

Periodic Table of Elements Atomic Numbers
Presentation
•
Science
•
8th Grade
•
Hard
Joseph Anderson
FREE Resource
8 Slides • 107 Questions
1
Atoms and the Periodic Table

2
What is an atom?
An atom is the building block of all matter.
Atoms are very small; smaller than the tip of your pencil.
Atoms are composed of particles called protons, electrons and neutrons.
Atoms can join together to form molecules, which in turn form most of the objects around you.

3
Multiple Choice

What is at the center of every atom?
an electron
only protons
a molecule
a nucleus
4
The Periodic Table of the Elements
display of the chemical elements, which are arranged by atomic number, electron configuration, and recurring chemical properties
The seven rows of the table, called periods, generally have metals on the left and nonmetals on the right.
The columns, called groups, contain elements with similar chemical behaviors.

5
Multiple Choice

What is the Periodic Table?
a chart that lists all the molecules that exist in the universe
a chart that shows all the elements scientists have discovered
a chart that shows all the compounds found in the world
a chart that shows what time periods elements were discovered in
6
Chemical Element
Atomic number- tells the number of protons, electrons and the location of the element on the Periodic Table
Atomic Mass- the sum of the protons and neutrons in the nucleus of the atom
To find the number of neutrons, subtract the atomic number from the atomic mass
APE MAN

7
Multiple Choice
What does an element's atomic number represent?
the element state at room temperature
how metallic an element is
the number of atoms in a compound
the number of protons in an element
8
Electron Shell Rules:
1st shell can hold up to 2 electrons
2nd shell can hold up to 8 electrons
3rd shell can hold up to 18 electrons

9
Multiple Choice
Which of the following is the correct atomic model for Boron?
10
Multiple Choice

How many neutrons are in Nitrogen?
*Click the image to make it larger.
14
7
21
11
The three basic states of matter

12

13
Multiple Choice
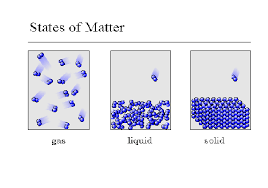
At higher temperatures
particles in an object have less energy
particles in an object move faster
a gas contracts
14
Multiple Choice
How is a gas defined?
no shape, no volume
Definite shape, definite volume
Found in stars
Definite volume, no shape
15
All matter have properties.

16
Multiple Choice
Physical properties are
are properties that can be observed without changing the matter's identity
are properties that can be observed that change the matter into new matter with new properties
17
Multiple Choice
Which of the following is a sign that a chemical reaction has occurred?
change in shape
melting
formation of a gas
dissolving
18
Poll
How would you rate your understanding of atoms and the Periodic Table?
19
Multiple Choice
the smallest particle that still can be considered an element
proton
neutron
electron
atom
20
Multiple Choice
a series of models that developed from experimental evidence
atomic law
atomic theory
elemental law
elemental theory
21
Multiple Choice
negatively charged particles, discovered by Thomson
proton
neutron
electron
22
Multiple Choice
the dense center of an atom
proton
nuetron
nucleus
election
23
Multiple Choice
a positively-charged particle in the atoms nucleus
proton
neutron
electron
24
Multiple Choice
a particle with no charge in the nucleus of the atom
proton
neutron
electron
25
Multiple Choice
the number of protons in the nucleus of an atom
protons
atomic number
electron
nucleus
26
Multiple Choice
average mass of all the isotopes of that element
proton
atomic mass
electon
nucleus
27
Multiple Choice
a chart showing all of the elements arranged according to their properties
atomic mass
proton
neutron
periodic table
28
Multiple Choice
a one or two letter abbreviation for the element
chemical symbol
atomic number
atomic mass
29
Multiple Select
Which TWO of the following subatomic particles are inside the nucleus?
protons
electrons
neutrons
There are no subatomic particles in the nucleus of an atom.
30
Multiple Choice

Tin (Sn) and Lead (Pb) are both members of the came group as carbon, as shown by the periodic table, which of the following elements would also be a member of this group and why?
Iron(Fe), because it is a metal like tin and lead
Krypton (Kr) because it is an inert gas like tin and lead
Calcium (Ca) because it has reactivity similar to that of tin and lead
Silicon (Si) because it has conductivity similar to that of tin and lead
31
Multiple Choice
32
Multiple Choice

33
Multiple Choice
In the modern periodic table, elements are arranged by:
atomic mass
atomic number
valence electrons
number of isotopes
34
Multiple Choice
Which of the following is an element?
salt
oxygen
vinegar
water
35
Multiple Choice

36
Multiple Choice
Which of the two elements below have similar properties?
Chlorine (Cl) and Zinc (Zn)
Sodium (Na) and Fluorine (F)
Potassium (K) and Francium (Fr)
Magnesium (Mg) and Helium (He)
37
Multiple Choice

38
Multiple Choice

A model of the most basic unit of oxygen is shown. Which of the following is true of oxygen?
Oxygen is an element because its most basic unit is a single type of atom
Oxygen is a compound because its most basic unit is a single type of atom
Oxygen is a mixture because its most basic unit contains more than one particle
Oxygen is a compound because its most basic unit contains more that one type of particle
39
Multiple Choice
40
Multiple Choice
a positively-charged particle in the atoms nucleus
proton
neutron
electron
41
Multiple Choice
42
Multiple Choice
How is an element different from a compound?
an element is highly reactive, and a compound is stable
an element exists in any state, and a compound is only a liquid
an element contains multiple substances, and a compound contains only one pure substance
an element is made of a single type of atom, and a compound is made of two or more atom types
43
Multiple Choice
44
Multiple Choice

Which label correctly identifies the nucleus of the atom?
A
B
C
D
45
Multiple Choice

46
Multiple Choice

This is a chemical formula for glucose, a type of sugar. What statement is true about glucose?
Glucose is an element, because it can be separated into smaller components
Glucose is an element, because it is made up of more than one kind of atom
Glucose is a compound, because it is made up of more than one element
Glucose is a compound, because it is made of protons, neutron and electrons
47
Multiple Choice

Which item on this element square represents the atomic mass?
13
Al
Aluminum
26.981538
48
Multiple Choice
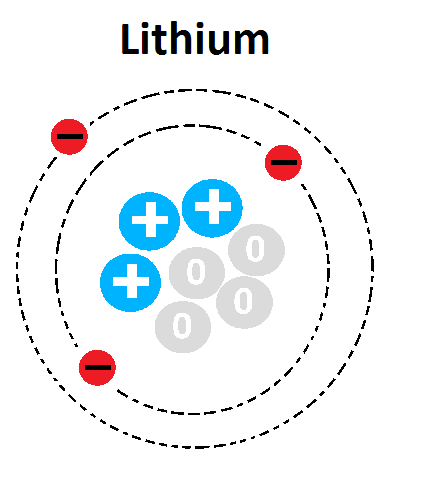
What is the mass number of this atom?
1
3
4
7
49
Multiple Choice

50
Multiple Choice

Nitrogen is a gas at room temperature and standard pressure. It is also a Group 15 element as shown. Which statement about the other elements in Group 15 is true?
They look similar to oxygen
They react in a way similar to how nitrogen reacts
They have the same density as nitrogen when under the same conditions
They are, like nitrogen, gases at room temperature and standard pressure
51
Multiple Choice

52
Multiple Choice
On the periodic table Li, Na, K and Rb are found in the same group because
they have the same number of neutrons
they have similar properties
they have the same number of electrons
they are compounds
53
Multiple Choice
54
Multiple Choice

Which elements in the periodic table have similar properties?
a metal and a noble gas
a metal and a non metal
two elements in the same group
two elements in the same period
55
Multiple Choice

56
Multiple Choice

57
Multiple Choice
58
Multiple Choice

Based on the periodic table of elements, which of the following sets of elements have similar properties?
Pb, Sn, Kr
Ne, N, O
Cl, Br, I
O, S, N
59
Multiple Choice
60
Multiple Choice
61
Multiple Choice
62
Multiple Choice
What is the atomic number of an element based upon?
mass of it's nucleus
number of electrons around it's core
number of protons in its nucleus
number of neutrons in its nucleus
63
Multiple Choice
If an atom has 24 protons and 24 neutrons, what is most likely the atomic mass of this element?
24
48
0
42
64
Multiple Choice
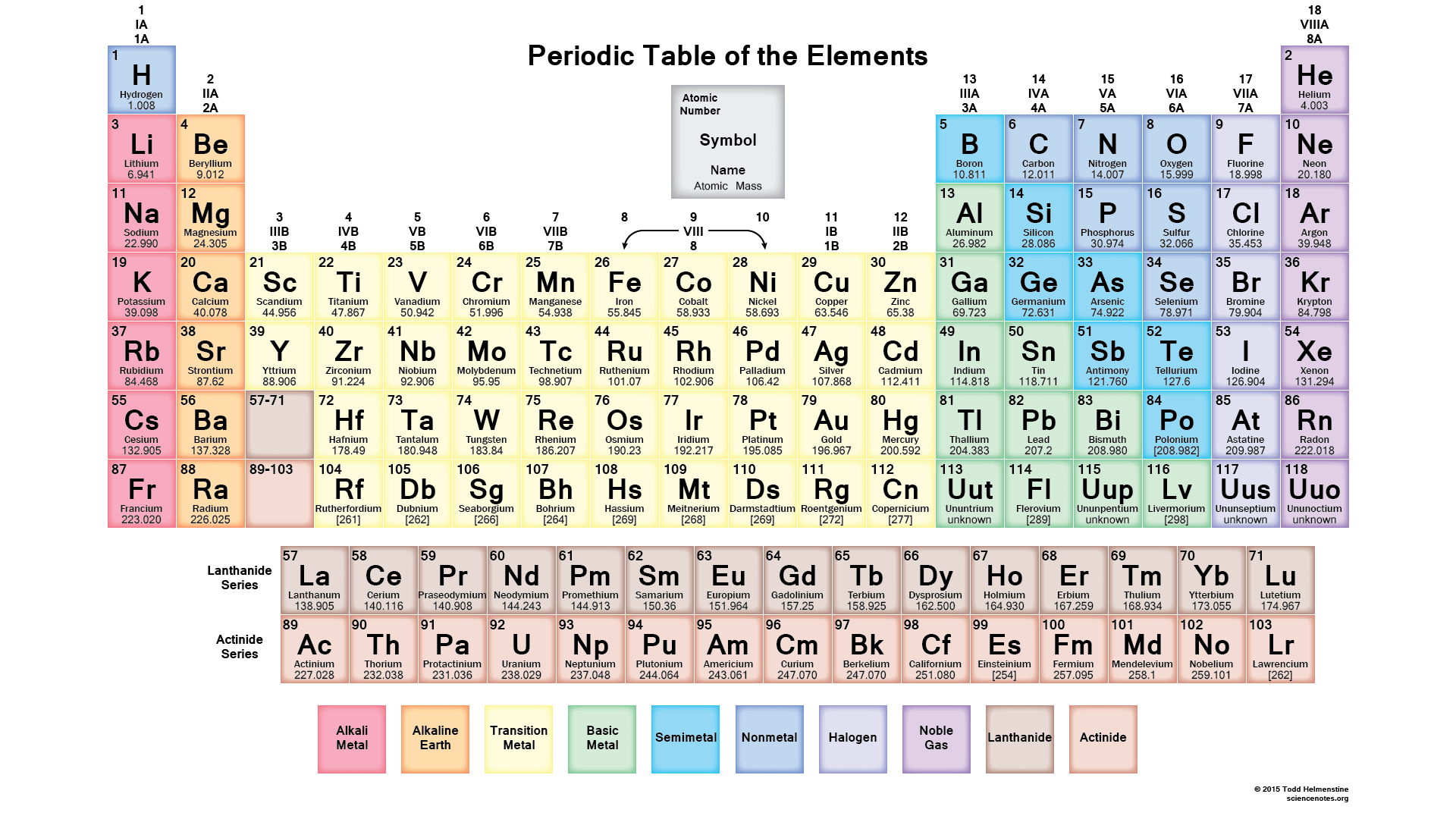
65
Multiple Choice

66
Multiple Choice
A chemist determines that the atom she is studying has eight negatively charged particles outside the nucleus. What does this indicate about the atom?
it is very unstable
it contains eight neutrons
it contains eight electrons
it is a negatively charged ataom
67
Multiple Choice

68
Multiple Choice
a chart showing all of the elements arranged according to their properties
atomic mass
proton
neutron
periodic table
69
Multiple Choice
70
Multiple Choice
Audra is looking at the diagram of an atom. Which particle(s) does she see inside the nucleus?
electrons only
protons only
electron and protons
protons and neutrons
71
Multiple Choice

A model of Nitrogen is shown, in this model, what do the blue spheres represent?
electrons
neutrons
photons
protons
72
Multiple Choice

A model of Nitrogen is shown, in this model, what do the blue spheres represent?
electrons
neutrons
photons
protons
73
Multiple Choice
The smallest unit found in silver is
element
nucleus
atom
cell
74
Multiple Choice
Which subatomic particle or particles have a negative charge?
protons and electrons
electrons and neutrons
electrons only
neutrons only
75
Multiple Choice
In which section of the periodic table could you find the most reactive metals?
the far left of the periodic table
the center of the periodic table
column 17 in the periodic table
the far right of the periodic table
76
Multiple Choice
Which best describes what happens when elements combine to form a compound?
the atoms in each compound dissolve
the atoms of both elements bind together
the nuclei in the atoms of both elements split and combine with the nuclei of other atoms
The subatomic particles in the atoms of both elements change to resemble each other
77
Multiple Choice

What period is this element in?
1
18
10
2
78
Multiple Choice
Kathy's teacher puts this question on the board:
All matter is made of ______, which may contain ______.
Atoms, molecules
Elements, atoms
molecules, atoms
molecules, elements
79
Multiple Choice
Everything in the periodic table is arranged by
the alphabet
their properties
their subatomic particles
their discovery date
80
Multiple Choice
Atoms combine in many different ways to form different substances. Iron is an element. Brass is not an element, instead brass is a combination of two elements. what is the difference between iron and brass?
Iron, but not brass, consists of atoms
Brass, but not iron, is a metallic substance
Iron is a compound, brass is not
Brass is a compound, iron is not
81
Multiple Choice

What group and period is sodium (Na) located?
period 1 and group 3
period 3 and group 1
period 2 and group 3
period 3 and group 2
82
Multiple Choice
Compounds are made up of different kinds of atoms that are chemically combined. What do compounds have in common?
They have the same color
They have the same melting point
They can be broken down into simpler substances
They can't combine with one another to from more complex substances
83
Multiple Choice

Which subatomic particle has a negative charge?
proton
neutron
electron
atom
84
Multiple Choice
Which of the following is a property of an element?
Elements have their own individual characteristic properties
Elements have at least two different types of atoms
Elements have the ability to change into a different element
Elements can be separated from one another
85
Multiple Choice
Which of the following is an element?
salt
oxygen
vinegar
water
86
Multiple Choice
A student notices that a basketball seems to act differently depending on whether the game is being played on a court with a wooden floor, on a concrete driveway, or on a blacktop in the park. they student hypothesizes that the surface on which a ball is bounced affects the height of the bounce.
How can the student design an experiment to test the hypothesis?
The student should drop the same ball from 3 different heights and have someone measure the height of each bounce
The student should drop 3 different balls from the same height and have someone measure the height of each bounce
The student should drop the same ball from the same height on 3 different surface and have someone measure the height of each bounce
The student should drop 3 different balls from the same height on 3 different surfaces and have someone measure the height of each bounce
87
Multiple Choice

88
Multiple Choice
Ena is measuring the pH of various liquids to see if they are acids or bases. Although she is following the same procedure each time, she notices that when she tests a particular liquid she obtains results she does not expect. She is not sure that the result is accurate.
What should Ena do to improve the reliability of the data she collects?
change her hypothesis
perform repeated trials on each liquid
measure a smaller volume of each sample
research more information about acids and bases
89
Multiple Choice

The graph shows the number of pythons removed from the Florida Everglades over a period of time.
Based on the data collected, which conclusion is the most logical?
Python presence is increasing each year in certain parts of Florida?
Python presence is due to the low water levels in surrounding lakes
Python presence in evidence of an exception to the theory of natural selection
python presence is a result of the lack of human ecosystem intervention in Florida
90
Multiple Choice
Devin is doing an investigation to address the question of how temperature affects the rate of crystal growth. He has 6 jars and adds the same amount of warm water to each jar. Then he adds salt to each jar, enough to make a saturated solution, and stirs until the salt dissolves completely. He observes the jars for several days.
Which experimental design allows for the collection of the evidence Devin needs to address this question?
He leaves all 6 jars on the counter and records the temperature and day the crystals appear
He puts all 6 jars in the fridge, and records the day and temperature the crystals appear
he puts 3 jars in the fridge and 3 jars at room temperature and, records the day the crystals appear
He puts 5 jars at room temperature and 1 in the fridge, and records the day the crystals appear
91
Multiple Choice
Which statement BEST describes a scientific hypothesis?
a scientific fact
a scientific theory
an inference based on scientific data
a testable scientific explanation based on observation
92
Multiple Choice
The vertical (up and down) columns in the Periodic Table are called
groups
towers
periods
atomic numbers
93
Multiple Choice
94
Multiple Choice

95
Multiple Choice

96
Multiple Choice
97
Multiple Choice
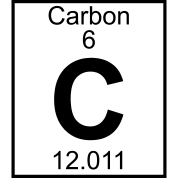
98
Multiple Choice
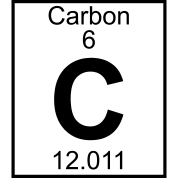
99
Multiple Choice

The smallest particle into which an element can be divided and still be the same element is a(n)
proton
neutron
electron
atom
100
Multiple Choice

Which subatomic particle has a positive charge?
proton
nuetron
electron
atom
101
Multiple Choice

Which subatomic particle has a negative charge?
proton
neutron
electron
atom
102
Multiple Choice

Which subatomic particle has a neutral (0) charge?
proton
nuetron
electron
atom
103
Multiple Choice

Why is the overall charge of an atom neutral (0)?
They have the same number of protons and neutrons.
They have the same number of protons and electrons.
They have the same number of electrons and neutrons.
104
Multiple Select
Which TWO of the following subatomic particles are inside the nucleus?
protons
electrons
neutrons
There are no subatomic particles in the nucleus of an atom.
105
Multiple Choice

106
Multiple Choice

107
Multiple Choice
108
Multiple Choice
109
Multiple Choice
110
Multiple Choice
111
Multiple Choice
112
Multiple Choice

113
Multiple Choice
114
Multiple Choice
115
Multiple Choice
Atoms and the Periodic Table

Show answer
Auto Play
Slide 1 / 115
SLIDE
Similar Resources on Wayground

108 questions
NE Chat Training
Presentation
•
KG

116 questions
8.3.1-Electricity
Presentation
•
8th Grade

109 questions
Noncontact Forces: Gravity
Presentation
•
6th - 8th Grade

108 questions
7 SINIF 2. ÜNİTE
Presentation
•
7th Grade

113 questions
Berfikir Komputasional
Presentation
•
9th Grade

111 questions
7 grade unit 3
Presentation
•
7th Grade

109 questions
67 67
Presentation
•
KG

108 questions
The Warm Christmas
Presentation
•
8th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade

48 questions
8th Grade NC Science EOG Review 2
Quiz
•
8th Grade

19 questions
Renewable and Nonrenewable Resources (CK)
Quiz
•
8th Grade

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade

10 questions
Exploring Weather Influences and Map Reading
Interactive video
•
6th - 10th Grade

43 questions
Amplify Earth's Changing Climate Unit Review
Quiz
•
6th - 8th Grade