review
Quiz
•
8th Grade

10 questions
Exploring the Rock Cycle
Interactive video
•
6th - 8th Grade

18 questions
Carbon Cycle
Quiz
•
8th Grade

10 questions
Exploring Earth's Seasons and Their Causes
Interactive video
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

10 questions
Evidence of Evolution
Presentation
•
6th - 8th Grade

20 questions
8th Grade Science NC EOG Review
Quiz
•
8th Grade