

PSU4.A Pg 3-4
Presentation
•
Science
•
9th - 12th Grade
•
Medium
Jolene Kayser
Used 1+ times
FREE Resource
23 Slides • 16 Questions
1

2
Multiple Choice
Beryllium
Be
B
Br
Ba
3
Multiple Choice
Carbon
C
Ca
Ce
Cs
4
Multiple Choice
Potassium
K
P
Kr
Po
5

6

7

8

9

10

11

12

13

14
Categorize
negative charge
No charge
Positive Charge
Found in the nucleus
Mass of approximately 1 atomic mass unit
Determines the atomic number
Contributes to the atomic mass
Positively charged subatomic particle
Found in the nucleus
Mass of approximately 1 atomic mass unit
Contributes to the atomic mass
Neutral subatomic particle
Helps stabilize the nucleus
Found in electron shells
Mass is negligible compared to protons and neutrons
Involved in chemical bonding
Negatively charged subatomic particle
Can be gained or lost in chemical reactions
Classify the charges
15
Multiple Choice
Which part of an atom occupies most of the volume?
Electron cloud
Neutrons
Nucleus
Protons
16
Multiple Choice
The center of an atom is called the __________ ?
neutron
nucleus
number
Centergon
17
Multiple Select
The nucleus is made up of 2 particles. Select the correct 2 particles below.
protons
neutrons
electrons
18

19

20

21

22
Multiple Choice
Parts of the atom that determine the identity of the atom (what kind of atom it is)
Protons
Neutrons
Electrons
23
Multiple Choice

How many protons are in Sodium?
11
22
12
33
24
Multiple Choice

How many electrons are in Potassium?
19
20
58
39
25

26

27

28

29

30

31

32

33

34
Multiple Choice
Parts of the atom that are the smallest of the three subatomic particles
Protons
Neutrons
Electrons
35
Multiple Choice

What is the Atomic Number of this Element
7
3
4
1
36
Multiple Select
Which subatomic particle(s) do we use to calculate mass number? (Select all that apply)
electron
proton
neutron
none of these
37
Multiple Select
Which subatomic particle(s) do we use to find atomic number? (Select all that apply)
electron
proton
neutron
none of these
38
Multiple Choice
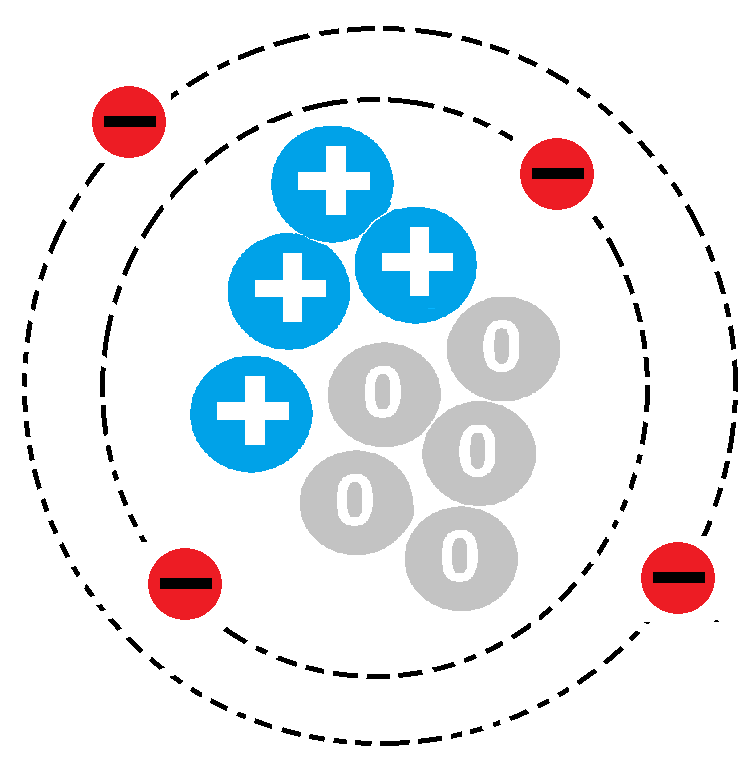
What is the Name of this atom?
Hydrogen
Fluorine
Boron
Beryllium
39
Multiple Choice

What is the mass number of this atom?
8
4
13
9

Show answer
Auto Play
Slide 1 / 39
SLIDE
Similar Resources on Wayground

33 questions
Astronomy
Presentation
•
9th - 12th Grade

32 questions
Basics on Chemical Bonding
Presentation
•
9th - 12th Grade

33 questions
Earth Science Review - The Solar System
Presentation
•
9th - 12th Grade

33 questions
Atoms and Atomic Structure
Presentation
•
8th - 11th Grade

34 questions
Ecology Review Part 1 - Food Webs and Energy Pyramids
Presentation
•
9th - 12th Grade

35 questions
Electron Configuration
Presentation
•
9th - 12th Grade

33 questions
Sectionalism
Presentation
•
9th - 12th Grade

36 questions
Unit 3 Lesson 1
Presentation
•
9th Grade - University
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

11 questions
EOC Domain 1: Cells
Presentation
•
9th - 10th Grade

14 questions
EOC Domain 2: Genetics
Presentation
•
9th - 10th Grade

10 questions
Exploring the Human Respiratory System
Interactive video
•
6th - 10th Grade