

State of Matter/Phase Changes
Presentation
•
Science
•
8th Grade
•
Easy
+9
Standards-aligned
Jennifer Napolitano
Used 32+ times
FREE Resource
13 Slides • 24 Questions
1

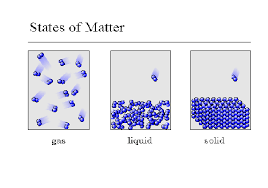
States of Matter
2

Kinetic Theory of Matter
Molecules are always moving and have kinetic energy.
The state of the matter (solid, liquid, or gas) depends on the particles' kinetic energy, which increases with temperature, causing them to move faster and spread farther apart.
2
3

Solids
●
Molecules are very close together
●
Molecules vibrate (move) slowly
●
Matter has a definite shape and volume
4

Liquids
●
Molecules are loosely packed
together.
●
Molecules can flow and move with
greater speed than a solid.
●
Matter has a definite volume but NOT a definite shape
5

Gas
●
Molecules are far apart.
●
Molecules can move very quickly.
●
Matter has NO definite shape or volume
6

Changing States of Matter
Changes in states of matter between solids, liquids and gases occur when objects are heated and cooled.
Watch the Video to learn more!
9
7

⦁Occurs when a SOLID
turns into a LIQUID.
⦁Heat is ADDED to
system.
Melting
11
8

⦁Occurs when a LIQUID
turns into a GAS.
⦁Heat is ADDED to
system.
Vaporization (Boiling)
12
9

⦁Occurs when a LIQUID
turns into a SOLID.
⦁Heat is REMOVED from
system.
Freezing
14
10

⦁Occurs when a GAS
turns into a LIQUID.
⦁Heat is REMOVED from
system.
Condensation
15
11

Graphing of
Changes of States
of Matter
19
12

A phase change diagram shows the relationship between temperature, heat energy and phase changes in matter.
Energy is added as you move from a SOLID to a GAS. It goes through the processes of melting and vaporization.
Energy is removed as you move from a GAS to a SOLID. It goes through the processes of condensation and freezing.
Phase Change Diagram
13
Drag and Drop

In this phase change the temperature
14
Drag and Drop

In this phase change the temperature
15
Drag and Drop

In this phase change the temperature
16
Drag and Drop

In this phase change the temperature
17

Let's Practice!
19
The practice questions are GRADED!
Use your notes to help you with the questions.
Take advantage of multiple attempts to earn the most points!
18
Multiple Choice
19
Multiple Choice
Which form of matter does not take the shape of its container and the particles move quickly?
20
Multiple Choice
Particles are far apart and move fast.
Particles are close together and move fast.
Particles are flowing and move fast.
Particles are far apart and move slowly.
21
Multiple Choice

Particles of a liquid
are tightly packed together and stay in a fixed position.
are free to move around one another but still touch.
22
Multiple Choice
The slower the particles in a substance move,
the colder it is.
the warmer it is.
the more energy it has.
23
Multiple Choice
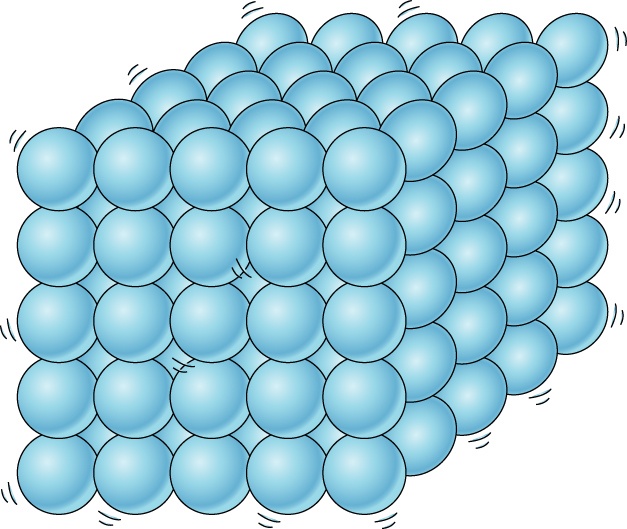
What state of matter?
Solid
Liquid
Gas
Plasma!!!
24
Multiple Choice
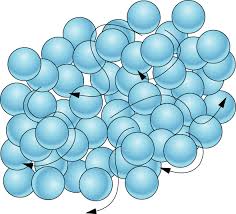
What state of matter?
Solid
Liquid
Gas
25
Multiple Choice
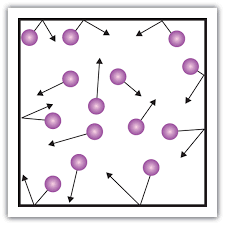
What state of matter?
Solid
Liquid
Gas
26
Multiple Choice
27
Multiple Choice
What is the phase change of a solid to a liquid?
freezing
melting
vaporization
condensation
28
Multiple Choice
What is it called when a solid turns into a liquid?
Melting
Freezing
29
Multiple Choice
All phase changes (changing states) requires energy to be added or taken away.
30
Multiple Choice
Change from Gas to Liquid is called........
Melting
Condensing
Freezing
Vaporizing
31
Multiple Choice
Change from Liquid to Solid is called ...
fusion
Freezing
sublimation
evaporation
32
Multiple Choice
33
Multiple Choice
Change from liquid to gas is called....
expansion
evaporation/vaporization
condensation
melting
34
Multiple Choice

In the phase change of freezing is energy being Added or Removed?
Removed
Added
35
Multiple Choice

What is happening to the temperature after a Gas turns into a Liquid?
Temperature Increases
Temperature Decreases
36
Multiple Choice

Is energy being added or removed during the vaporization process?
Energy is added
Energy is removed
37
Multiple Choice
Are there molecules moving in ice?
yes, they are moving slow
no, it is frozen so molecules don't move
yes, they are moving quickly
no, ice is too cold for movement

States of Matter
Show answer
Auto Play
Slide 1 / 37
SLIDE
Similar Resources on Wayground

31 questions
Solutions: Elements, Compounds, and Mixtures
Presentation
•
8th - 9th Grade

33 questions
Amplify Magnetic Fields Ch 1 L 1.5
Presentation
•
8th Grade

32 questions
Rigid Transformation Practice
Presentation
•
8th Grade

26 questions
Ecology Review
Presentation
•
8th Grade

33 questions
Atoms and the periodic table
Presentation
•
8th Grade

34 questions
Carbon, Nitrogen, and Oxygen Cycles
Presentation
•
8th Grade

28 questions
Moon Phases and Tides
Presentation
•
8th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

12 questions
PS.8.1.2, PS.8.1.3, PS.8.1.5 Review
Quiz
•
8th Grade

20 questions
Moon Phases and Eclipses
Quiz
•
6th - 8th Grade

22 questions
6th & 8th Grade Science Material SOL Review
Quiz
•
6th - 8th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade

48 questions
8th Grade NC Science EOG Review 2
Quiz
•
8th Grade

19 questions
Renewable and Nonrenewable Resources (CK)
Quiz
•
8th Grade

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade