

PS A Semester Test Review
Presentation
•
Science
•
9th - 12th Grade
•
Practice Problem
•
Medium
+9
Standards-aligned
Rebecca Cmelik
Used 1+ times
FREE Resource
11 Slides • 17 Questions
1
Physical Science A
End-of-semester review
Text was AI-generated and edited by the teacher
2
Multiple Choice
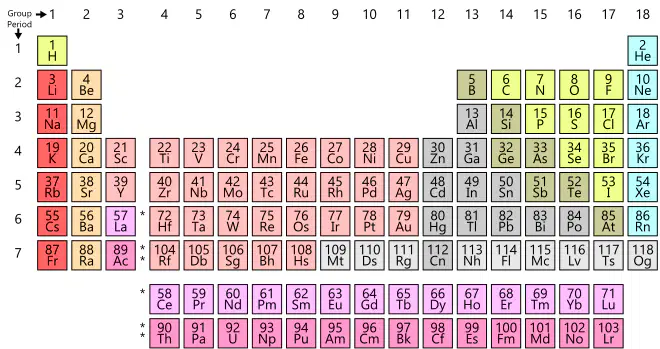
Two elements are in the same group (column) on the periodic table. These elements most likely have the same...
atomic mass
number of energy levels
number of valence electrons
state of matter at room temperature
3
Multiple Choice
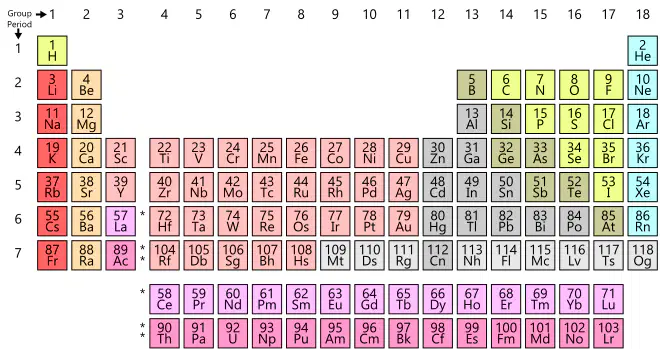
Which element is most likely to react in a similar way as fluorine (F)?
sodium (Na)
aluminum (Al)
argon (Ar)
chlorine (Cl)
4
Parts of an atom
Positive protons and neutral neutrons in the nucleus (center of atom)
Negative electrons in energy levels outside nucleus
Valence electrons are in the outermost energy level

5
Periodic table patterns
Elements are arranged by atomic number (number of protons)
Same group (column) = same number of valence electrons
Valence electrons help predict reactivity and properties

6
Multiple Choice

Which pair of elements is most likely to form an ionic compound?
nitrogen with nitrogen (N)
oxygen (O) and sulfur (S)
boron (B) with carbon (C)
lithium (Li) with chlorine (Cl)
7
Multiple Choice

How will carbon (C) react with oxygen (O)?
carbon will transfer valence electrons to oxygen
oxygen will transfer valence electrons to carbon
carbon and oxygen will share valence electrons
carbon and oxygen will form a sea of shared valence electrons
8
Why reactions happen
Atoms react to get full valence shells
In ionic bonds, metals transfer valence electrons to nonmetals
In covalent bonds, nonmetals share valence electrons
In metallic bonds, metals share a sea of valence electrons with each other


9
Multiple Choice

Which substance likely has the weakest electrical forces between particles?
diamond
salt
liquid water
oxygen gas
10
Multiple Choice

A substance is very hard and melts at a very high temperature.
What does this suggest about the forces between its particles?
They are weak
They are strong
They only exist at low temperatures
They are temporary
11
Structure → properties
Strong forces of attraction between particles lead to solids and high melting points. Examples include diamonds and table salt.
Weak forces of attraction between particles lead to gases and low melting points. Examples include carbon dioxide (dry ice) and oxygen gas.


12
Multiple Choice

A reaction feels cold to the touch. What does this indicate?
Energy was released by the reaction
Energy was absorbed by the reaction
No chemical bonds changed
Energy was destroyed
13
Drag and Drop
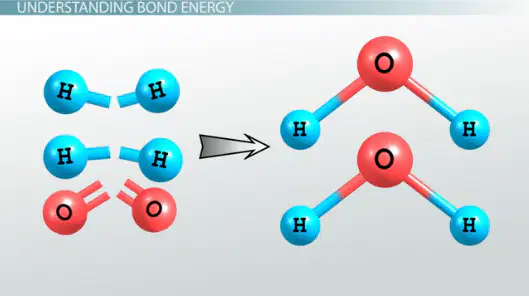
14
Energy in reactions
It requires energy to break chemical bonds.
Making new chemical bonds releases energy.
Exothermic reaction = more energy released than absorbed
Endothermic reaction = more energy absorbed than released


15
Multiple Choice

Which of these would probably slow down a chemical reaction?
Increasing the temperature
Decreasing the size of solid particles
Decreasing the temperature
Increasing the concentration of a reactant
16
Reaction Rates
Temperature: Higher temperatures usually increase the rate of a reaction
Concentration: Higher concentrations of a reactant usually increase the rate of a reaction
Surface area: Crushing solid reactants into smaller particles increases their surface area, which usually increases the rate of a reaction


17
Multiple Choice
Is this chemical equation balanced?
Na + Cl₂ → NaCl
Yes
No
18
Multiple Choice
Is this chemical equation balanced?
C + O₂ → CO₂
Yes
No
19
Chemical equations
Mass and atoms are not created or destroyed in chemical reactions.
Balanced equations show that the total number of each type of atom is the same before and after a reaction.

20
Multiple Choice
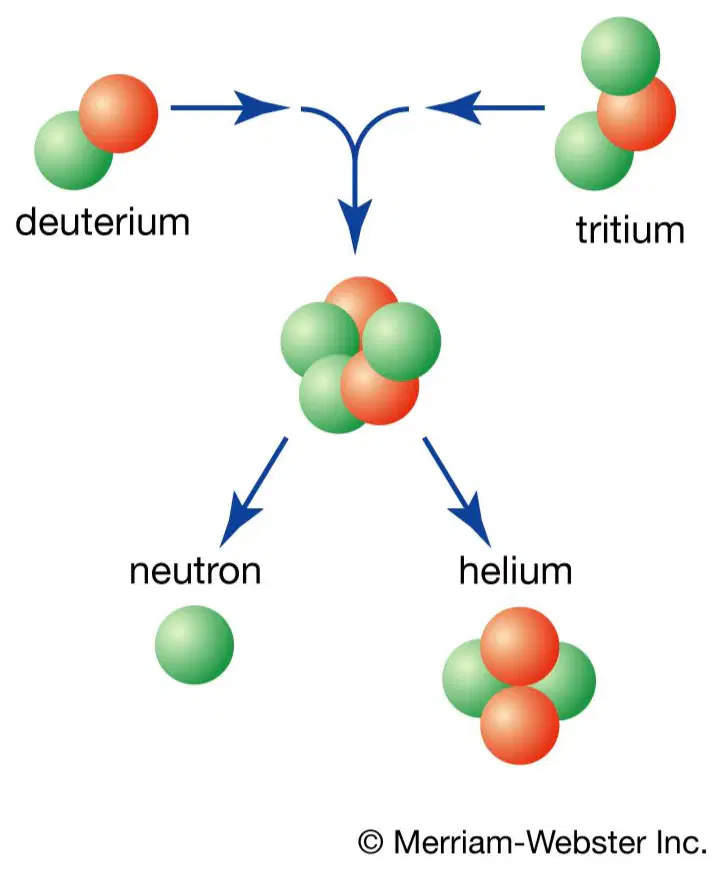
What type of reaction is happening in this picture?
(2 small atoms combining to form a larger atom)
nuclear fission
nuclear fusion
radioactive decay
chemical reaction
21
Multiple Choice
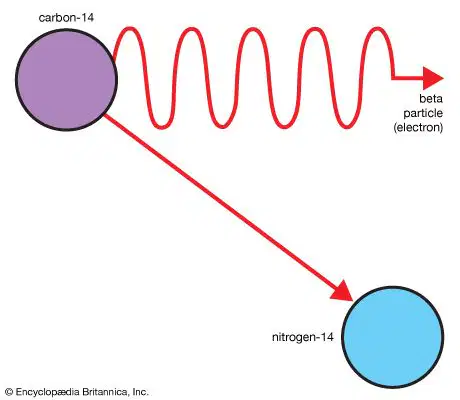
What type of reaction is happening in this picture?
(an atom releasing a particle)
nuclear fission
nuclear fusion
radioactive decay
chemical reaction
22
Nuclear reactions
Nuclear reactions involve changes to the nucleus of atoms. In nuclear reactions, tiny amounts of mass get converted into HUGE amounts of energy.
Fusion: Small atoms → larger atom + energy
Fission: Large atom → smaller atoms + energy
Nuclear decay: Nucleus releases particles and/or energy


23
Multiple Choice

What forces are acting on TV sitting on a desk?
No forces because the TV isn't moving
Gravity is pulling down
Gravity is pulling down and the table is pushing up
24
Multiple Choice

How much force is required to accelerate a car with a mass of 1,000 kg and an acceleration of 4 m/s²?
250 N
1,000 N
4,000 N
25
Force, mass, and acceleration
A force is a push or pull (Newtons, N). Mass is the amount of stuff an object has (kilograms, kg). Acceleration is how an object's motion changes (meters per second squared, m/s²).
Force = mass × acceleration

26
Multiple Choice

Which object has more momentum, a flying bird or a parked jet?
The flying bird
The parked jet
They have the same momentum.
It's impossible to tell from this information.
27
Multiple Choice
Object A: 5 kg and +5 m/s
Object B: 10 kg and -2 m/s
What is the total momentum of objects A and B?
+25 kg × m/s²
-20 kg × m/s²
+5 kg × m/s²
-5 kg × m/s²
28
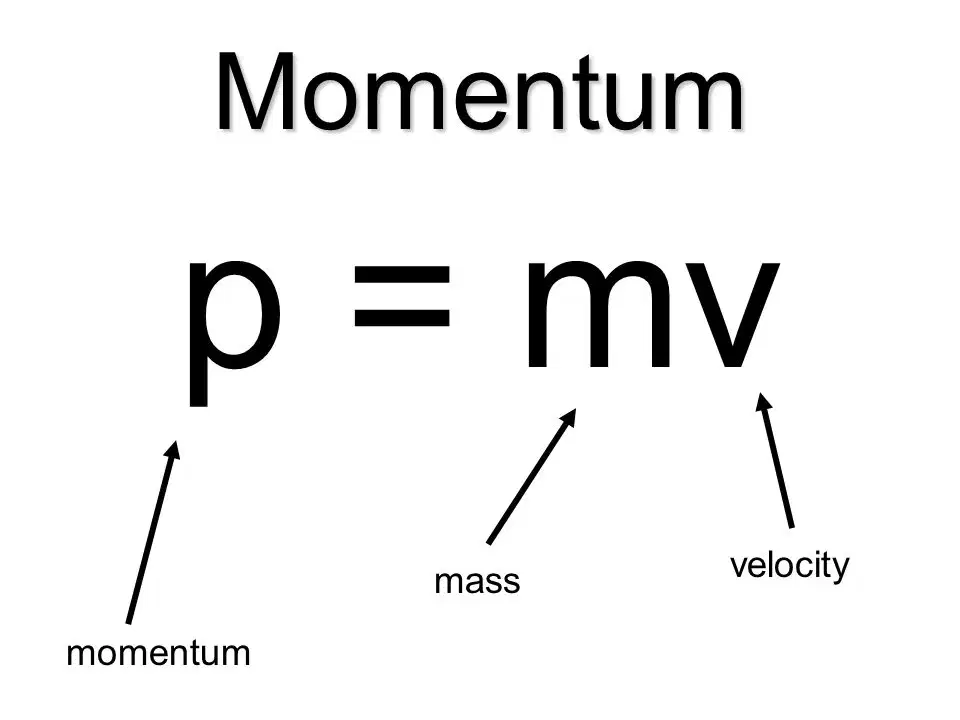
Conservation of momentum
Momentum (p) = mass (m) × velocity (v)
Momentum of a closed system stays the same, even if objects in the system collide and change their velocity.

Physical Science A
End-of-semester review
Text was AI-generated and edited by the teacher
Show answer
Auto Play
Slide 1 / 28
SLIDE
Similar Resources on Wayground

21 questions
Naming Ionic Compounds
Presentation
•
9th - 12th Grade

20 questions
The Ozone Layer
Presentation
•
9th - 12th Grade

23 questions
Acids and Bases
Presentation
•
9th - 12th Grade

24 questions
Nuclear Decay
Presentation
•
9th - 12th Grade

23 questions
Chemical Reactions - Chemical Changes and Conservation
Presentation
•
9th - 12th Grade

20 questions
Protein Synthesis Part 1
Presentation
•
9th - 12th Grade

22 questions
Unit 2.7 Energy Conservation
Presentation
•
9th - 12th Grade

19 questions
Enzymes Notes
Presentation
•
9th - 11th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Science

20 questions
Earth Day Trivia
Quiz
•
9th - 12th Grade

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

20 questions
Earth Day
Quiz
•
3rd - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

10 questions
Exploring Natural Selection Concepts
Interactive video
•
6th - 10th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

10 questions
Exploring Air Masses and Weather Fronts
Interactive video
•
6th - 10th Grade