

Reaction with acids
Presentation
•
Science
•
10th Grade
•
Medium
Standards-aligned
Céline Sailly
Used 1+ times
FREE Resource
8 Slides • 24 Questions
1
Open Ended
What is the definition of an acid?
2
Multiple Choice
sulfuric acid
HSO3
H2SO3
H2SO4
H3SO4
3
Open Ended
What is the definition of an alkali?
4
Multiple Select
Which of the following are bases?
sodium oxide
sodium nitrate
sodium hydroxide
potassium oxide
5
Multiple Choice

HCl is found in household products, including some toilet bowl cleaners. Is HCl an acid or a base?
Acid
Base
Neither (Neutral)
6
Multiple Choice
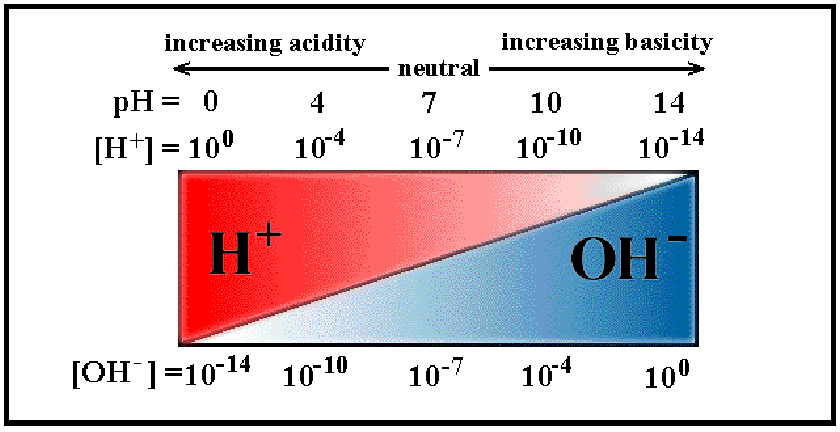
True or false: a neutral solution has equal amounts of H+ and OH-.
True
False
7
Multiple Choice
8
Multiple Choice
A substance is found to have the following characteristics:
Very bitter taste
Feels soapy to the touch
Produces OH- ions when dissolved in water
In what category would the substance be classified?
acid
base
9
REACTIONS with ACIDs
10
NEUTRALISATION REACTIONS

Don't forget:
- To balance the reaction
- To show the states of reactants and products
11
Multiple Choice
Which of the following is the ionic eqution for neutralisation?
H++OH−→H2O
H2+O2→H2O2
2H++OH−→H3O
NaOH +HCl →NaCl +H2O
12
Fill in the Blanks
Type answer...
13
Multiple Choice
Sulfuric acid reacts with sodium hydroxide to form salt X and water. Name the salt.
sodium sulfate
sodium nitrate
sodium chloride
14
Multiple Choice
Complete the following reaction:
Hydrochloric acid + magnesium hydroxide -->
Magnesium chloride + water
Magnesium + water
Magnesium chloride + hydrogen
Magnesium chloride + water + carbon dioxide
15
Multiple Choice
What type of salt can hydrochloric acid produce?
calcium hydrochloric
calcium hydrochloride
calcium chloride
calcium nitrate
16
REACTION WITH A METAL

17
Fill in the Blanks
Type answer...
18
Multiple Choice
HCl + Zn --> ZnCl2 + H2
19
REACTION WITH METAL OXIDE
What is a METAL OXIDE?
A metal oxide is a compound that is made with a metal and oxygen anion O2-
Examples: MgO -> magnesium oxide,
Fe2O3 is iron(III) oxide
20
Multiple Choice
21
Multiple Choice
Determine the formula unit: Lead(IV) oxide
Pb2O
Pb4O
PbO4
PbO2
22
Multiple Choice
Which is the correct formula for the compound: Calcium Oxide
CaO
CaO2
Ca2O
23
COPY IN YOUR NOTEBOOK

24
Multiple Choice
What are the products of a reaction between a metal oxide and an acid
Oxygen gas and water
Salt and water
Oxygen gas and salt
hydrogen gas and salt
25
REACTION WITH METAL CARBONATE
What is a METAL CARBONATE?
A metal carbonate is a compound that is made with a metal and carbonate CO32-
Examples: MgCO3 -> magnesium carbonate,
CaCO3 -> Calcium carbonate
26
Multiple Choice
27
Multiple Choice
Name: (NH4)2CO3
Ammonium (I) carbonate
Ammonium(II) carbonate
Ammonium carbonate
Ammonium carbon trioxide
28
COPY IN YOUR NOTEBOOK

29
Fill in the Blanks
Type answer...
30
Multiple Choice
Potassium carbonate reacts with an acid Y to form potassium nitrate, water and carbon dioxide. What is the acid used for this reaction?
HNO3
H2SO4
HCl
31
WRAP UP QUESTION
32
Multiple Select
Which of the following produce magnesium chloride salt? (multiple answer possible)
magnesium and hydrochloric acid
magnesium oxide and hydrochloric acid
magnesium hydroxide and hydrochloric acid
magnesium carbonate and hydrochloric acid
What is the definition of an acid?
Show answer
Auto Play
Slide 1 / 32
OPEN ENDED
Similar Resources on Wayground

23 questions
Naming Covalent Compounds
Presentation
•
10th Grade

28 questions
Genetic Mutation
Presentation
•
10th Grade

26 questions
Forensic Science Lesson 06: Fingerprints 1
Presentation
•
10th Grade

27 questions
PS03-3 Classifying Matter
Presentation
•
9th - 11th Grade

27 questions
DNA Structure Lesson 3
Presentation
•
10th Grade

26 questions
Chemical Bonds
Presentation
•
10th Grade

26 questions
Water Pollution
Presentation
•
9th Grade

26 questions
DNA Replication - Base Pairs
Presentation
•
10th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade

49 questions
AP Environmental Science Final Exam Review
Quiz
•
10th Grade - University

10 questions
Exploring Chemical and Physical Changes
Interactive video
•
6th - 10th Grade

10 questions
Exploring Animal Adaptations: Physical, Behavioral, and Life Cycle Changes
Interactive video
•
6th - 10th Grade