

Electrons in Atoms
Presentation
•
Science
•
10th - 12th Grade
•
Hard
Joseph Anderson
FREE Resource
13 Slides • 10 Questions
1
Electrons in Atoms

2

3

4

5
Open Ended

How is the Bohr Model similar to a ladder?
6
Open Ended

1. Why is less energy required in higher levels (n=5) to go to the next level?
7
Multiple Choice
1. The amount of energy required to move an electron from one energy level to another is called_______________________________
orbitals
Quantum of energy
energy level
8
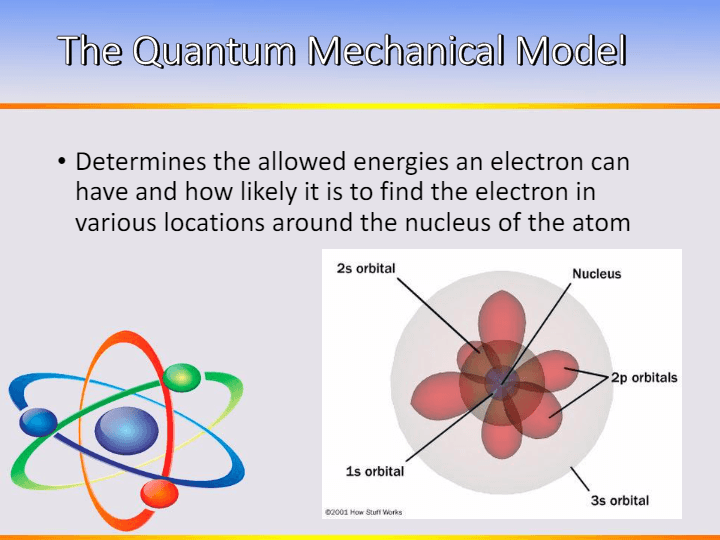
9

10
Multiple Choice
1. What Determines the allowed energies an electron can have and how likely it is to find the electron in various locations around the nucleus of the atom?
Rutherford's model
The Bohr Model
Quantum Mechanical Model
11
Multiple Choice
Who derived the equation for the quantum mechanical model?
Ernest Rutherford
Niels Bohr
Erwin Schrodinger
JJ Thomson
12
Multiple Select
1. Compare the Bohr Model and the Quantum Mechanical Model.
both have electrons
both travel in a circular orbit
both have energy levels
both have a maximum number of electrons per energy level
both are based on the probability an electron will be there
13

14
Multiple Choice
1. What is the principal quantum number?
how many electrons are in the atom
indicates the energy level the electron is on
how many valence electrons an atom has
15
Multiple Choice
1. What are the shapes described by Schrodinger’s equation called?
electrons
atomic orbitals
energy levels
atoms
16
Multiple Choice
1. What are atomic orbitals?
regions where there is a high probability of finding an electron
regions where it is hghly unlikely to find an electron
same as the nucleus
17

18
Multiple Choice
1. How can you determine the maximum number of electrons that can fit in an energy level? (formula)
n
n2
2n2
none of the above
19

20

21

22

23

Electrons in Atoms

Show answer
Auto Play
Slide 1 / 23
SLIDE
Similar Resources on Wayground

19 questions
Marine and brackish biomes
Presentation
•
9th - 12th Grade

20 questions
Cell Differentiation and Specialized Cells
Presentation
•
9th - 12th Grade

18 questions
Passive and Active transport
Presentation
•
9th - 11th Grade

17 questions
Waves
Presentation
•
9th - 11th Grade

20 questions
Evolution of Plant Transport System
Presentation
•
9th - 12th Grade

21 questions
Unit 7: Energy Transformations
Presentation
•
9th - 12th Grade

17 questions
Genetics Review Part 2 (Practice)
Presentation
•
6th - 9th Grade

20 questions
ions
Presentation
•
10th - 12th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade

49 questions
AP Environmental Science Final Exam Review
Quiz
•
10th Grade - University

10 questions
Exploring Chemical and Physical Changes
Interactive video
•
6th - 10th Grade

10 questions
Exploring Animal Adaptations: Physical, Behavioral, and Life Cycle Changes
Interactive video
•
6th - 10th Grade