

Self paced: Chemical reactions Part 1
Presentation
•
Science
•
7th - 9th Grade
•
Easy
+2
Standards-aligned
STEPHANIE SLUSS
Used 8+ times
FREE Resource
13 Slides • 28 Questions
1
Chemical Equations
Today you will:
Review chemical changes
Learn parts of a chemical equation
Learn how to apply LOCOM to chemical equations
Some text here about the topic of discussion
2
The next slide is a reaction- it is a twist on one we did in class...
Watch the video and see if you can figure out what kind of change is taking place.
3
4
Open Ended
What kind of a change was that and why?
5
How can we tell when a chemical reaction has occured?
Remember these?
color change change in odor
Production of a gas ( bubbles) formation of a precipitate
Energy Production
Write these on your paper.....

6
Draw
Circle the signs you saw in the alka seltzer reaction?
7

8
Multiple Choice
A real world example of color change is
Train rusting
Patina on the statue of liberty
apple turning brown
all of these are real world examples
9

10

11
Draw
What are the bubbles? Circle these on the slide
12
Multiple Choice
We know that there was a chemical reaction occurring when 2 clear liquids were mixed together. That is because we show the production of a _____________.
solid.
precipitate.
gas.
13
Multiple Select
You know that a chemical reaction is occurring here because you see __________________ . (Pick 3)
purple color
smoke
ice formation
fire
a yellow precipitate
14
Models are important in Science
How do scientists model atoms?
Some text here about the topic of discussion
15
Draw
Draw a model of Lithium. It has 3 protons, 4 neutrons and 3 electrons
16
Chemical Equation: models chemical reactons and shows how the atoms are rearranged.

17
Draw
This equation models what happens when Hydrogen and oxygen come together to make water. Circle water.
18
Multiple Choice
How do scientist model chemical reactions?
chemical equations
Bohr models
They dress the atosm up adn put them on a runway
music and lots of clay
19


20
Draw
Circle the reactants. what you start with
21
Draw
Circle the products. What you end with
22
Multiple Choice

What are the reactants in this reaction?
H2 and O2
H2
O2
H2O
23
Multiple Choice

The reactants in the image above are:
CH4 and 2 O2
2 H2O and CO2
All of these are reactants
All of these are products
24
Multiple Choice
What is the left part of a chemical equation called?
2H2 + O2 ---> 2H2O
Reactants
Yields
Products
Chemical equation
25
Multiple Choice
What is the right part of a chemical equation called?
2H2 + O2 ---> 2H2O
Reactants
Yields
Products
Chemical Equation
26
Multiple Choice
Complete the sentence.
During a chemical reaction, atoms ________.
can appear
can disappear
get rearranged
stop moving and vibrating
27
Multiple Choice
28
Law of Conservation of Matter
Matter can be transformed but it is never created or destroyed.
glow party memories...

29
What does this have to do with equations?
If you start with 100 grams of reactants you will end with 100 grams of products.
Some text here about the topic of discussion
30
Multiple Choice
31
Multiple Choice
32
Multiple Choice
The Law of Conservation of Mass States
Energy cannot be created nor destroyed, it can only change form.
Mass cannot be created nor destroyed, it can only change form.
Mass can be created or destroyed, it cannot change form.
33
Multiple Choice
According to the Law of Conservation of Mass, what happens to the wood and oxygen when it is burned?
The wood disappears.
It stays the same chemically.
The atoms in the wood and oxygen rearrange and form smoke and ash.
Nothing.
34
Multiple Choice
35
Multiple Choice
Hint: Read this twice.....
In a reaction A + B ----> C, reactant A has 5g and product C has 9g. How many grams does reactant B should have?
36
Multiple Choice
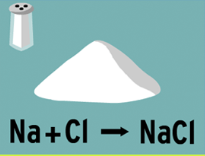
When sodium and chlorine combine to form sodium chloride, sodium chloride is the:
Originator
Reactant
Product
Valence wielder
37
Multiple Choice
38
Multiple Choice

39
Poll

How do you feel about labeling a chemical equation?
I am a labelling expert. People will come from everywhere to get my help
I get it but would like more practice
I sort of get it but need more help to really understand
I have no idea how to do any of this... What is an equation
40
Poll

How do you feel about applying the Law of conservation of matter to equations
i totally get it. Ask me anything
I understood some problems but not all of them
I am pretty confused, s....o....s....s...o....s....
41
1. Now, fill out the key words on your notes...
2. Then, label the eqution and answer the "today I learned" portion...
Some text here about the topic of discussion
Chemical Equations
Today you will:
Review chemical changes
Learn parts of a chemical equation
Learn how to apply LOCOM to chemical equations
Some text here about the topic of discussion
Show answer
Auto Play
Slide 1 / 41
SLIDE
Similar Resources on Wayground

35 questions
synch: lunar cycle
Presentation
•
7th - 8th Grade

34 questions
7U3L4 Energy Pyramids
Presentation
•
7th - 8th Grade

34 questions
Acids and bases
Presentation
•
7th - 8th Grade

35 questions
Scale of the Universe and Characteristics of Stars
Presentation
•
7th - 8th Grade

38 questions
Electricity Lesson
Presentation
•
6th - 8th Grade

38 questions
Force and Motion
Presentation
•
8th Grade

32 questions
Metric System
Presentation
•
7th - 8th Grade

36 questions
Natural Selection Info
Presentation
•
7th - 8th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

20 questions
genetics, punnett squares, heredity
Quiz
•
7th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

30 questions
GMAS Physical Science Review
Quiz
•
8th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

20 questions
Food Webs + Energy Pyramids
Quiz
•
7th Grade

10 questions
Exploring the Rock Cycle
Interactive video
•
6th - 8th Grade