

Physical Science 4-1: Electrons & Chemical Bonding
Presentation
•
Science
•
9th - 12th Grade
•
Easy
+1
Standards-aligned
Abby Fancsali
Used 2+ times
FREE Resource
13 Slides • 19 Questions
1
Electrons & Chemical Bonding

2
Multiple Choice
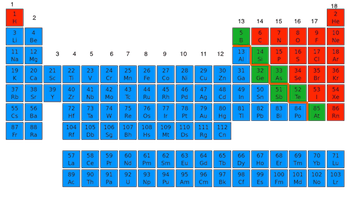
Left side
Right Side
Center
They are scattered throughout the table
3
Drag and Drop
row
isotope
4
Multiple Choice
What is the name of group 1 on the Periodic Table?
halogens
noble gases
alkali metals
alkaline earth metals
5
Multiple Choice
What group number are the halogens found?
18
1
2
17
6
Lesson Objectives
Describe what Chemical Bonding is
Identify what Valence Electrons are
Explain the role of valence electrons in Bonding
Draw an electron dot diagram

7
All Matter is made up of either individual atoms or molecules and compounds
Molecules and Compounds are held together by bonds
Chemical Bonding: The Joining of atoms to form a new stable substance
Chemical Bond: An interaction that holds atoms or ions together
Can act between different elements or the same type of element
All chemical bonds form when electrons in an atom interact
What is a Chemical Bond?
8
Recall: Electrons in atoms arrange themselves in energy levels that can hold a certain number of electrons
Closest to the nucleus = 2 electrons
Every other level = 8 electrons
We split electrons into two main groups
Valence Electrons: The electrons on the outermost level of an atom
Core Electrons: The electrons on the inner and middle levels of an atom
Atoms are most stable when they have 8 valence electrons
Will react with other atoms to try and make it to 8
The types of bonds an atom forms depend on the number of valence electrons it has.
Electrons in Atoms
9
Multiple Choice
The interaction between atoms or ions that holds them together is a....
Mixture
Compound
Chemical Bond
Formula
10
Multiple Choice
To be bigger
11
Multiple Choice
12
Fill in the Blanks
Type answer...
13
Recall: The atomic number tells you how many protons are in an element
All elements are neutral, so they have to have the same number of electrons
When you draw a diagram of the atom, the electrons on the final ring are the valence electrons
Determining the Number of Valence Electrons

14
The group number of an element on the periodic table can be used to find the number of valence electrons it has
Elements in the same group have the same number of valence electrons
Group 1= 1 valence electron
Group 2= 2 Valence electrons
Groups 13-18= Group # -10
Exception: Helium only has two valence electrons
Using the Periodic Table to Find Valence Electrons
15
The closer an atom is to having 8 valence electrons in its outermost shell, the more likely it is to bond
Can get to 8 either by gaining or losing electrons
The Noble Gases already have 8 valence electrons, so they do not form bonds at all
Why Do Atoms Bond?
16
Multiple Choice
An electron that is found in the outermost shell of an atom.
An electron that is found in the innermost shell of an atom.
An electron that is found in the middle shell.
An electron that is found in the nucleus of the atom
17
Multiple Choice

This is a Bohr Diagram of Aluminum. How many valence electrons does Aluminum have?
3
4
5
8
18
Multiple Choice
Using your Periodic Table, How many valence electrons does Lithium (Li) have?
8
1
5
2
19
Multiple Choice
How many valence electrons does Tin (Sn) have?
8
7
5
4
20
Lewis Dot Structures
Sometimes we just want to diagram out the valence electrons in an atom
They are the most important in reactions
In order to do this, we draw Lewis Dot Structures
Show the element Symbol with the number of Valence Electrons

21
Electron Dot Diagrams Practice Problem 1
Draw the Lewis Dot Structure for the element Sodium
Step 1: Identify the number of Valence electrons
Sodium is in Group 1, so it has 1 Valence Electron
Step 2: Draw the Element Symbol
Step 3: Place the Valence electrons around the Symbol
Start at the top and add one to each side before doubling up
22
Electron Dot Diagrams Practice Problem 2
Draw the Lewis Dot Structure for the element Fluorine
Step 1: Identify the number of Valence electrons
For Groups 13-18: Valence electrons = Group # - 10
23
Fill in the Blanks

Type answer...
24
Electron Dot Diagrams Practice Problem 2
Draw the Lewis Dot Structure for the element Flourine
Step 1: Identify the number of Valence electrons
Sodium is in Group 17
Valence electrons = Group # - 10
Step 2: Draw the element Symbol
Step 3: Place the Valence electrons around the Symbol
Start at the top and add one to each side before doubling up
25
Electron Dot Diagrams Practice Problem 3
Draw the Lewis Dot Structure for the element magnesium
Step 1: Identify the number of Valence electrons
26
Fill in the Blanks

Type answer...
27
Electron Dot Diagrams Practice Problem 3
Draw the Lewis Dot Structure for the element magnesium
Step 1: Identify the number of Valence electrons
Magnesium is in group 2, so it has two valence electrons
Step 2: Draw the element Symbol
Step 3: Place the Valence electrons around the Symbol
Start at the top and add one to each side before doubling up
28
Multiple Choice
What determines the number of valence electrons an element it has.
period/row
group/column
Atomic Number
Atomic Mass
29
Multiple Choice

This is a correct dot diagram for nitrogen (N)
true
false
30
Multiple Choice

This is a correct dot diagram for neon (Ne)
true
false
31
Multiple Choice

Which Element could this be the dot diagram of
32
Multiple Choice
What is the number of valence electrons needed for atoms to be stable?
1
2
8
18
Electrons & Chemical Bonding

Show answer
Auto Play
Slide 1 / 32
SLIDE
Similar Resources on Wayground

28 questions
Evidence of Evolution
Presentation
•
9th - 12th Grade

24 questions
Earth's Resources
Presentation
•
9th - 11th Grade

24 questions
Food Label questions
Presentation
•
9th - 12th Grade

26 questions
Cell Cycle Regulation
Presentation
•
9th - 12th Grade

25 questions
Physical and Chemical Changes Review
Presentation
•
8th Grade

26 questions
Introduction to Matter
Presentation
•
9th - 12th Grade

23 questions
3.3 Fluids
Presentation
•
9th - 12th Grade

23 questions
Forensics: Observation pt 2
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade