

SCIENCE YEAR 9 REVIEW PACKET
Presentation
•
Science
•
8th Grade
•
Practice Problem
•
Medium
+18
Standards-aligned
Gil legaspi
Used 2+ times
FREE Resource
101 Slides • 67 Questions
1
SCIENCE YEAR 9 REVIEW PACKET
HCIS

2
Keys, Variations, Inheritance
UNIT 3

3

4

5

6

7

8

9
Multiple Choice
Where do offspring get their genes?
Half from their mom, half from their dad
From their mom
From their dad
From their brains
10
Multiple Choice
Where are genes found?
In the mitochondria
In our bone marrow
Chromosomes, in the nucleus of a cell
In our brains
11
Multiple Choice
What is variation?
Racial diversity
The variation of females and males
Variation of personalities
The differences between the individuals in a species
12
Multiple Choice
What causes variation?
Your environment
Your genes and your environment
Your genes
Your names
13
Multiple Choice
What is a key (genetics)?
Something to open a lock
Something that tells us which object something is
A method of identifying a unknown organism
The key to life
14
Selective Breeding and Natural Selection

15

16

17

18

19

20
Multiple Choice

The long and sharp beak of a woodpecker is an example of
natural selection
selective breeding
21
Multiple Choice
Which organisms are most likely to survive?
The strongest
The best adapted
The fittest
The most athletic
22
Multiple Choice

Which is an example of selective breeding?
A giraffe's long neck.
Clipping a horse's tail so it's not too long.
Training a horse to run faster.
Cross breeding a labrador retriever with a poodle .
23
Multiple Choice
"Survival of the fittest" describes which theory
Natural selection
Selective breeding
24
Multiple Choice
Selective breeding
organisms better adapted to their environment tend to survive and produce more offspring
process used by humans to develop new organisms with desirable characteristics
25
Multiple Choice
Natural selection
organisms better adapted to their environment tend to survive and produce more offspring
process used by humans to develop new organisms with desirable characteristics
26
Multiple Choice

Change over time
Natural Selection
Evolution
Selective Breeding
Homeostasis
27
Material Properties
UNIT 4

28
The Structure of Atoms

29

30

31

32

33

34
Multiple Choice

35
Multiple Choice

36
Multiple Choice

37
Multiple Choice

38
Multiple Choice

39
Multiple Choice

40
Periodic Table Trends

41

42

43

44

45

46

47

48
Multiple Choice
49
Multiple Choice
50
Multiple Choice
51
Multiple Choice
52
Multiple Choice
53
Multiple Choice
54
Multiple Choice
55
Multiple Choice
56
Multiple Choice
57
Multiple Choice
58
Energy Changes
UNIT 5

59

60

61

62

63

64

65

66
Multiple Choice
What type of chemical reaction absorbs energy and requires energy for the reaction to occur?
Endothermic
Exotermic
Exothermic
Endotermic
67
Multiple Select
Examples of endothermic reactions
Photosynthesis
Ice melting
Respiration
Sodium carbonate + ethanoic acid
Water + calcium oxide
68
Multiple Choice
if less heat energy is released in making bonds in the products than is taken in when breaking bonds in the reactants
Endothermic
Exotermic
Exothermic
Entrothermic
69
Multiple Choice
if more heat energy is released in making bonds in the products than is taken in when breaking bonds in the reactants
Exothermic
Endothermic
Exotermic
Endotermic
70
Multiple Choice
When calcium reacts with water, the temperature changes from 18°C to 39°C. Which statement is correct?
The solution at the end is acidic
The reaction is reversible
The reaction is exothermic
The reaction is endothermic
71
Multiple Choice
ΔH value in an endothermic reaction is a positive number.
True
False
72
Reactivity
UNIT 6

73

74

75

76

77

78

79

80
Multiple Choice
81
Multiple Choice
82
Multiple Choice
83
Multiple Choice
84
Multiple Choice
85
Multiple Choice

86
Multiple Choice
87
Multiple Choice
Which of the following the most reactive in dilute acid
magnesium
calcium
aluminum
zinc
88
Salts
Unit 7

89

90

91

92

93

94

95

96

97

98

99

100

101

102

103

104

105

106

107

108

109

110
Multiple Choice
Bryan wants to prepare potassium sulphate by reacting an acid with potassium hydroxide. Name the acid used in this experiment.
hydrochloric acid
nitric acid
sulphuric acid
111
Multiple Choice
Bryan wants to prepare silver chloride in the science laboratory. Which of the following reactants should he use?
silver + hydrochloric acid
silver hydroxide + hydrochloric acid
silver nitrate + potassium chloride
silver + chlorine gas
112
Multiple Choice
Precipitation involves mixing ___________ to form an insoluble solid that separates out from the reaction mixture.
acid and alkali
acid and metal
two acids
two solutions
113
Multiple Choice
114
Multiple Choice
115
Multiple Choice
116
Multiple Choice
What type of reaction produced an insoluble salt?
Neutralisation reaction
Precipitation reaction
Titration reaction
Insoluble reaction
117
Multiple Choice
How do you remove the water to leave solid copper sulfate crystals?
Filter
Evaporate
Distill
118
Multiple Choice
Complete the following word equation:
Metal + Acid -->
Salt + water
Salt + hydrogen
Salt + Water + Carbon dioxide
119
Rates of Reaction
UNIT 8

120

121

122

123

124

125

126

127

128

129

130

131

132

133

134

135

136

137

138

139

140

141

142

143

144

145
Multiple Select
Which factors affect the rate of a reaction?
temperature
concentration
surface area
pressure
catalyst
146
Multiple Choice
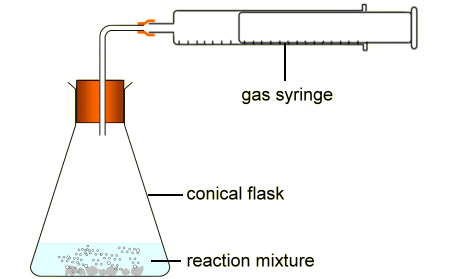
How is this equipment being used to measure the rate of reaction?
The gas syringe measures how much gas is produced in a certain time
The reaction mixture will increase in volume in a certain time
147
Multiple Choice

The amount of something; how many particles in a volume of a mixture
Rate
Collision
Catalyst
Concentration
148
Multiple Choice
The speed at which a chemical reaction occurs
Fast
Slow
Rate
Ratio
149
Multiple Choice
150
Multiple Choice

Icing sugar has a greater ______ _____ than a solid cube of
sugar.
Surface Area
Catalyst
Temperature
151
Multiple Choice
How can you increase the rate of a reaction?
Decrease the pressure
Decrease the concentration
Increase the concentration
152
Multiple Choice
What is the rate of reaction?
How fast a reaction is
How big a reaction is
How loud a reaction is
153
Multiple Choice
Which factors increase the rate of a reaction?
increasing temperature
increasing concentration
increasing surface area
all of these
154
Forces in Action
UNIT 9

155

156

157

158

159

160

161

162
Multiple Choice

#16 Show all your work on your answer document. m=D⋅V
0.625 g
1.6 g
10 g
7 g
163
Multiple Choice

#15 Show all your work on your answer document. m=D⋅V
0.25 g
3 g
4 g
5 g
164
Multiple Choice
165
Multiple Choice
166
Multiple Choice
167
Multiple Choice

168
Multiple Choice
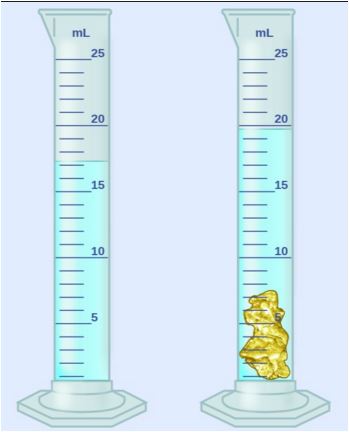
An irregularly shaped piece of gold was lowered into a graduated cylinder holding a volume of water equal to 17 ml. The height of the water rose to 20 ml. If the mass of the gold was 27 g, what was its density?
9 g/mL
10.5 g/mL
6.5 g/mL
8 g/mL
SCIENCE YEAR 9 REVIEW PACKET
HCIS

Show answer
Auto Play
Slide 1 / 168
SLIDE
Similar Resources on Wayground

165 questions
IB Psychology Qualitative Research
Presentation
•
11th - 12th Grade

161 questions
SP - S8 2026
Presentation
•
KG
![[tutoring] root words and affixes](https://cf.quizizz.com/image/image-loader.svg)
159 questions
[tutoring] root words and affixes
Presentation
•
6th - 8th Grade

167 questions
Integrated Science Final Reveiw
Presentation
•
9th - 12th Grade

151 questions
DiHybrid Crosses
Presentation
•
9th Grade

149 questions
Math 8 Module 15
Presentation
•
8th Grade

155 questions
Medical Term - Digestive System
Presentation
•
10th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade

48 questions
8th Grade NC Science EOG Review 2
Quiz
•
8th Grade

19 questions
Renewable and Nonrenewable Resources (CK)
Quiz
•
8th Grade

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade

10 questions
Exploring Weather Influences and Map Reading
Interactive video
•
6th - 10th Grade

43 questions
Amplify Earth's Changing Climate Unit Review
Quiz
•
6th - 8th Grade