

Classification & Phys/Chem Prop/Changes REVIEW LESSON
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
Tracy Rodgers
Used 4+ times
FREE Resource
0 Slides • 80 Questions
1
Multiple Choice
Matter can be classified as either a __________ or a _____________.
Element or a Compound
Compound or a Solid
Pure Substance or a Mixture
Mixture or a Solid
2
Multiple Choice
Which two types of matter are pure substances?
elements and compounds
mixtures and elements
homogenous mixtures and elements
3
Multiple Select
What is the difference between a pure substance and a mixture? Select all that apply.
substances are made of many different things while mixtures are just made of 1
a substance has one type of particle while a mixture has 2 or more types
a mixture is physically combined and can be physically separated while a substance is chemically bonded
a mixture is chemically bonded together while a substance is physically combined
4
Multiple Choice
Which of the following represents an element?
H2O
HCl
NaCl
Au
CaCO3
5
Multiple Choice
6
Multiple Choice
7
Multiple Choice
A mixture that is uniformly mixed is called?
Element
Compound
Homogeneous
Heterogenous
8
Multiple Choice
9
Multiple Choice

The diagram shows
element
mixture
compound
10
Multiple Choice
11
Multiple Choice
What is a type of mixture in which different parts can easily be seen (Ex. Vegetable soup)?
Heterogeneous mixture
Homogeneous mixture
Solution
Colloid
12
Multiple Choice

What does this pic illustrate?
Element
Mixture of Compunds
Mixture of elemets
Compounds
13
Multiple Choice

Identify the following
compound
homogeneous mixture
element
heterogeneous mixture
14
Multiple Choice
A material made up of two more more substances that can be easily separated by physical means is called?
Mixture
Element
Compound
Pure Substance
15
Multiple Choice
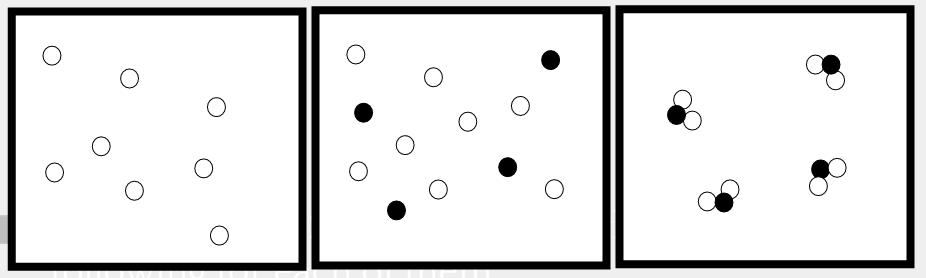
16
Multiple Choice
Water and sugar dissolved is an example of:
Compound
Homogeneous mixture
Substance
Heterogeneous mixture
17
Multiple Choice
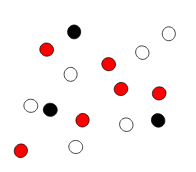
18
Multiple Choice

Identify the following
compound
homogeneous mixture
element
heterogeneous mixture
19
Multiple Choice
A chocolate chip cookie is an example of:
Substance
Compound
Homogeneous mixture
Heterogeneous mixture
20
Multiple Choice
What is a compound?
Two different elements bonded together.
Two of the same elements bonded together.
A mixture of different substances.
Two substances stuck together with glue.
21
Multiple Choice

What does this pic illustrate?
Element
Mixture of Compunds
Mixture of elemets
Compounds
22
Multiple Choice
True or False.
23
Multiple Choice
What is a compound?
Two different elements bonded together.
Two of the same elements bonded together.
A mixture of different substances.
Two substances stuck together with glue.
24
Multiple Choice
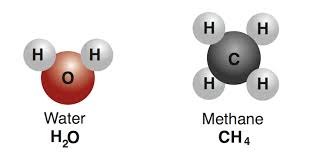
25
Multiple Choice

26
Multiple Choice

27
Multiple Choice

28
Multiple Choice
29
Multiple Choice
30
Multiple Choice
a property that DOESN'T change the composition of a substance
physical property
chemical property
density
volume
31
Multiple Choice

32
Multiple Choice
the amount of SPACE a substance takes up
matter
density
mass
volume
33
Multiple Choice
a characteristic of matter
flammability
property
substance
chemical symbol
34
Multiple Choice
properties that HAVE the ability to change chemical bonds and atomic composition
physical property
chemical bonds
chemical property
atomic composition
35
Multiple Choice
36
Multiple Choice

A wall that was once white is painted black. Which of the following is definitely true of the painted wall?
Its chemical properties have changed.
Its physical properties have changed.
Both its physical properties and chemical properties have changed.
None of its characteristic properties have changed.
37
Multiple Choice
A solid is crushed into a powder?
38
Multiple Choice
Solubility, the ability to dissolve, is a --- property.
Chemical
Physical
39
Multiple Choice

Hydrogen peroxide bubbles when you put it on a cut on your finger indicates a ?????
physical challenge
physical change
chemical corrosion
chemical change
40
Multiple Choice

Iron fillings and Sulfur that are stirred together indicates ???
physical charade
chemical change
physical change
41
Multiple Choice

Acids have the ability to react with bases to become salts. This is a statement about the acids'
chemical properties
physical properties
chemical and physical properties
visual properties
42
Multiple Choice

Which of the following is a physical property of matter?
reactivity
volume
solubility
combustibility
43
Multiple Choice

44
Multiple Choice

Making lemonade indicates a ??????
physical challenge
physical change
chemical corrosion
chemical change
45
Multiple Choice

Which of the following involves a change in chemical properties?
boiling water to make steam
mixing baking soda and vinegar to make a bubbles of carbon dioxide
melting a bar of solid gold to make liquid gold
tearing a piece of paper to make smaller pieces of paper
46
Multiple Choice

Which of the following is a chemical property
acidity
electrical conductivity
color
volume
47
Multiple Choice
48
Multiple Choice
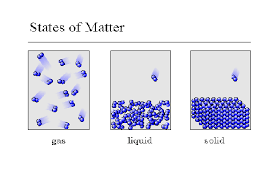
49
Multiple Choice
a property that describes how well one substance dissolves in another substance
malleablility
density
phase chane
solubility
50
Multiple Choice
What is the outcome of observing a chemical property?
Nothing
A new substance is formed with different properties
NaCl
A rise in thermal conductivity
51
Multiple Choice

Paint being sanded off a car is an indicator for a ?????
physical challenge
physical change
chemical change
chemical corrosion
52
Multiple Choice
53
Multiple Choice

The process of roasting marshmallows indicates a ?????
physical challenge
physical change
chemical corrosion
chemical change
54
Multiple Choice

Your body breaking down a candy bar when eaten.
physical challenge
physical change
chemical corrosion
chemical change
55
Multiple Choice

Which of the following involves a change in physical properties only?
baking a loaf of bread
freezing water into ice cubes
burning a match
mixing an acid and a base
56
Multiple Choice

A piece of chalk becomes shorter as it is used. Which of the following is NOT true of the shorter piece of chalk?
Its mass has changed.
Its volume has changed.
Its shape has changed.
Its characteristic properties have changed
57
Multiple Choice
58
Multiple Choice

Freezing
F
C
A
D
59
Multiple Choice
Liquid to Gas
melting
vaporization
condensation
sublimation
60
Multiple Choice
Melting
endothermic
exothermic
61
Multiple Choice

Sublimation
A
B
C
F
62
Multiple Choice
Condensation
endothermic
(absorbs energy)
exothermic
(releases energy)
63
Multiple Choice
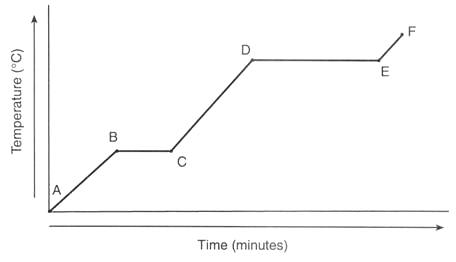
What is happening to the particles in the substance between points A and B?
Melting
Freezing
Speeding Up
Slowing Down
64
Multiple Choice

What letters would be only a liquid?
A-->B
C-->D
E-->F
None of these are correct
65
Multiple Choice

66
Multiple Choice

Which answer would have the greatest particle motion?
A-->B
C-->D
E-->F
none of these are correct
67
Multiple Choice

Use the graph below to answer this question. Which phase change is taking place if the substance is losing energy from point C to point B?
melting
freezing
evaporation
condensation
68
Multiple Choice

69
Multiple Choice

70
Multiple Choice

71
Multiple Choice

72
Multiple Choice

73
Multiple Choice

What letters would be only a solid?
A-->B
C-->D
E-->F
None of these are correct.
74
Multiple Choice

What letters would be only a gas?
A-->B
C-->D
E-->F
none of these are correct
75
Open Ended

When Substance X and Substance Y are mixed together, a magnet can remove particles of Substance X.
Type "Y" for yes & "N" for no
76
Open Ended

When Substance X and Substance Z are heated together in test tube, drops of Substance B form on the inside of the test tube.
Type "Y" for yes & "N" for no
77
Open Ended

Particles of Substance B are suspended in Substance T.
Type "Y" for yes & "N" for no
78
Open Ended

When Substance Q is added to Substance R, Substance R tastes salty.
Type "Y" for yes & "N" for no
79
Open Ended

Substance A is added to Substance B. The freezing point of B is lowered by 9oC.
Type "Y" for yes & "N" for no.
80
Open Ended

When solid Substance Y is added to liquid Substance R, there is a violent explosion. The resulting matter has less mass than the combined mass of Y and R.
Type "Y" for yes & "N" for no.
Matter can be classified as either a __________ or a _____________.
Element or a Compound
Compound or a Solid
Pure Substance or a Mixture
Mixture or a Solid
Show answer
Auto Play
Slide 1 / 80
MULTIPLE CHOICE
Similar Resources on Wayground

76 questions
Powers Unit 5 Notes Final
Presentation
•
9th - 12th Grade

73 questions
LAST CHANCE Biology SOL Review!!!
Presentation
•
9th - 12th Grade

76 questions
Equations and Inequalities
Presentation
•
9th - 12th Grade

76 questions
Gr 11 Exam Prep and Review - Content
Presentation
•
9th - 12th Grade

74 questions
Neo-Confucianism
Presentation
•
9th - 12th Grade

74 questions
Polymer and Plastics
Presentation
•
9th Grade - University

73 questions
Mitosis vs Meiosis
Presentation
•
9th - 12th Grade

75 questions
ILD 4 Jan (Sec 5)
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Chemistry

25 questions
Waygrounds 5/20 PS.8.1.2
Quiz
•
9th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

15 questions
Types of chemical reactions
Quiz
•
9th - 12th Grade

32 questions
Acids/Bases, Electrochem, Orgo
Quiz
•
9th - 12th Grade