
15 Q
10th - Uni

25 Q
12th - Uni

13 Q
12th - Uni

15 Q
12th - Uni

15 Q
10th - Uni

15 Q
10th - Uni

25 Q
10th - Uni

25 Q
10th - Uni

21 Q
9th - 12th

25 Q
10th - Uni

40 Q
9th - 12th

25 Q
10th - Uni

15 Q
10th - Uni

25 Q
10th - Uni

25 Q
10th - Uni

15 Q
10th - Uni

28 Q
12th

38 Q
9th - 12th

10 Q
10th - 12th

15 Q
10th - Uni
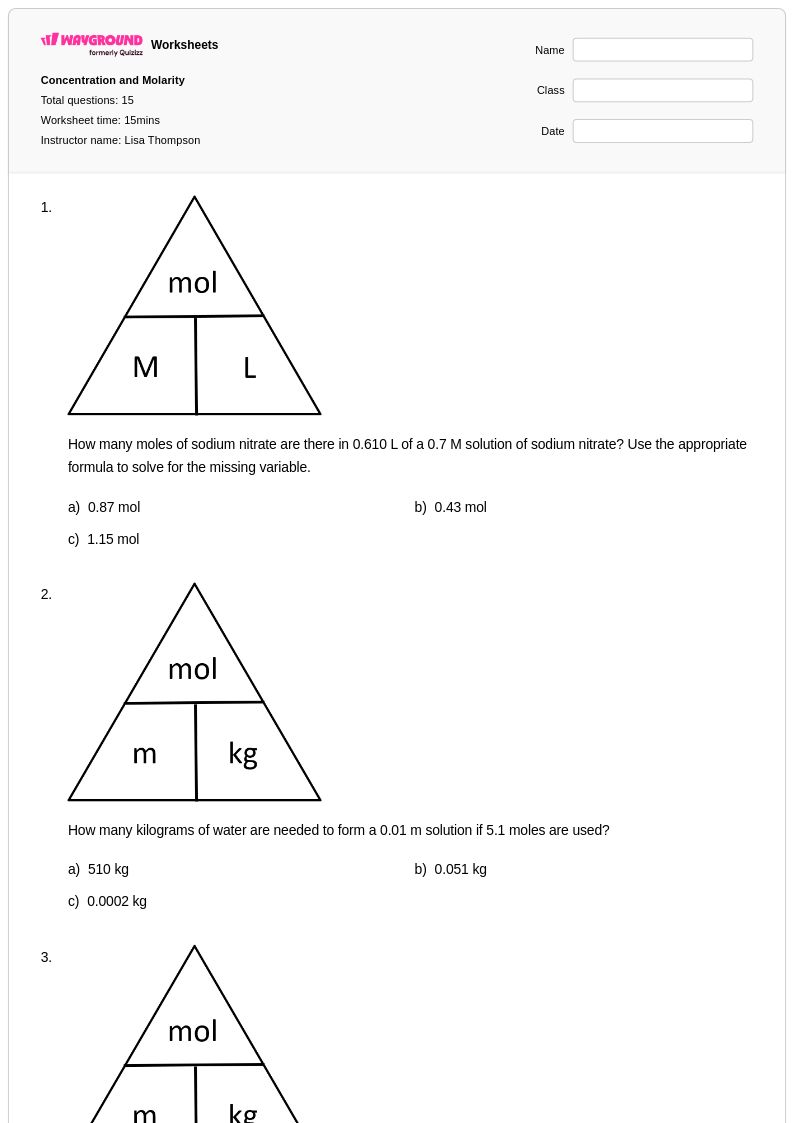
15 Q
10th - Uni
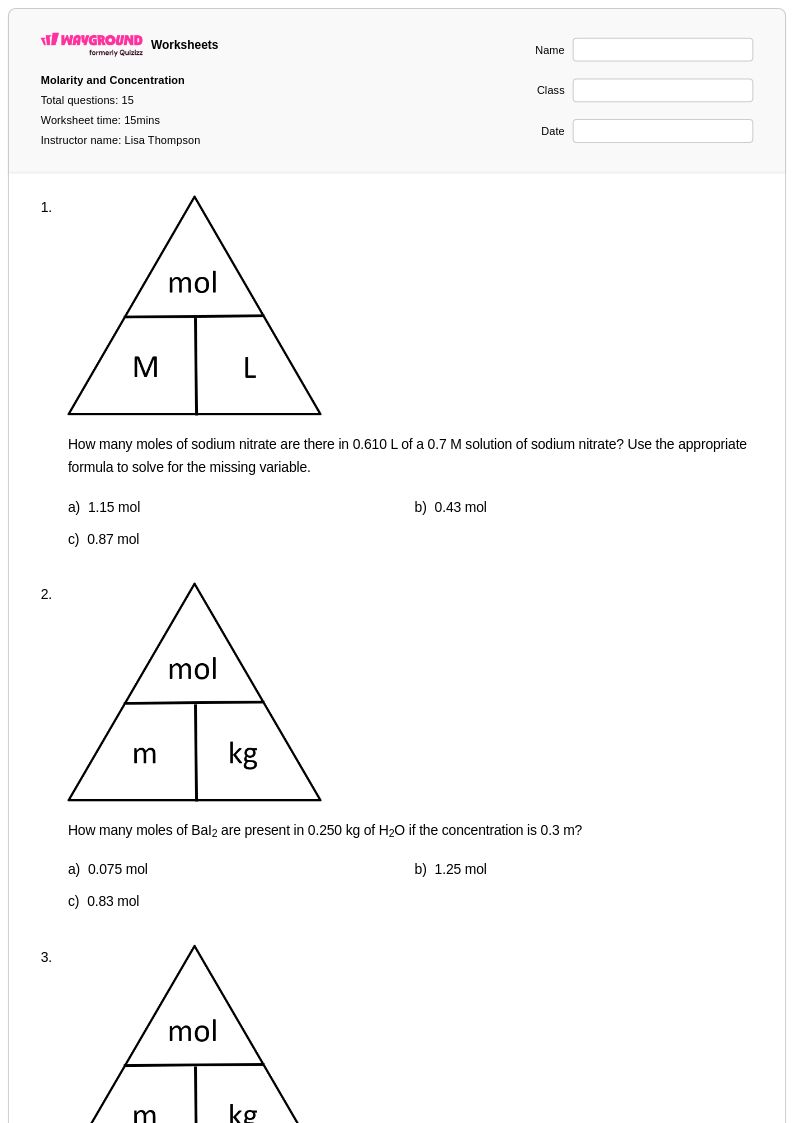
15 Q
10th - Uni

25 Q
10th - Uni

25 Q
10th - Uni
Explore Other Subject Worksheets for class 12
Explore printable Molality worksheets for Class 12
Molality worksheets for Class 12 chemistry students available through Wayground (formerly Quizizz) provide comprehensive practice with this essential concentration measurement concept. These expertly designed worksheets strengthen students' abilities to calculate molality using the moles of solute per kilogram of solvent formula, distinguish between molality and molarity, and apply molality calculations to real-world chemical scenarios including colligative properties and solution preparation. The collection includes diverse practice problems ranging from basic molality calculations to complex multi-step problems involving temperature-dependent solutions, with each worksheet featuring detailed answer keys and step-by-step solutions. Teachers can access these free printables in convenient PDF format, making them ideal for homework assignments, laboratory preparation, assessment review, and independent study sessions that reinforce critical analytical chemistry skills.
Wayground (formerly Quizizz) supports chemistry educators with millions of teacher-created molality worksheet resources that feature robust search and filtering capabilities, allowing instructors to quickly locate materials aligned with specific learning standards and student proficiency levels. The platform's differentiation tools enable teachers to customize worksheets for varying ability levels within Class 12 chemistry classes, while flexible formatting options provide both printable PDF versions for traditional classroom use and digital formats for online learning environments. These comprehensive worksheet collections facilitate effective lesson planning by offering ready-to-use materials for skill practice, targeted remediation for students struggling with concentration calculations, and enrichment opportunities for advanced learners exploring complex solution chemistry applications, ensuring that all students develop mastery of molality concepts essential for success in advanced chemistry coursework.
FAQs
How do I teach molality to chemistry students?
What practice problems help students get better at molality calculations?
What mistakes do students commonly make when calculating molality?
How is molality different from molarity, and why does it matter?
How can I use Wayground's molality worksheets in my chemistry class?
