

Kinetics Review 2
Presentation
•
Science
•
10th Grade
•
Easy
+1
Standards-aligned
Linda Cannon
Used 6+ times
FREE Resource
0 Slides • 31 Questions
1
Multiple Choice
Why does a higher concentration increase the rate of reaction?
it increases the amount of reactants
it lowers the activation energy
it increases the energy of reactant particles
it increases the frequency of collision
2
Multiple Choice
How fast a reaction is
How big a reaction is
How loud a reaction is
How much gas a reaction produces
3
Multiple Choice
Grinding an effervescent tablet into powder increases the rate of reaction due to increase of
concentration
total surface area
temperature
size
4
Multiple Choice
Increasing pressure causes the reacting particles
to bond together
to repel from each other
gain more kinetic energy
to move closer together
5
Multiple Choice
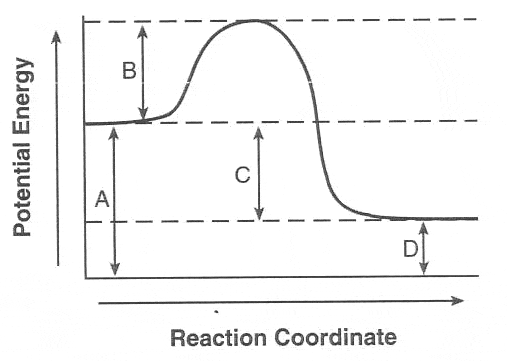
A
B
C
D
6
Multiple Choice
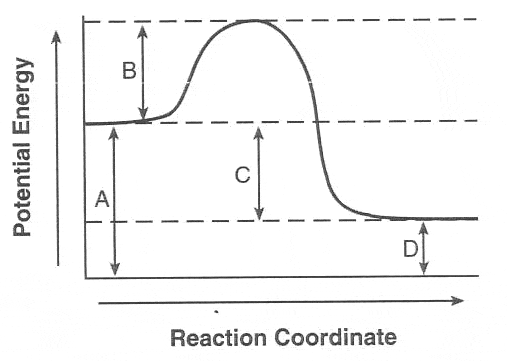
Endothermic
Exothermic
7
Multiple Choice
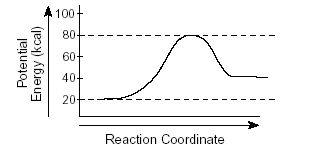
What is the ΔH of this reaction?
40 kJ
20 kJ
80 kJ
-60 kJ
8
Multiple Choice
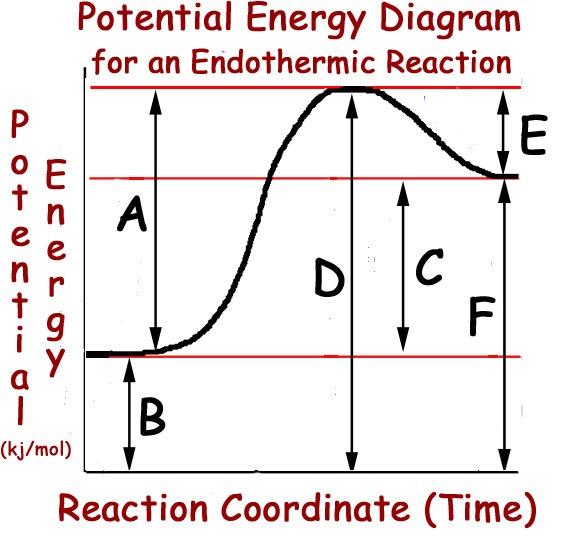
A
B
C
D
9
Multiple Choice

100 kJ
175 kJ
50 kJ
75 kJ
10
Multiple Choice
True
False
11
Multiple Choice

This symbol indicates that a reaction is _____
reversible
irreversible
12
Multiple Choice
What are the two factors that help determine that equilibrium is reached?
Forward reaction rate is faster than the reverse and the concentrations are equal
Forward and reverse reaction rates are equal and concentrations are equal
Forward and reverse reaction rates are equal and concentrations are constant
13
Multiple Choice
changing the order of the reaction
increasing the temperature
lowering the activation energy
making the activated complex
14
Multiple Choice
If the temperature is reduced, a reaction rate will ______
increase
decrease
stay the same
15
Multiple Choice
If the concentration of a reactant is increased, the reaction rate will ___________.
increase
decrease
stay the same
16
Multiple Choice
catalyst
product
reactant
solute
17
Multiple Choice
Slow collision frequency
Allow less effective collision between the particles
Cause particles to lose speed
Increase collision between the particles thus increasing the rate.
18
Multiple Choice

Concentration
Pressure
Temperature
States of matter
19
Multiple Choice
2SO2(g)+O2(g) ⇌ 2SO3(g) + Heat
Adding SO2(g) will
shift equilibrium right
shift equilibrium left
increase rate of reaction
have no change
20
Multiple Choice
2SO2(g)+O2(g) ⇌ 2SO3(g) + Heat
Increasing the temperature will...
shift equilibrium right
shift equilibrium left
increase pressure
have no change
21
Multiple Choice
2SO2(g)+1O2(g) ⇌ 2SO3(g) + Heat
Increasing the pressure on the system will...
shift equilibrium right
shift equilibrium left
slow rate of reaction
have no change
22
Multiple Choice
2SO2(g) + O2(g) ⇌ 2SO3(g) + Heat
Using a catalyst
shift equilibrium right
shift equilibrium left
increase the rate of reaction
have no change
23
Multiple Choice
temperature
concentration
particle size (surface area)
All of these affect reaction rates
24
Multiple Choice
the particle size of the reactants are larger.
temperature is decreased.
concentration of the reactants are increased.
the reaction is not stirred.
25
Multiple Choice
catalyst
product
reactant
solute
26
Multiple Choice
Catalysts permit reactions to proceed along a ___________energy path.
lower
higher
magnetic
psycho's
27
Multiple Choice
Smaller particle size allows for a _________ surface area to be exposed for the reaction.
larger
smaller
rectangular
spherical
28
Multiple Choice
________ is the measure of how much area of an
object is exposed.
Surface Area
Catalyst
Temperature
Concentration
29
Multiple Choice
Grains of sugar have a greater _______ than a solid cube of
sugar of the same mass, and therefore will dissolve quicker in water.
Surface Area
Catalyst
Temperature
Concentratrion
30
Multiple Select
Pick the TWO (2) options that will INCREASE the rate of a reaction.
Reducing Heat
Adding Catalyst
Adding Heat
Removing Catalyst
31
Multiple Choice
Which of the following will lower the rate
of reaction?
adding an enzyme to the reaction
decreasing the temperature from 40°C to
10°C
breaking a chunk of calcium up into
smaller pieces
increasing the amount of solute
dissolved in a solution
Why does a higher concentration increase the rate of reaction?
it increases the amount of reactants
it lowers the activation energy
it increases the energy of reactant particles
it increases the frequency of collision
Show answer
Auto Play
Slide 1 / 31
MULTIPLE CHOICE
Similar Resources on Wayground

21 questions
Net Force, Inertia, and Friction
Presentation
•
10th Grade

26 questions
Cellular Respiration
Presentation
•
10th - 11th Grade

20 questions
States of Matter Review
Presentation
•
3rd Grade

23 questions
Precipitation Reactions
Presentation
•
10th Grade

25 questions
Scientific Method Review
Presentation
•
9th Grade

25 questions
Human Impact
Presentation
•
10th Grade

23 questions
Population Pyramids
Presentation
•
10th Grade

24 questions
Taxonomy
Presentation
•
10th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

11 questions
EOC Domain 1: Cells
Presentation
•
9th - 10th Grade

14 questions
EOC Domain 2: Genetics
Presentation
•
9th - 10th Grade

10 questions
Exploring the Human Respiratory System
Interactive video
•
6th - 10th Grade

10 questions
Exploring Newton's Laws of Motion
Interactive video
•
6th - 10th Grade

10 questions
Exploring the Formation of the Solar System
Interactive video
•
6th - 10th Grade