

Physical and Chemical Reactions Lesson
Presentation
•
Science
•
8th Grade
•
Hard
Joseph Anderson
FREE Resource
27 Slides • 43 Questions
1
Chemical Reactions

2
Fill in the Blanks
Type answer...
3
Multiple Choice
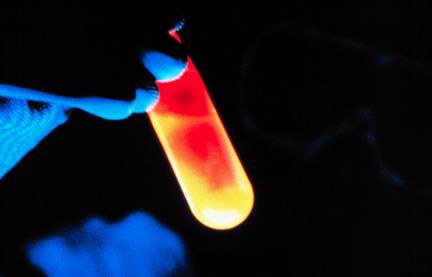
4
Multiple Choice
Bases turn blue litmus paper red.
True
False
5
Multiple Choice
Which of the following is an example that includes evidence of a chemical reaction?
A sample of zinc is placed in water and it settles to the bottom
A solid block of ice is heated and it completely melts into a liquid.
Sugar that is burned gives off an odor and turns brown and then black.
A tomato is placed into a blender and chopped up into smaller pieces.
6
Multiple Choice
Which of the following is a base?
baking soda in water
orange juice
vinegar
lemonade
7
Multiple Choice
8
Multiple Choice
Which of the following pH values represents a base?
2.1
4.6
6.8
8.1
9
Multiple Choice
Which of the following examples shows that a chemical reaction has occurred?
A rock breaks into smaller pieces when it is struck with a hammer.
Rust forms on a hammer that has been left outside to long.
A cup of water turns pink when a few drops of red food coloring are added.
A solid is formed when heat is removed from a sample of water.
10
Multiple Choice
Which of the following is an acid?
vinegar
bleach
sugar in water
starch in water
toothpaste in water
11
Multiple Choice
12
Multiple Choice
Which of the following are properties of acids?
They conduct electricity when dissolved in water
They taste sour
They react with metals to produce hydrogen gas
All of the answer choices are correct
13
Multiple Choice
14
Multiple Choice
When an acid is dissolved in water, it turns red litmus paper blue.
True
False
15
Multiple Choice
In a ___________change, a substance changes into a different substance.
Physical
Chemical
16
Multiple Choice

Milk is a very weak acid. What might its pH value be?
6.5
7.8
4.2
12.2
17
Open Ended
What is a Chemical Reaction?
18
Multiple Choice
Which of these pH values represent an acid?
4
8
10
12
19
A chemical reaction occurs when substances interact with each other . Bonds are broken and new ones are formed

20
Chemical Reactions results in Chemical change

21
Evidence of Chemical Change include
Temperature Change
Formation of Precipitate
Color Change
Formation of gases
22
What is the statement below saying?
2H2 + O2 = 2H2O
23
Chemical Reactions are represented by Chemical Equation
24
Open Ended
What is an Equation?
25
In Math an equation is a statement where both side are equal
An equation is separated by an equal sign
5+5=10
When we add 5 and 5 on the left side we get 10 which is the same on the right side

26
Equation in Math deals with numbers and variables
Chemical Equation deals with elements and compounds
Numbers are sometimes placed below elements
These numbers are called subscript .
They tell how many atoms of the elements are present
Numbers placed before an element or compound are called Coefficient

27
A chemical Equation has reactants which are on the left side, products on the right and an arrow
THe reactants are the substances which are interacting
The Products are what are formed
The Arrow shows the direction of Reaction

28
Multiple Choice
What is the number before a chemical formula called?
Ex: 2H2 + O2 --> 2H2O
Coefficient
Subscript
Atom
Equation
29
Multiple Choice
What is the little number after an element in a chemical equation called.
Example: H2
Coefficient
Subscript
Atom
Equation
30
Multiple Choice
What is the role of the arrow sign in a chemical reaction?
Making sure atom counts on each side are the same.
Separating products and reactants.
Showing which substances were in the container first and which substances were added later.
Showing which substances are solids and which are liquids or gases.
31
Multiple Choice
Identify the product(s) in the following chemical reaction:
Chromium oxide + Carbon → Chromium + Carbon monoxide
Chromium and carbon.
Chromium and carbon monoxide.
Chromium and carbon dioxide.
Chromium oxide and carbon.
32
Multiple Choice
Identify the reactants(s) in the following chemical reaction:
Chromium oxide + Carbon → Chromium + Carbon monoxide
Chromium and carbon.
Chromium and carbon monoxide.
Chromium and carbon dioxide.
Chromium oxide and carbon.
33
The number of similiar elements on the left should always equal to those on the right
After a chemical reactions that is not the case
Example when a piece of paper is burned there is far less matter than what previously existed
34
The matter is not lost it just changed formed
The Law of Conservation of Matter state that Matter is neither created or destroyed, but just change in form
For example a burning paper releases gases and water into a different form
It is not destroyed just changed
35
Closed System
In a closed system nothing is lost as all the atoms of an element at the beginning can be accounted for at the end because nothing escaped
36
Open System
In an open system matter is allowed to escaped to its surrounding
The number of atoms of each element on either side will be different
This would not satisfy the Law of Conservation of Matter since it would appear that matter was lost
37
Multiple Choice
Are the number of atoms of each elements of the reactants the same as the products ?...
Al + O2 --> Al2O3
Yes!
No!
38
Balancing Equations
To satisfy the Law of Conservation of Matter equations are balanced
To balance an Equation Coefficient are added
Coefficient are used to multiply the subscript (number of atoms ) of each elements until both sides are equal
39

Google Slides - create and edit presentations online, for free.
You can open this webpage in a new tab.
40

How to Balance a Chemical Equation : 7 Steps (with Pictures) - Instructables
You can open this webpage in a new tab.
41
42
Balance this equation
Li + Cl2 -> LiCl
43
Balance this Equation
MgCl2 --> Mg4+ Cl2
44
Multiple Choice
Balance this equation:
MgCl2 --> Mg4+ Cl2
It's already balanced.
4MgCl2 --> Mg4+ 4Cl2
4MgCl2 --> Mg4+ 5Cl2
45
Multiple Choice
Balance this equation:
2Li + Cl2 -> LiCl
2Li + Cl2 -> 4LiCl2
2Li + Cl2 -> LiCl2
2Li + Cl2 -> 2LiCl
46
Multiple Choice
Is the following equation balanced or unbalanced ?
N2 +3 H2 --> 2NH3
Balanced
Unbalanced
47
Multiple Choice
Al + O2 --> Al2O3
48
Multiple Choice
_ H2 + O2 -> 2 H2O
49
Multiple Choice
Which chemical equation best represents the Law of Conservation of Mass?
H + O → H2O
H2 + O2 → H2O
2H2 + O2 → 2H2O
H2 + O2 → 2H2O2
50
Multiple Choice

Observe the reaction shown here. The formula for this equation is N2 + H2 → NH3 . Does this represent a balanced equation that obeys the Law of Conservation of Mass?
No. The reactants and products are not equal in numbers of atoms of each element.
Yes. The reactants and products are equal in numbers of atoms of each element.
51
Multiple Choice

Is this equation balanced?
Yes
No
52
Multiple Choice
53
Multiple Choice
__NaClO3 --> __NaCl + __O2
2,2,2
2,2,3
3,2,2
1,2,3
54
Multiple Choice
__Na + __Cl2 --> __NaCl
1,1,2
2,1,2
2,1,1
already balanced
55
Types of Chemical Reaction
Synthesis
Decomposition
Replacement

56
Synthesis
Two or more elements joined together to become a compound

57
2Na + Cl2 → 2NaCl
2NO + O2 → 2NO2
2 H2O + O2 → 2 H2O
58
Decomposition
A compound is broken down into individual elements

59
H2CO3 → H2O + CO2
2 H2O2 → 2 H2O + O2
2 H2O → 2 H2 + O2
60
Single Replacement
One element replaces one element in a compound

61
2K + 2H2O → 2KOH + H2
F₂ + 2NaCl → 2NaF + Cl₂
62
Combustion
Combustion is a type of chemical reaction where Oygen breaks down compounds

63
Multiple Choice
Which type of chemical reaction has two or more reactants to make one product?
combustion
single displacement
double displacement
synthesis
decomposition
64
Multiple Choice
Combustion reactions will always involve
Oxygen and a solid
Liquid nitrogen and heat
Oxygen and heat/energy
CO and H2O
65
Multiple Choice
What reaction has the following general formula:
AB --> A + B
Combination
Decomposition
Single Replacement
Double Replacement
Combustion
66
Multiple Choice
Which type of chemical reaction has two or more reactants to make one product?
combustion
single displacement
double displacement
synthesis
decomposition
67
Multiple Choice
68
Multiple Choice
In a chemical reaction, everything to the right of the arrow is called ______________.
Products
Yields
Molecules
Reactants
69
Multiple Choice
Which phrase describes what happens to atoms in a chemical reaction?
never lost or gained, just rearranged
sometimes lost, never rearranged
sometimes lost, gained, or rearranged
lost or gained, never rearranged
70
Multiple Choice
What reaction has the following general formula:
A + B --> AB
Combination
Decomposition
Single Replacement
Double Replacement
Combustion
Chemical Reactions

Show answer
Auto Play
Slide 1 / 70
SLIDE
Similar Resources on Wayground

61 questions
Ecological Relationships
Presentation
•
8th Grade

63 questions
Form 3 Reading Practice
Presentation
•
9th Grade

65 questions
Tuesday JAnuary 14 Lesson
Presentation
•
9th Grade

67 questions
Stratigraphy and Dating Rocks
Presentation
•
8th Grade

64 questions
Newton's Second Law of Motion
Presentation
•
7th - 8th Grade

62 questions
Drones Lesson Activity
Presentation
•
8th Grade

67 questions
Grade 8 Matter Summer School
Presentation
•
8th Grade

63 questions
Biogeochemical Cycles Review
Presentation
•
9th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

30 questions
GMAS Physical Science Review
Quiz
•
8th Grade

10 questions
Exploring the Rock Cycle
Interactive video
•
6th - 8th Grade

18 questions
Carbon Cycle
Quiz
•
8th Grade

10 questions
Exploring Earth's Seasons and Their Causes
Interactive video
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

10 questions
Evidence of Evolution
Presentation
•
6th - 8th Grade

20 questions
8th Grade Science NC EOG Review
Quiz
•
8th Grade