

Sub plan 2
Presentation
•
Science
•
8th Grade
•
Hard
+25
Standards-aligned
Beryl Lopez
Used 4+ times
FREE Resource
34 Slides • 88 Questions
1
Atomic Theory
Timeline of scientists and their findings

2
Multiple Choice
Electrons orbit a certain distance from the nucleus. Either radiate or absorb energy when jumping orbitals.
Dalton
Thomson
Rutherford
Bohr
3
Multiple Choice
The atom is mostly large empty space with a small, dense nucleus at the center
Dalton
Thomson
Rutherford
Bohr
4
Multiple Choice
Atom is a positively charged cloud with embedded negative electrons
Dalton
Thomson
Rutherford
Bohr
5
Multiple Choice
Atoms are indivisible (cannot be broken down)
Dalton
Thomson
Rutherford
Bohr
6
Multiple Choice

What scientist proposed this atomic model?
Dalton
Thomson
Rutherford
Bohr
7
Multiple Choice

What scientist proposed this atomic model?
Dalton
Thomson
Rutherford
Bohr
8
Multiple Choice

What scientist proposed this atomic model?
Dalton
Thomson
Rutherford
Bohr
9
Multiple Choice

What scientist proposed this atomic model?
Dalton
Thomson
Rutherford
Bohr
10
Multiple Choice
In a famous experiment conducted by Ernest Rutherford, positively charged alpha particles were scattered by a thin gold foil. Which of the following is a conclusion that resulted from this experiment?
The nucleus is negatively charged
The atom is a dense solid and is indivisible
The mass is conserved when atoms react chemically
The nucleus is very small and the atom is mostly empty space
11
Multiple Choice
In a famous experiment conducted by Ernest Rutherford, positively charged alpha particles were scattered by a thin gold foil. Which of the following is a conclusion that resulted from this experiment?
The nucleus is negatively charged
The atom is a dense solid and is indivisible
The mass is conserved when atoms react chemically
The nucleus is very small and the atom is mostly empty space
12
Multiple Choice
What subatomic particle was discovered in the cathode ray tube experiment?
Proton
Neutron
Electron
Nucleus
13
Multiple Choice
What subatomic particle was discovered in the cathode ray tube experiment?
Proton
Neutron
Electron
Nucleus
14
Multiple Choice
What evidence led Niels Bohr to believe that electrons occupy specific energy levels in atoms?
Excited electrons emit colored light at every possible wavelength.
Excited electrons emit colored light at only certain wavelengths.
Electrons emit colored light only when in an excited state.
Electrons emit colored light only when in ground state.
15
Multiple Choice
16
Multiple Choice
17

18

19

20

21
Multiple Choice
The vertical (up and down) columns in the Periodic Table are called
groups
towers
periods
atomic numbers
22
Multiple Choice
Elements in a .................. have similar chemical properties.
period
group
row
23

24

25
Multiple Choice
Which group of the periodic table is composed of inert (not reactive) gases?
answer choices
alkali metals
alkaline earth metals
halogens
noble gases
26

27

28

29
Multiple Choice
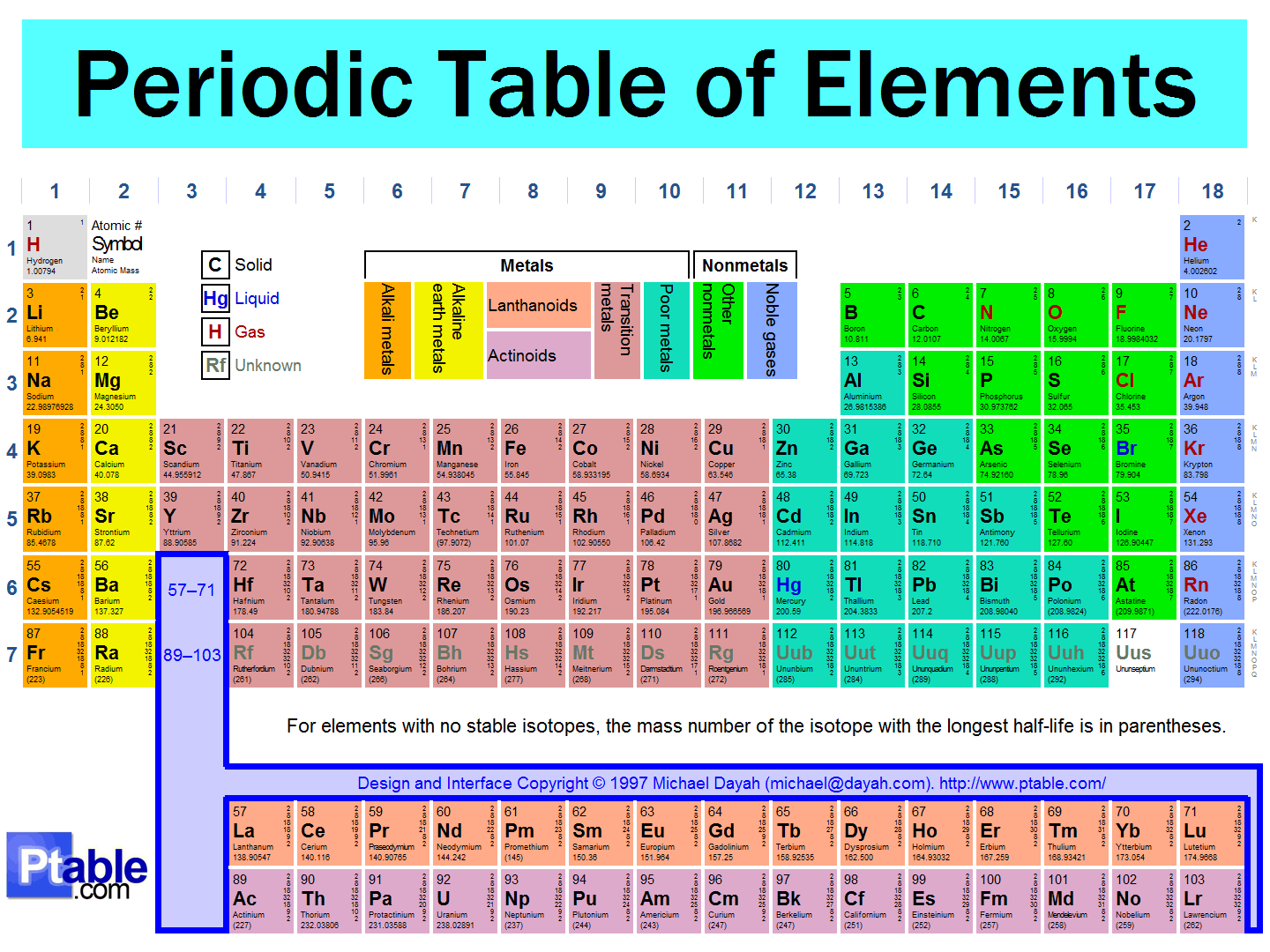
30
Multiple Choice

31
Multiple Choice
Which group of elements are the most reactive nonmetals?
Group 2
Group 1
Group 18
Group 17
32
Multiple Choice
What are the vertical columns on the periodic table called?
Groups
Periods
Elements
Valence electrons
33
Multiple Choice
Which group of elements have one valence electron(s)?
Group 13
Group 2
Group 18
Group 1
34
Multiple Choice
Which group of elements are nonreactive due to already being chemically stable?
Group 17
Group 1
Group 2
Group 18
35
Multiple Choice
Which classification of elements are shiny, silvery solids and good conductors of heat and electricity?
Transition metals
Metalloids
Nonmetals
Metals
36
Multiple Choice
Which classification of elements are gases or dull brittle solids and poor conductors?
Transition metals
Metals
Nonmetals
Metalloids
37
Multiple Choice
Which classification of elements are solids, semi-conductors, and have physical properties like metals and chemical properties like nonmetals?
Transition metals
Metalloids
Nonmetals
Metals
38
Categorize
shiny, silvery solids
good conductors of heat and electricity
malleable and ductile
located ot the left of the metalloids
gases, or dull brittle solids
poor conductors
right of the metalloids
solids
semi-conductors
physcial properties like metals
chemical properties like nonmetals
39
Multiple Choice
What is a Bohr Model
diagrams for funsies
simple diagrams that show the atomic structure of an atom
diagrams that show where it is at on the periodic table
not the answer
40
Multiple Choice

Who discovered the electrons?
Rutherford
Bohr
Thomson
Dalton
41
Pure Substances & Mixtures
Some text here about the topic of discussion

42
Multiple Choice
Both elements and compounds are pure substances.
AGREE
DISAGREE
43
Multiple Choice
Which of these is a compound?
Aluminium (Al)
Carbon Dioxide (CO2)
Nitrogen (N)
Sand
44
Multiple Select
Identify which images are PURE SUBSTANCES! (Check all that apply) : )
45

Homogeneous Mixture is uniform and evenly mixed!
46
Heterogeneous Mixture is NOT uniform and NOT evenly mixed!

47
Multiple Choice

Elements and Subatomic Particles
Protons and Electrons
Atoms and Compounds
Homogenous and Heterogeneous
48
Multiple Choice
In which mixture can you not see all of the parts?
homogeneous
heterogeneous
49
Multiple Choice
In which mixture can you see all of the parts?
homogeneous
heterogeneous
50

51
Drag and Drop
52
Multiple Choice
A solute is usually a ....
Solid
Liquid
Gas
53
Multiple Choice

Water is the universal ____.
solute
solvent
solution
54
Multiple Choice
What is another name for a homogeneous mixture?
Solute
Solution
Heterogeneous mixture
Solvent
55
Multiple Choice
The substance that is doing the dissolving in a solution is called the ---
solute
solvent
concentrated part
diluted part
56
Multiple Choice
If Mohammed dissolves 25g of sugar into 200mL of water that has a mass of 200g, what is the mass of the new solution?
225 g
425 g
175 g
between 200 g and 225 g
57
Multiple Select

Stefanie wants to dissolve a sugar cube into water. Which of the following will speed up the dissolving rate of the sugar cube? (Choose all that are correct!)
Using warm water instead of cold water
Add more than 1 cube at a time
Crushing the cube into small pieces
Adding the cubes to the cup and then pouring on the water
Stirring the water after adding the cubes
58
Multiple Choice

At 60 degrees Celsius, how much NaNO3 can be dissolved in 100g of water?
120 g
105 g
140 g
90 g
59
Multiple Choice

According to the graph, which substance is not much affected in solubility as the temperature rises?
NaNO3
KNO3
NaCl
Na2SO3
60
Multiple Choice
Adam makes coffee by mixing coffee and sugar into hot water. The coffee and sugar have dissolved completely. What type of mixture did Adam make?
Colloid
suspension
solution
compound
61
Multiple Choice
Which of the following groups are ALL mixtures?
soil, water, blood, salt
soil, blood, air, salt water
Oxygen, water, air, salt
salt water, coffee, oxygen, sugar
62
Multiple Choice
Milk is an example of which type of mixture?
solution
suspension
colloid
compound
63
Multiple Select

Which of the following are true statements about the picture? (Choose all that are correct!)
A is the most concentrated
G is the most diluted
C is more diluted than E
D is more diluted than E and more concentrated than C
B is more diluted than A and more concentrated than C
64
Multiple Choice

In the glass of salt water shown, what is the solute in the solution?
water
salt
salt water
this does not have a solute because it is not a solution
65
Multiple Select
Which of the following are homogeneous mixtures? (Choose all that are correct!)
cereal with milk
chocolate chip icecream
salt water
pizza
Soda
66
Multiple Choice

Which picture represents a compound?
A
B
C
67
Multiple Choice

Which of the pictures represents a mixture?
A
B
C
68
Multiple Choice
A substance made of two or more PHYSICALLY combined substances is--
an element
a mixture
a salt
a compound
69
Multiple Choice
A substance made of only 1 type of atom is called --
an element
a salt
a compound
a mixture
70
Multiple Choice

Name the separation technique shown in the diagram.
evaporation
distillation
filtration
71
Multiple Choice
72
Multiple Choice
73
Multiple Choice
74
Multiple Choice

What mixture could you use this tool to best separate that mixture?
Sugar and Salt
Sand and rocks
Apples and grapes
75
Multiple Choice

76
Multiple Choice

If I have a mixture of wooden blocks and metal blocks. What is a fast way to separate them?
relative density (put them in water)
filter
sieve
77

78
Multiple Choice
How do we know a chemical reaction has taken place?
Substances change in appearance
Substances change in size
Substances change in states, like from a liquid into a solid.
New substances are formed
79

80
Multiple Choice
Which of the following is a reactant in the following equation?
O2 + C6H12O6 > CO2 + H2O
CO2
H2O
O2
81
Multiple Choice
Which of the following is a product in the following equation?
O2 + C6H12O6 > CO2 + H2O
CO2
C6H12O6
O2
82

83

84

85

86
Multiple Choice
Lighting a match is an example of
an endothermic reaction
an endergonic reaction
both an endothermic and an endergonic reaction
both an exothermic and an exergonic reaction
87

88
Multiple Choice
In the following equation, which is the catalyst?
H2O2 + Catalase > H2O + O2 + Catalase
H2O2
H2O
O2
Catalase
89

90

91

92

93

94
Multiple Choice
In which of the following has a chemical reaction taken place?
An apple turning brown
Milk developing a sour taste
Smoke being released as a candle burns
All of the previous
95

96
Multiple Choice
Which of the following is an example of a CHEMICAL change?
Shattering a window with a baseball
Ripping paper into 1,000,000,000 tiny pieces
Rusting an iron bar on a humid day
Melting a block of copper metal
97
Multiple Choice
Which of the following is an example of a PHYSICAL change?
Neutralizing vinegar with baking soda
Breaking your iPad with a rock due to school stress
Creating a precipitate by mixing Lead Nitrate and Sodium Iodide
Burning popcorn in the microwave
98
Multiple Choice

What are the products of ALL combustion reactions?
Carbon Dioxide and Water
Smoke and Ash
Fire and Carbon Dioxide
Water and Ash
99
Multiple Choice
What does the symbol (aq) stand for?
Aquatic
Aqueous
Aqua
Aquarium
100
Multiple Choice
Which law states that Matter cannot be created or destroyed?
Law of Conservation of Matter
Law of Gravity
Law of Equivalent Exchange
Law of Matter ad Infinitum
101
Multiple Choice
When a chemical reaction takes in more energy than it gives off it becomes _______________.
colder
hotter
bigger
smaller
102

103

104
Multiple Choice
What does 'atomos' mean?
not to be cut
not to be broken
not very big
not to be seen
105
Open Ended
What did his model of the atom look like?
106

107
Multiple Choice
What is Dalton best known for?
the first modern atomic theory
playing pool
saying that atoms are small
108

109
Fill in the Blanks
Type answer...
110
Multiple Select
Which two desserts look like Thomson's model?
chocolate chip cookie
plum pudding
cake
brownies
111

112
Multiple Choice
What element is most famous in Rutherford's experiment?
gold
silver
platinum
nickel
113
Open Ended
What did Rutherford prove?
114
Multiple Choice
What did Rutherford call his new discovery?
the electron cloud
the nucleus
the orbital
115

116
Multiple Choice
Where is most of the mass of an atom located?
the nucleus
the electron cloud
the space in between particles
117
Open Ended
What did Bohr compare the atom to?
118
Multiple Choice
Bohr is the first to give definite information regarding which subatomic particle?
electron
proton
neutron
119

120
Open Ended
What do we now know about electrons?
121
Multiple Choice
What does an electrons location depend on?
how fast it is moving
how much energy it has
122
Multiple Choice
What do we call the area in which electrons are located?
the electron sky
the electron space
the electron cloud
the electron solar system
Atomic Theory
Timeline of scientists and their findings

Show answer
Auto Play
Slide 1 / 122
SLIDE
Similar Resources on Wayground

109 questions
Intro to Similarities
Presentation
•
8th Grade

119 questions
Ch5 : Networks
Presentation
•
10th Grade

120 questions
Elements of Art: 7
Presentation
•
7th Grade

119 questions
WW3 Review Lessons 7-9
Presentation
•
8th Grade

116 questions
Civics EOC Lesson
Presentation
•
7th - 8th Grade

113 questions
Chapter 6 PPT (Causes of the American Revolution
Presentation
•
8th Grade

115 questions
3E Sample Revision PPT L50
Presentation
•
8th - 10th Grade

109 questions
F4 (R) Buy It! L35 2021
Presentation
•
8th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade

48 questions
8th Grade NC Science EOG Review 2
Quiz
•
8th Grade

19 questions
Renewable and Nonrenewable Resources (CK)
Quiz
•
8th Grade

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade

10 questions
Exploring Weather Influences and Map Reading
Interactive video
•
6th - 10th Grade

43 questions
Amplify Earth's Changing Climate Unit Review
Quiz
•
6th - 8th Grade