

1st Quarter Review
Presentation
•
Science
•
8th Grade
•
Medium
+5
Standards-aligned
Sabrina Robinson
Used 37+ times
FREE Resource
39 Slides • 112 Questions
1
Pure Substances and Mixtures

2

3

4

5
Multiple Choice
A combination of rocks and water is an example of
A mixture
A solution
A solute
6
Multiple Choice
7
Multiple Choice

What type of mixture is this?
heterogeneous
homogeneous
8
Multiple Choice

Consider the following samples.
Which model accurately classifies the samples?
9

10
Multiple Choice

Pure Substance
MIxture
11
Multiple Choice

What is it?
Pure Substance
Mixture
12
Multiple Choice

What is it?
Pure Substance
Mixture
13
Multiple Choice

What is it?
Pure Substance
Mixture
14
Multiple Choice

What is it?
Pure Substance
Mixture
15
Multiple Choice

Element
Compound
Homogeneous Mixture
Heterogeneous MIxture
16
Multiple Choice

What is this?
Element
Compound
Heterogeneous MIxture
Homogeneous Mixture
17
Multiple Choice

What is this?
Element
Compound
Homogeneous Mixture
Heterogeneous Mixture
18
Multiple Choice

What is this?
Element
Compound
Homogeneous Mixture
Heterogeneous MIxture
19
Multiple Choice

What type of substance is pictured?
element
compound
mixture
solution
20
Multiple Choice
Which is a mixture of compounds?
21
Multiple Choice

What type of substance is pictured?
element
compound
mixture
solution
22
Multiple Choice

What type of substance is pictured?
element
compound
mixture
23
Multiple Choice
Which is a mixture of compounds?
24
Phase Changes

25
Open Ended

A phase change diagram is used to show the relationship between temperature, pressure and state of matter. Explain what is happening on the horizontal lines and why the lines are horizontal.
26
States of Matter
Remember that the state of matter is a Physical Property because the actual substance doesn't change, only the form changes.

27
Each State of Matter is defined by how their particles behave.
1How close the particles are to one another, and 2how quickly they move determine what state of matter a substance is in.
28
There are 4 states of matter.
- solid
- liquid
- gas
- plasma

29
Multiple Choice

What line segment represents only the solid state? (Diagram F)
A-B
B-C
C-D
D-E
30
Multiple Choice
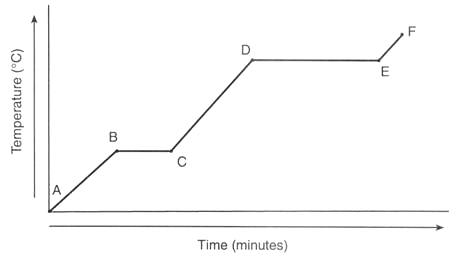
Between which points is the temperature of the substance remaining constant? (Diagram F)
A-B only.
A-B, C-D, E-F
B-C only.
B-C, D-E
31
Multiple Choice
32
Multiple Choice

A substance's heating curve is shown in the graph. What is its boiling point? (Diagram B)
100 C
60 C
80 C
20 C
33
Multiple Choice
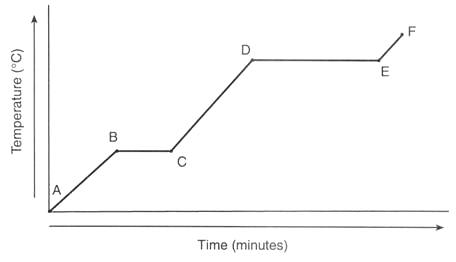
Are the particles moving faster or slower as time goes on? (Diagram F)
Faster
Slower
34
Multiple Choice

Which line segment demonstrates vaporization occurring? (Diagram E)
B-C
C-D
D-E
E-F
35
Multiple Choice

How long does it take this substance to change from a liquid to a gas? (Diagram E)
1 minute
2 minutes
10 minutes
15 minutes
36
Multiple Choice

37
Multiple Choice
38
Solid
In a solid, particles are packed tightly together so they don't move much, but are dense. The electrons of each atom are constantly in motion, giving the atoms a small vibration, but they are fixed in their position.
Solids have a definite shape, as well as mass and volume, and do not conform to the shape of the container in which they are placed.

39
Multiple Choice

What state of matter is represented in this picture?
solid
liquid
gas
40
Liquid
In a liquid, the particles are more loosely packed than in a solid, but closely enough that it is not easily compressible.
The particles are able to flow around each other, giving the liquid an indefinite shape. Therefore, the liquid will conform to the shape of its container.

41
Multiple Choice
Which state of matter do the particles slip and slide past each other?
solid
liquid
gas
42
Gas
In a gas, the particles have a great deal of space between them, so they move easily, and have high kinetic energy (energy of movement). When a gas is put under pressure by reducing the volume of the container, the space between particles is reduced and the gas is compressed.

43
Liquid on the other hand...
In a liquid, the particles are already close together, so it is not easily compressed.

44
Multiple Choice

What state of matter is represented in the picture above?
solid
liquid
gas
45
Multiple Select
Which type(s) of matter have a definite volume?
solid
liquid
gas
46
Multiple Choice
Which state of matter has the ability for its particles to be easily compressed?
Solid
Liquid
Gas
Plasma
47
Gas
A gas has no definite shape or volume. If unconfined, the particles of a gas will spread out indefinitely; if confined, the gas will expand to fill its container.

48
Multiple Select
Which type(s) of matter take on the shape of the container they are in?
solid
liquid
gas
49
Plasma
Plasma is not a common state of matter here on Earth, but it may be the most common state of matter in the universe. Stars are essentially superheated balls of plasma.
Plasma consists of highly charged gas particles with extremely high kinetic energy (energy in motion).
Think of plasma as gas, with an electrical charge.

50
More on plasma.
The noble gases (helium, neon, argon, krypton, xenon and radon) are often used to make glowing signs by using electricity to ionize (an atom with a charge) them to the plasma state.

51
Multiple Choice
Which state of matter is much like a gas, but with particles that are charged?
Solid
Liquid
Gas
Plasma
52

53
Multiple Choice
While an object is melting, what happens to the temperature?
increases
decreases
stays the same
54
Multiple Choice
In which state of matter are particles packed tightly together in fixed positions?
gas
solid
liquid
plasma
55
Multiple Choice
Which has more energy?
Hot water
Room temperature water
Ice water
56
Multiple Choice
Which state of matter contains particles that flow freely past one another?
Solid
Liquid
Gas
Plasma
57
Multiple Choice
In which state of matter do particles spread apart and fill all the space available to them?
crystal
liquid
gas
solid
58
Multiple Choice
Which state of matter has a fixed volume and mass?
Solid
Liquid
Gas
Plasma
59
Multiple Choice
In a(n) _______________________, the particles are packed closely together, but they can move past each other freely.
solid
liquid
gas
plasma
60
Multiple Choice
Which state of matter fills a container?
Solid
Liquid
Gas
Plasma
61
Multiple Choice

In which state of matter are the particles least able to move?
A
B
C
62
Multiple Choice
Which state of matter contains particles that slowly vibrate, but in a fixed position?
Solid
Liquid
Gas
Plasma
63
Multiple Choice
Particles in this state of matter are moving the fastest.
solid
liquid
gas
64
Multiple Choice
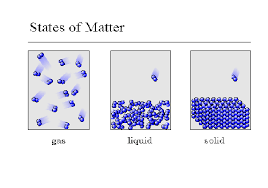
At higher temperatures
particles in an object have less energy
particles in an object move faster
a gas contracts
65
Multiple Choice
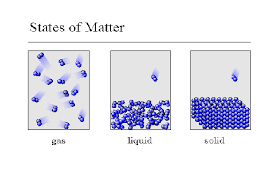
A gas
has a definite shape but no definite volume
has a definite volume but no definite shape
has fast-moving particles
66
Multiple Choice
Which state of matter has a definite shape?
Solid
Liquid
Gas
Plasma
67
Multiple Choice
Superheating gas so that it becomes ionized is one of the methods to form this state of matter.
Solid
Liquid
Gas
Plasma
68
Multiple Choice
Atoms or molecules in this phase of matter have the highest energy.
Solid
Liquid
Gas
Plasma
69
Multiple Choice
When water sits in the freezer and freezes into ice, it is because the water has...
gained mass
gained energy
lost mass
lost energy
70
Multiple Choice
When ice sits out on a counter and melts, it is because the ice has...
gained mass
gained energy
lost mass
lost energy
71
Physical and Chemical Properties
Properties of Matter

72
All substances have properties that we can use to identify them. There are TWO basic types of properties:
PHYSICAL properties and CHEMICAL properties
73
Does not change the identity or composition of matter
Observed with senses or measurements
PHASE CHANGES are physical, since only the speed, spacing, and attraction of particles are altered.
Physical Properties

74
Examples of Physical Properties
magnetism
boiling point and freezing point
density, mass, and volume
viscosity (how thick a liquid is)
size, shape, and color
texture
conducts electricity
75
Describes how the substances may change when it transforms through a chemical reaction (reacts with another substance)
Changes the identity of the original substance
Chemical Properties

Chemical changes can't be "undone", since the substance has changed into something new!
76
Examples of Chemical Properties
reactivity (reacting with other chemicals)
Signs of a chemical reaction to look for:
Release of heat or light
Color change
Formation of a gas
Formation of a precipitate (solid falling out of a liquid)
pH (measuring how acidic or basic a solution is)
flammability (how likely to burn)
77

78
Multiple Choice

You measure the DENSITY of a rock compared to water.
physical property
chemical property
79
Multiple Choice

A candle burning because the wick is flammable.
physical property
chemical property
80
Multiple Choice
Which of the following is NOT a physical property?
flammability
melting point
density
luster
81
Multiple Choice
A property that can only be observed when a substance reacts or does not react with another substance
chemical property
physical property
82
Multiple Choice
Which of the following is defined as the likelihood of a substance to undergo a chemical reaction?
oxidation
rusting
reactivity
flammability
83
Multiple Choice
Which of the following is NOT an example of a chemical reaction?
flammability
oxidation
reactivity
melting point
84

The different densities of objects determines if it will sink or float in water.
Higher density than water will SINK!
Lower density than water will FLOAT!
The density of water is defined as 1 gram / milliliter3
What makes objects sink or float?
85
Multiple Choice
If water has a density of 1 g/cm3, what would an object with a density of 1.5 g/cm3 do in water?
sink
float
I don't know
86
Multiple Choice

Which box has a higher density?
A
B
They are the same
87
Multiple Choice

88
Multiple Choice
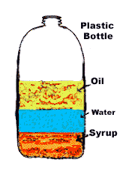
89
Multiple Choice

In the picture, a can of Pepsi sunk and a can of Diet Pepsi are put into water. The Pepsi must be
more dense than water
less dense than water
it has no density
it has a density of 1 g/mL
90
Multiple Choice
Why does ice float
Liquid water is less dense than solid water.
Solid water is less dense than liquid water.
Gaseous water is less dense than solid water.
Liquid water is less dense than gaseous water.
91
Multiple Choice

Which of the following substances would float in water? (Hint: water has a density of 1g/cm3)
gold
rubber
water
cork
92

93

94
Multiple Choice
What is an example of a physical change?
baking brownies
explosion
cutting paper
rust on metal
95

96

97
Multiple Choice
Is baking a cake a physical or chemical change?
Physical
Chemical
98

99

100

101

102

103
Multiple Choice
Chopping wood
physical change
chemical change
104
Multiple Choice
Mixing different colored marbles
physical change
chemical change
105
Multiple Choice
Firework
physical change
chemical change
106
Multiple Choice
Making eggs for breakfast
physical change
chemical change
107
Multiple Choice
Burning wood
physical change
chemical change
108
Multiple Choice
109
Multiple Choice
110
Multiple Choice
111
Multiple Choice
112
Multiple Choice
113
Multiple Choice
changes affecting the form of a chemical substance, but not its chemical identity.
114
Multiple Choice
What is a chemical change?
How two chemicals react with one another when combined
A property that we can measure or observe with our 5 senses or other tools
When two chemicals react with one another to form a new substance
When a change occurs but it does not result in the formation of a new substance
115
Multiple Choice
A student mixes yellow powder with water. Which observation would prove a CHEMICAL change occurred?
The powder DISSOLVED in the water
The water turned the COLOR yellow
The water's TEMPERATURE changed
The STATE OF MATTER changed
116
Multiple Choice
Which of the following options provides evidence of a CHEMICAL change?
Wood is burned releasing ash and smoke
Rocks separate when soil is sifted
Boiling water bubbles when heated
Salt dissolves in water
117
Multiple Choice
Which of the following examples provides evidence of a PHYSICAL change?
Wood is burned and releases ash and smoke
Iron rusts over time
When mixed, baking soda and vinegar form gas bubbles
Boiling water bubbles when heated
118
Multiple Choice
When mixing milk and vinegar, a white, chunky, solid substance is formed. What has occurred?
Physical change: state of matter
Chemical change: precipitate
Physical change: freezing
Chemical change: new gas
119
Multiple Choice
You mix a yellow powder and a clear liquid. The liquid turns light yellow and the powder seems to disappear. This is a...
Endothermic Chemical Change
Exothermic Chemical Change
Physical Change
120

121

122

123
Multiple Choice
What is a group
Up and down row on the table
Up and down column on the table
Side to side row on table
Side to side column on the table
124
Multiple Choice
What is a period
Up and down row on the table
Up and down column on the table
Side to side row on table
Side to side column on the table
125
Multiple Choice
What group is carbon in?
1
2
13
14
15
126
Multiple Choice
Why are noble gases not reactive
Too few electrons
Too many electrons
They have a full set of electrons
They need more electrons
127
Multiple Choice

128
Multiple Choice

129
Multiple Choice
130
Multiple Choice

131
Multiple Choice
The Atomic number is known as the _______
Number of electrons in an atom
Number of Protons in an atom
Number of neutrons in an atom
Number of Protons and electrons in an atom
132
Multiple Choice
What charge do protons have?
Positive
Negative
Neutral
133
Multiple Choice
What is Atomic Mass Number ?
Number of Protons
Number of Neutrons
Number of Protons and Neutrons
Number of Protons and Electrons
134
Multiple Choice
The number of electrons in an atom is equal to the...
Atomic number
Protons
Atomic Mass
Neutrons
135
Multiple Choice
The formula used to calculate the number of neutrons is:
Mass Number - Atomic Number
Protons + Electrons
Atomic Number - Mass Number
Protons x2
136
Multiple Choice
How many periods are on the periodic table?
5
6
7
8
137
Multiple Select
Elements in the same group have : (2 correct answers)
Similar chemical properties
Similar names
Same number of outer (valence) electrons)
The same number of protons
138
Atoms
Atoms are made of protons, electrons, and neutrons
Protons are positive
Neutrons are neutral
Electrons are negative
Protons and neutrons are in the nucleus
Electrons are on the outside
139
Multiple Choice
140
Multiple Choice

141
Multiple Choice
How many atoms of Hydrogen are in H2O
1
2
3
more than 3
142
Multiple Choice
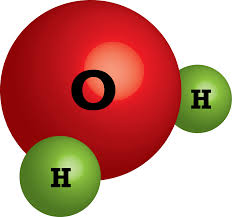
How many atoms of Hydrogen are in this compound?
1
2
3
4
143

144
Multiple Choice
Substances present at the start of a chemical reaction.
subscripts
reactants
coefficients
products
145
Multiple Choice
How many atoms of Carbon (C) are in C6H12O6?
3
6
12
24
146
Multiple Choice
In chemical reactions, what does the principle of conservation of mass mean?
Matter is not created or destroyed
The total mass of the reactants is greater than the total mass of the products
The total mass of the reactants is less than the total mass of the products
Matter is not changed
147
Multiple Choice
Is this equation balanced?
N2 + H2 --> NH3
Yes
No
148
Multiple Choice
149
Multiple Choice
The Law of Conservation of Mass says that matter can neither be __________ or __________.
created; destroyed
recycled; renewed
subtracted; added
joined; separated
150
Multiple Choice
In a reaction A + B ----> C, reactant A has 5g and product C has 9g. How many grams does reactant B have?
4g
5g
9g
14g
151
Multiple Choice
Pure Substances and Mixtures

Show answer
Auto Play
Slide 1 / 151
SLIDE
Similar Resources on Wayground

148 questions
tata surya
Presentation
•
7th Grade

145 questions
Energy Transformation Lesson Plus Questions
Presentation
•
6th - 8th Grade

139 questions
7.PS1.2-Mixtures, Reactions, and Changes in Matter-notes/slides
Presentation
•
7th Grade

145 questions
học môn địa lý
Presentation
•
8th Grade

142 questions
Benefits of Biodiversity
Presentation
•
6th Grade

150 questions
E7- UNIT 5- Grammar- count,uncount,How much, How many, some,any
Presentation
•
7th Grade

145 questions
Benchmark Review
Presentation
•
6th - 10th Grade

143 questions
Law & Justice- Crime
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade

48 questions
8th Grade NC Science EOG Review 2
Quiz
•
8th Grade

19 questions
Renewable and Nonrenewable Resources (CK)
Quiz
•
8th Grade

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade

10 questions
Exploring Weather Influences and Map Reading
Interactive video
•
6th - 10th Grade

43 questions
Amplify Earth's Changing Climate Unit Review
Quiz
•
6th - 8th Grade