

7.3.1-Solids, Liquids, & Gases
Presentation
•
Science
•
7th Grade
•
Easy
+13
Standards-aligned
Jessica Freeman
Used 3+ times
FREE Resource
44 Slides • 27 Questions
1

2

3

4

5

6

7

8
Multiple Choice
What are the states of matter?
solid
liquid
gas
all of these
9
Multiple Choice
What is matter made of?
particles
big blocks
only solids
10

11

12

13

14

15

16

17

18

19

20
Particle motion in a solid

21

22
Multiple Choice

What type of solid contains particles that are jumbled and form no particular pattern?
Crystalline
Amorphous
High-density
Low-volume
23

24

25

26
Particle motion in a liquid

27

28

29

30
31
Multiple Choice
Water is polar. What does that mean?
it is a molecule with opposite charges on opposite ends
it is a molecule with no charge
it is a molecule with identical charges on opposite ends
it is a molecule with too many protons
32
Multiple Choice
Water sticks well to many materials. What term relates to this property of water?
cohesion
adhesion
density
surface tension
33
Multiple Choice
Which of the following is an example of COHESION?
water sticking to water
water sticking to glass
oil sticking to plastic
hydrogen sticking to oxygen
34
Multiple Choice
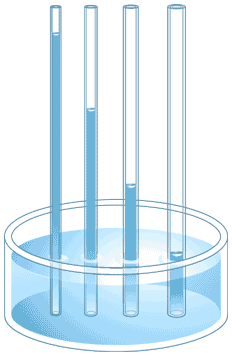
What is the term for water's ability to defy gravity and climb up a tube?
Capillary Action
Specific Heat
Universal Solvent
Magic
35
Multiple Choice

Why does ice float?
water expands when it freezes, making ice less dense than water
water compacts when it freezes, making ice more dense than water
hydrogen bonds in water push the ice to the surface
ice is afraid of water, and avoids it
36
Multiple Choice
37
Multiple Choice

38
Multiple Choice
A water molecule is a _______ since the negative end of one water molecule is attracted to the positive end of another water molecule.
polar molecule
sustain life
surface tension
specific heat
39
Multiple Choice

An attraction between molecules of different substances is called...
adhesion
cohesion
40
Multiple Choice

A measure of how difficult it is to stretch or break the surface of a liquid is called...
specific heat
polar molecule
capillary action
surface tension
41
Multiple Choice
An attraction between molecules of the same substance is called...
adhesion
cohesion
42
Multiple Choice
Water molecules ________ since the slightly positive portion of a water molecule is attracted to the slightly negative portion of the next water molecule.
stick together
spread out
43

44
Low-viscosity: water

45
High-viscosity: Honey

46
Multiple Choice

Notice how the liquid on the left forms round drops while the one on the right spreads out.
Which phrase describes the difference between the liquids?
Fixed volume vs changing volume
Different particle arrangement
Low-viscosity vs high-viscosity
Low-surface tension vs High-surface tension
47

48

49

50
Particle motion in a gas.

51

52
Multiple Choice
What is the best way to describe particles in a gas?
Very energetic
Vibrating in place
Completely still
Closely packed
53

54

55

56
Multiple Choice
Which of the following depicts particles in a solid?
57
Properties of matter
Solids- particles are tightly packed, vibrate in place and have a definite volume and shape.
Liquids- Particles are loose and can move, they have a definite volume, but not a definite shape.
Gases- Particles are spread far and move easily, they have no definite shape or volume
Plasma- particles are loose and can move like water, but have no definite shape or volume like gas and are electrically charged.

58
Other properties of liquid
surface tension- the pulling of molecules toward each other.
viscosity- the resistance of flowing

59
Draw
Draw the particle organization of each of the 4 states of matter.
60

61

62

63

64
Multiple Choice
What effect does heat have on atomic motion?
It causes them to vibrate slower
It causes them to vibrate faster
65
Multiple Choice
What effect does the loss of heat have on atomic motion?
It causes them to vibrate faster
It causes them to vibrate slower
66
Multiple Choice
Why does a liquid become less dense when heat is added?
Heat causes the atoms to vibrate slower and move closer together, making the liquid less dense.
Heat causes the atoms to vibrate faster and move further apart, making the liquid less dense.
67
Multiple Choice

a
b
c
d
68
Multiple Choice

a
b
c
d
69
Multiple Choice

70
Multiple Choice

a
b
c
d
71
Multiple Choice

a
b
c
d

Show answer
Auto Play
Slide 1 / 71
SLIDE
Similar Resources on Wayground

63 questions
Communications
Presentation
•
7th Grade

68 questions
Quizizz #1 - TEST REVIEW - Earth's Layers and Tectonic Plates
Presentation
•
6th Grade

70 questions
Chemical Reactions
Presentation
•
8th Grade

66 questions
Photosynthesis and Cellular Respiration
Presentation
•
6th - 8th Grade

69 questions
Present Coninuous lesson
Presentation
•
7th Grade

67 questions
Grade 7 Unit 6 Cells Homework
Presentation
•
7th Grade

67 questions
Matter
Presentation
•
7th Grade

67 questions
Physical/Chemical Changes
Presentation
•
6th - 8th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

20 questions
genetics, punnett squares, heredity
Quiz
•
7th Grade

20 questions
Food Webs + Energy Pyramids
Quiz
•
7th Grade

10 questions
Exploring Weather Influences and Map Reading
Interactive video
•
6th - 10th Grade

43 questions
Amplify Earth's Changing Climate Unit Review
Quiz
•
6th - 8th Grade

62 questions
Super Science Trivia
Quiz
•
6th - 8th Grade