

Chemical Bonds
Presentation
•
Chemistry
•
10th Grade
•
Medium
Janice Taylor
Used 8+ times
FREE Resource
12 Slides • 84 Questions
1

CHEMICAL BONDS
2


1.Why do atoms form bonds?
2.What are the three types of chemical bonds?
3.Which group of elements do not form bonds?
3

1.Why do atoms form bonds? Atoms form chemical bonds
to become stable and have a full outer shell.
2.What are the three types of chemical bonds?
Ionic, covalent and metallic.
3.Which group of elements do not form bonds?
Group zero - the noble gases

4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
Why do atoms bond?
to find a date
to become bigger
to go to dog jam
to become stable
7
Multiple Choice
When atoms bond it is called a ___________________ because they make a new substance.
chemical reaction
physical reaction
8

9

10

11
Multiple Choice
12
Multiple Choice
an element from Group 7
an element with atoms that have eight electrons in its outer shell
13
Multiple Choice
14
Multiple Choice
15
Multiple Choice

Positive ions are formed when atoms _____________ electrons.
lose
gain
16
Multiple Choice

Negative ions are formed when atoms _____________ electrons.
lose
gain
17
Multiple Choice
When an atom loses an electron, it becomes a
positive ion
negative ion
neutral ion
neutral atom
18
Multiple Choice
Why do all bonds form?
so the number of protons equals the number of electrons
so an atom can become unstable
to fill the outermost energy level
19
Multiple Choice
20
Multiple Choice
21
Multiple Choice
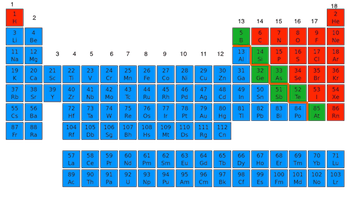
22
Multiple Choice
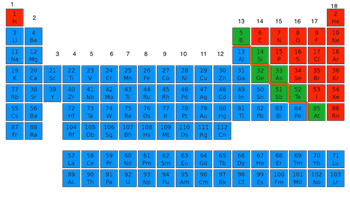
23
Multiple Choice

What type of elements form cations?
metals
nonmetals
metalloids?
24
Multiple Choice
Anions
Gain electrons, has an overall positive charge
Lose electrons, has an overall positive charge
Lose electrons, has an overall negative charge
Gain electrons, has an overall negative charge
25
Multiple Choice
How does calcium become an calcium ion?
it loses 2 electrons
it gains 2 electrons
26
Multiple Choice
How does magnesium become an magnesium ion?
it loses 2 electrons
it gains 2 electrons
27
Multiple Choice
How does oxygen become an oxide ion?
it loses 2 electrons
it gains 2 electrons
28
Multiple Choice
29

30

31
Multiple Choice
32
Multiple Choice
33
Multiple Choice

34
Multiple Choice
35
Multiple Choice
36
Multiple Choice
37
Multiple Choice
38
Multiple Choice
39
Multiple Choice
Why do atoms share electrons?
to increase their atomic number
to become ions
to obtain noble gas electron configuration
to become less polar
40
Multiple Choice
In covalent bonds electrons are:
shared
transferred
mobile
41
Multiple Choice
Which bond type is more likely to be liquids or gases at room temperature?
ionic bonds
covalent bonds
metallic bonds
42
Multiple Choice
Which of the following compounds is considered a covalent compound?
NH3
NaCl
MgO
K2S
43
Multiple Choice
Ice, H2O(s), is classified as
an ionic compound
a molecular compound
a homogeneous mixture
a heterogeneous mixture
44
Poll

In Carbon Dioxide, the bond between the carbon and oxygen is,
single bond
double bond
triple bond
45
Multiple Choice

This is the displayed formula for what covalent compound?
Water
Hydrogen peroxide
Hydrogen oxide
Ethanol
46
Multiple Choice

This is the displayed formula for what covalent molecule?
Water
Chlorine
Chloride
Ethanol
47
Multiple Select

What does this picture illustrate? (Select all that apply)
Sharing valence electrons
Covalent bonding
transferring valence electrons
ionic bonding
two nonmetals
48
Multiple Choice

How many covalent bonds are created in a single water molecule?
0
1
2
3
4
49
Multiple Choice

How many covalent bonds are created in a single ammonia molecule?
0
1
2
3
4
50
Multiple Choice
What type of elements share electrons and form covalent bonds?
Nonmetals
Metals
Metalloids
A Metal & A Nonmetal
51
Multiple Choice

What type of bond will this compound likely form H-Cl?
Ionic compound
Covalent compound
Metallic compound
52

Metallic bonding is the electrostatic
attraction between positive metal
ions and delocalised electrons.
The organisation of the metal ions
and the free-moving electrons give
metals certain properties, such as
malleability and ability to conduct
heat and electricity.
METALLIC BONDING
53

54
Multiple Choice
55
Multiple Choice
56
Multiple Choice
57
Multiple Choice
58
Multiple Choice
59
Multiple Choice
60
Multiple Choice
61
Multiple Choice
Why do metals conduct heat and electricity
62
Multiple Choice
63
Multiple Choice
64
Multiple Choice
Why are metals malleable?
They are shiny
The electrons are held tightly within the lattice structure making it strong
The electrons are delocalized and able to move between the atoms
The electrons are shared between two metal ions and this holds the atoms together
65
Multiple Select
Which of these are considered properties of metals? (choose ALL that apply)
brittleness
low melting point
luster (shininess)
malleability
ductility
66
Multiple Choice
Why do metallic compounds conduct electricity as solids?
core electrons are mobile, allowing electricity to flow through the metal
valence electrons are mobile, allowing electricity to flow through the metal
protons are mobile, allowing electricity to flow through the metal
the metal cations are mobile, allowing electricity to flow through the metal
67
Multiple Choice
68
Multiple Choice
69
Multiple Choice
70

71

72
Multiple Choice
73
Multiple Choice
74
Multiple Choice
75
Multiple Choice
76
Multiple Choice
77
Multiple Choice
78
Multiple Choice
Why do jewelers not create jewelry with pure gold, silver, or copper?
It is a soft metal, so it will rub or scratch away over time.
It will not be shiny.
It is impossible to find a pure form of these metals.
It is too hard to shape into a design.
79
Multiple Choice
All alloys are mixtures.
True
False
80
Multiple Choice
Steel generally speaking is made of
iron and carbon
copper and iron
tin and carbon
aluminum and nickel
81
Multiple Choice
Brass is an alloy made of
copper and zinc
copper and tin
copper and nickel
copper and aluminum
82
Multiple Choice
Bronze is an alloy made of
aluminum and magnesium
copper and tin
lead and tin
copper and nickel
83
Multiple Choice
Steel can become stainless when alloyed with _____.
chromium and nickel
copper and zinc
tin and lead
iron aand carbon
84
Fill in the Blanks
85
Multiple Choice
Many alloys are softer than the elements that are in them.
True
False
86
Fill in the Blanks
87
Multiple Choice
In steel, iron is mixed with small amounts of non-metal, _______ .
Copper
Carbon
Diamonds
Wood
88
Fill in the Blanks
89
Multiple Choice
Pure iron is _________________ .
soft and weak
hard and stong
bendy and brittle
90
Multiple Select
Which of the following are alloys? (choose 3)
Lead
Bronze
Brass
Aluminium
Stainless Steel
91
Multiple Choice
Iron is a good conductor, malleable and magnetic. What type of element is Iron?
metal
non-metal
metalloid
alloy
92
Multiple Choice
Which substances do you find on the left of the Periodic Table?
metalloid
non-metals
metals
alloy
93
Multiple Choice
Alloys are much stronger because...
the particles from the different metals help the arrangement to stay in fixed positions
the different metals bond well
the particles from the different metals mix and disrupt the pattern making it hard for them to slide
94
Multiple Choice
Which metal is the best conductor of electricity?
steel
copper
zinc
brass
95
Multiple Choice
‘Bronze is harder than copper’. Which of the following is the best to explain the statement?
Atoms in bronze are arranged closely packed and less empty spaces.
Layers of atoms in bronze are not easily slide over each other.
Atomic size of copper and tin is different.
Bronze is a mixture of copper and tin
96
Multiple Choice
Which of these pictures shows the particles in a solid, pure metal?

CHEMICAL BONDS
Show answer
Auto Play
Slide 1 / 96
SLIDE
Similar Resources on Wayground

93 questions
EOC 2
Presentation
•
11th Grade

91 questions
Copy of Welcome to Animal Science I
Presentation
•
10th - 12th Grade

88 questions
Nubia 1b: The Kushites & Meroë Study Guide
Presentation
•
8th Grade

84 questions
Decade week
Presentation
•
11th Grade

100 questions
C1: States of Matter
Presentation
•
9th Grade

81 questions
Chemistry Semester 1 Review
Presentation
•
10th Grade

97 questions
Photosynthesis and Cellular Respiration Lesson
Presentation
•
9th - 12th Grade

99 questions
self guided periodic table intro
Presentation
•
11th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade