

Chapter 2 Example Explainations and Practice
Presentation
•
Science
•
9th - 12th Grade
•
Easy
Standards-aligned
Chelsea McBride
Used 1+ times
FREE Resource
32 Slides • 50 Questions
1
Chemistry: Chapter 2 Explained
By Chelsea McBride
2
Mixtures
When we talk about types of matter, we can place the matter into categories; mixtures and pure substances. Today is an introduction into mixtures.

3
Homogeneous Mixtures
The first type of mixture is homogenous. "Homo-" means the same so a homogenous mixture is the same throughout the entire matter.

4
Heterogeneous Mixtures
The second type of mixture is heterogeneous. "Hetero-" means different so a heterogeneous. mixture is different throughout the entire matter.

5
Multiple Choice

Is cereal homogeneous or heterogeneous?
homogeneous
heterogeneous
6
Multiple Choice

Is the drink "Hawaiian Punch" homogeneous or heterogeneous?
homogeneous
heterogeneous
7
Multiple Choice

Are M&M's homogeneous or heterogeneous?
homogeneous
heterogeneous
8
Multiple Choice

Is a cheeseburger homogeneous or heterogeneous?
homogeneous
heterogeneous
9
Multiple Choice

Is coffee homogeneous or heterogeneous?
homogeneous
heterogeneous
10
Multiple Choice

Is this cake homogeneous or heterogeneous?
homogeneous
heterogeneous
11
Multiple Choice

Is lemonade homogeneous or heterogeneous?
homogeneous
heterogeneous
12
Multiple Choice

Is guacamole homogeneous or heterogeneous?
homogeneous
heterogeneous
13
Multiple Choice

Is water (H2O) homogeneous or heterogeneous?
homogeneous
heterogeneous
Wait, this is a trick question. H2O is a chemical compound.
14
Pure Substances
There are 2 types of pure subtances: elements and compounds.

15
Elements
ALL are listed on the periodic table
Made of only 1 type of atom
Cannot be broken down with a physical or chemical changes
Have consistent properties

16
Compounds
Are made of two or more types of atoms chemically combined
Can be broken down with chemical changes, but not physical
Have consistent properties

17
Multiple Select
Select all of the ELEMENTS from the list below.
Water
Aluminum
Oxygen
Air
Copper
18
Multiple Select
Select all the COMPOUNDS from the list below.
Table Salt (NaCl)
Air
Oxygen (O2)
Carbon Monoxide (CO)
Baking Soda
19
Multiple Choice
Which describes COMPOUNDS?
made of one type of atom
made of two or more types of atoms
20
Multiple Select
Select all of the ELEMENTS shown below.
21
Multiple Select
Select all the COMPOUNDS shown below.
22
Multiple Choice
Which best describes ELEMENTS?
made of only 1 type of atom
made of 2 or more types of atoms
23
Multiple Select
Select all the ELEMENTS shown below.
24
Multiple Select
Select all the COMPOUNDS shown below.
25
Multiple Select
Select all the COMPOUNDS shown below.
26
Density
You know, just a general measure of how much "stuff" is in there, very technical...

27
Particle Diagrams and Density
We can imagine the "stuff" as particles and recognize that both the amount of particles (mass) and the space they occupy determine the density
Let's compare and discuss A and B...
Now let's compare and discuss A and C...

28
Multiple Choice

Which diagram shows the MOST dense object?
left
center
right
29
Multiple Choice

Which diagram shows the LEAST dense object?
left
center
right
30
Multiple Choice

Rank the objects from MOST dense to LEAST dense.
left, center, right
right, center, left
left, right, center
right, left, center
center, left, right
31
Calculating Density
This is in the Edpuzzle video for the week and the Notes so make sure you get it down!
mass is typically in units of g or kg
volume of liquids is in mL
volume of liquids is in cm3
density has a "combined" unit of g/mL or g/cm3

32
Practice calculating density
33
Multiple Choice
A block of aluminum occupies a volume of 15.0 mL and weighs 45.0 g. What is its density?
3 g/mL
0.3333 g/mL
675 g/mL
I have no idea
34
Multiple Choice
A sample of liquid has a mass of 300 g and occupies 50 mL in a graduated cylinder. What is the density?
15,000 g/mL
.16667 g/mL
6 g/mL
Yup, still no clue
35

36

37
38
Multiple Choice

39
Multiple Choice
Isotopes of an element have a different number of...
Protons
Neutrons
Electrons
Mass
40

41
Multiple Choice
42
Multiple Choice

43
Multiple Choice
44

45

46

47
Multiple Choice
Using the atomic masses and relative abundance of the isotopes of nitrogen given below, determine the average atomic mass of nitrogen.
N-14: 14.003 amu; 99.63% N-15: 15.000 amu; 0.37%
14.003 amu
14.007 amu
14.506 amu
15.000 amu
48
Multiple Choice
Gallium has two stable isotopes. The mass of gallium-69 is 68.926 amu and has an abundance of 60.11%. The mass of gallium-71 is 70.925 amu and has an abundance of 39.89%. Which is the average atomic mass of gallium?
69.93 amu
69.72 amu
292.47 amu
139.85 amu
49
Fill in the Blanks
Type answer...
50
Intro to mole
By Monica Youssef
1. What is a mole?
2. Avogadro's number
51

52

53

54

55
Multiple Choice
Which of the following statements regarding the mole is NOT correct?
A mole is a counting unit
Equal to 6.02 x 1023 particles.
The concept of mole is similar to the concept of dozen
none of the above
56

57
Multiple Choice
When comparing a mole of oxygen and a mole of sulfur, how many atoms does each have?
oxygen has 16 grams of atoms
sulfur has 32.06 grams of atoms
both contains 6.02 x 1023 atoms
can not be determined
58
Multiple Choice
1 mole of Li
1 mole of Au
1 mole of Si
None, all are equal
59
Multiple Choice
1 mole H2O
1 mole Al(OH)3
1 mole NaCl
There are all the same
60
Multiple Choice
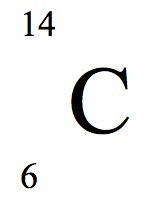
61

62
Multiple Choice
53
63.55g
126.9
6.02 x 1023
63
Multiple Choice
2
2 x (6.02 x 1023)
2 x (atomic mass)
2 ÷ (6.02 x 1023)
64
Multiple Choice
How many Carbon Atoms are in 4 moles of Carbon?
6.02 x 1023
1.33 x 1023
4.52
2.408 x 1024
65

66

67

68

69
Multiple Choice
A mole = Avogadro's number = 6.022x1023
true
false
70
Multiple Choice
Molar number
Scientist's number
71
Multiple Choice
Which of the following statements regarding the mole is correct?
A mole is a counting unit equal to 6.02 x 1023 particles.
The number of particles in a mole is known as Avogadro’s number.
1 mole = Avogdro's number
all of the above
72
Molar Mass
molar mass = 1 mole
CO2
C=12 O=16x2
44 g/mol

73
Multiple Choice
CO2 = 44 g/mol
How much would 3 moles of CO2 equal?
132
33
404
74
Multiple Choice

Molar Mass of CH4
12 g/mol
16 g/mol
4 g/mol
75
Multiple Choice
Molar Mass of K2CO3
138.205 g/mol
35 g/mol
78.2 g/mol
76
Multiple Choice
Molar Mass of Ag2O
231.7 g/mol
56.2 g/mol
734.4 g/mol
77
Mass converted to molar mass
to find the moles of any molecule
mole = mass/molar mass

78
20 grams of H2O is how many moles?
Molar mass of H2O = 18
Moles = 20/18
1.1 moles of H2O

79
Multiple Choice
88 grams of H2O (18 g/mol) is how many moles?
4.88 moles
.54 moles
6.67 moles
80
Multiple Choice
52 grams of O2 (32 g/mol) is how many moles?
1.63 moles
.615 moles
26 moles
81
Multiple Choice
73 grams of AlBr3 (267 g/mol) is how many moles?
.273 moles
263 moles
4.23 moles
82
Multiple Choice
800 grams of 2NH3 (34 g/mol) is how many moles?
23.52 moles
.0425 moles
834 moles
Chemistry: Chapter 2 Explained
By Chelsea McBride
Show answer
Auto Play
Slide 1 / 82
SLIDE
Similar Resources on Wayground

76 questions
Cell Energy
Presentation
•
9th Grade - University

75 questions
Bone Formation
Presentation
•
9th - 12th Grade

80 questions
APES Unit 2 Videos Biodiversity
Presentation
•
9th - 12th Grade

80 questions
Flow of Energy and Matter
Presentation
•
9th - 12th Grade

77 questions
Introduction to Biochemistry Lesson
Presentation
•
9th - 12th Grade

78 questions
2.1 Matter Notes
Presentation
•
9th - 12th Grade

76 questions
Pre AP Biology Natural Selection and Hardy Weinberg
Presentation
•
9th - 12th Grade

75 questions
Skeletal SYS
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

11 questions
EOC Domain 1: Cells
Presentation
•
9th - 10th Grade

14 questions
EOC Domain 2: Genetics
Presentation
•
9th - 10th Grade

10 questions
Exploring the Human Respiratory System
Interactive video
•
6th - 10th Grade