

PC Review Ch.1 + 2
Presentation
•
Science
•
7th Grade
•
Easy
+2
Standards-aligned
Denisse Duran
Used 8+ times
FREE Resource
16 Slides • 48 Questions
1
PC Review Ch.1 + 2

2

3
Multiple Choice
When energy is transferred into a substance...
kinetic energy, temperature, and speed changes
Only temperature changes
4
Multiple Choice
The colder a substance is, the
less freedom of movement it has
more freedom of movement it has
5
Multiple Choice
The hotter a substance is, the
less freedom of movement it has
more freedom of movement it has
6

7

8

9

10

11

12

Yellow arrow= energy transferred in
Green arrow= energy transferred out
13
Multiple Choice

Phase change going from a Solid to a Liquid...
Freezing
Condensation
Melting
Sublimation
14
Multiple Choice
Phase change going from Liquid to Solid...
Melting
Freezing
Condensation
Sublimation
15
Multiple Choice
16
Multiple Choice
17
Multiple Choice
18
Multiple Choice
19
Multiple Choice
20

21
Multiple Choice

A student left a sealed jar of water in her freezer. When she returned twelve hours later, the water had changed phase and was a solid. What happened to the water molecules?
Before, the molecules were moving in place. After, the molecules were moving around each other.
Before, the molecules were moving around each other. After, the molecules were moving in place.
Before, the molecules were moving away from each other. After, the molecules were moving around each other.
22
Multiple Choice
23

24

25

26
Multiple Choice
Blake is doing a science experiment with a substance in a sealed jar. At first, the substance’s molecules are moving around each other. What will happen to Blake’s substance if he transfers energy out of the substance and causes a phase change? After the phase change, the substance’s molecules will move . ..
faster and be a gas
faster and be a liquid
slower and be a liquid
slower and be a solid
27
Multiple Choice
Cohen had a Popsicle but left it out and it started to melt. What caused the Popsicle to melt?
kinetic energy was taken away from the Popsicle
kinetic energy was added to the Popsicle
Cohen doesn't deserve treats if he is going to waste them
28
Multiple Choice
Lily is doing a science experiment with a substance in a sealed jar. At first, the substance’s molecules are moving around each other. What will happen to Lily’s substance if she transfers energy into the substance and causes a phase change?
After the phase change, the substance’s molecules will move . . .
faster, and the substance will be a liquid.
faster, and the substance will be a gas.
slower, and the substance will be a solid.
slower, and the substance will be a liquid.
29
Multiple Choice
Scientists digging in a cave found an unknown substance. The scientists found that the substance’s molecules were moving around each other. The scientists increased the speed of the substance’s molecules and caused a phase change. How did the scientists do this, and how did this affect the substance?
The scientists transferred energy . . .
into the substance, and the substance changed into a gas.
into the substance, and the substance changed into a liquid.
out of the substance, and the substance changed into a solid.
out of the substance, and the substance changed into a liquid.
30
Multiple Choice
31
Multiple Choice
The "measure of the average kinetic energy of the molecules of a substance" is also known as:
temperature
heat
potential energy
speed
32
Multiple Choice
A "measure of how hot or cold something is" is called:
temperature
heat
kinetic energy
potential energy
33
Multiple Choice
A DECREASE in molecules' motion is evidence that:
their kinetic energy has DECREASED
their kinetic energy has INCREASED
their kinetic energy has STAYED THE SAME
34
Multiple Choice
An INCREASE in molecules' motion is evidence that:
their kinetic energy has INCREASED.
their kinetic energy has DECREASED.
their kinetic energy has STAYED THE SAME.
35
Multiple Choice
When energy is REMOVED from a substance, it leads to:
a DECREASE in the kinetic energy of the molecules that make up that substance
an INCREASE in the kinetic energy of the molecules that make up that substance
NO CHANGE in the kinetic energy of the molecules that make up that substance
BOTH a simultaneous INCREASE and DECREASE in the kinetic energy of the molecules that make up that substance
36
Multiple Choice
When energy is ADDED to a substance, it leads to:
an INCREASE in the kinetic energy of the molecules that make up that substance
a DECREASE in the kinetic energy of the molecules that make up that substance
NO CHANGE in the kinetic energy of the molecules that make up that substance
BOTH a simultaneous INCREASE and DECREASE in the kinetic energy of the molecules that make up that substance
37

38
Multiple Choice
If you want to CONDENSE a gas, you have to:
transfer energy OUT of it
transfer energy into it
keep the energy the same
transfer energy both INTO and OUT of it at the same time
39
Multiple Choice
If you want to FREEZE a solid, you have to:
transfer energy OUT of it
transfer energy INTO it
keep the energy the same
transfer energy both INTO and OUT of it at the same time
40
Multiple Choice
If you want to EVAPORATE a liquid, you have to:
transfer energy INTO it
transfer energy OUT of it
keep the energy the same
transfer energy both INTO and OUT of it at the same time
41
Multiple Choice
If you want to MELT a solid, you have to:
transfer energy INTO it
transfer energy OUT of it
keep the energy the same
transfer energy both INTO and OUT of it at the same time
42
Multiple Choice
If energy is transferred OUT OF a substance, the substance will:
gets warmer/hotter
gets cooler/colder
stay the same temperature
43
Multiple Choice
If energy is transferred INTO a substance, the substance will:
get warmer/hotter
get cooler/colder
stay the same
44

45

46
Multiple Choice
When a substance gets hotter or warmer, its freedom of movement:
increases
decreases
stays the same
47
Multiple Choice
When a substance heats up, its freedom of movement:
increases
decreases
stays the same
48
Multiple Choice
When a substance gets colder, its freedom of movement:
increases
decreases
stays the same
49
Multiple Choice
When a substance cools down, its freedom of movement:
decreases
increases
stays the same
50
Multiple Choice
Which of the following causes a substance's freedom of movement to DECREASE?
condensing
evaporating
melting
energizing
51
Multiple Choice
Which of the following causes a substance's freedom of movement to DECREASE?
freezing
melting
evaporating
stagnating
52
Multiple Choice
Which of the following causes a substance's freedom of movement to INCREASE?
freezing
evaporating
condensing
regulating
53
Multiple Choice
Which of the following causes a substance's freedom of movement to INCREASE?
freezing
melting
condensing
activating
54
Multiple Choice
If you transfer energy out of a substance how will the movement of the molecules change?
molecules will move faster
molecules will move slower
molecules will stop moving
molecules do not move
55
Multiple Choice
If you transfer energy into a substance how will the molecules change?
molecules will move faster
molecules will move slower
molecules will stop moving
56
Multiple Choice
How do the molecules in a liquid behave
do not move
move away from each other
move in place
move around each other
57
Multiple Choice
How do the molecules in a gas behave?
move in place
move around each other
move away from each other
do not move
58
Multiple Choice
How do the molecules in a solid behave?
Move around each other
move in place
Moving away from each other
Do not move
59
Multiple Choice
What happens to molecules when their kinetic energy decreases?
They move slower.
They move faster.
They get heavier.
It depends on the type of molecules.
60
Multiple Choice

The diagram above describes the movement of molecules in a substance used to make fuel. What change did the fuel scientist observe in the substance?
Before the process, the substance was a solid. After the process, it was a liquid.
Before the process, the substance was a gas. After the process, it was a liquid.
Before the process, the substance was a liquid. After the process, it was a solid.
Before the process, the substance was a liquid. After the process, it was a gas.
Before the process, the substance was a liquid. After the process, it was a gas.
61
Multiple Choice

The medicine in the diagram above has molecules that are moving around each other. The doctor needs to slow down the molecules enough to cause a phase change. How will she do this, and how will this affect the medicine?
She transfers energy . . .
into the medicine until it is a liquid.
into the medicine until it is a gas.
out of the medicine until it is a solid.
out of the medicine until it is a liquid.
62
Multiple Choice

A measure of how hot or cold something is
Phase
Molecule
Density
Temperature
63
Multiple Choice

The energy that an object has because it is moving
Molecule
Refute
Density
Kinetic Energy
64
Multiple Choice
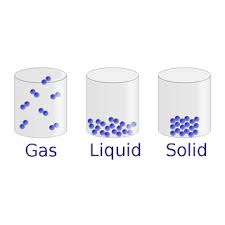
The way molecules in a substance move around relative to each other
Kinetic Energy
Molecule
Phase
Freedom of Movement
PC Review Ch.1 + 2

Show answer
Auto Play
Slide 1 / 64
SLIDE
Similar Resources on Wayground

55 questions
Upwellings and Currents
Presentation
•
8th Grade

56 questions
Unit 5 Adaptations
Presentation
•
7th Grade

59 questions
Energy
Presentation
•
8th Grade

60 questions
Using Food Pyramids to Describe Energy Transfer
Presentation
•
7th Grade

59 questions
Science Lesson Vocab 2
Presentation
•
7th Grade

60 questions
Criterion B and C T&C M1 Q4
Presentation
•
7th Grade

60 questions
Multiplying Integers
Presentation
•
7th Grade

56 questions
Body Systems
Presentation
•
7th - 8th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Science

19 questions
Introduction to Properties of Waves
Presentation
•
6th - 8th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

45 questions
Food Webs
Quiz
•
6th - 8th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

20 questions
Abiotic & Biotic Factors
Quiz
•
7th Grade

20 questions
Asexual/Sexual Reproduction
Quiz
•
7th Grade

20 questions
Energy Transformations
Quiz
•
6th - 8th Grade

20 questions
Food Webs + Energy Pyramids
Quiz
•
7th Grade