

Heating Curves
Presentation
•
Science
•
9th - 12th Grade
•
Medium
Standards-aligned
Stacy Kowlsen
Used 19+ times
FREE Resource
11 Slides • 9 Questions
1

Copyright © 2011 MsRazz ChemClass
Bell Ringer
Complete the handout
while watching the video.
Use the word bank to help
with labeling the heating
curve
Word Bank:
Solid, Liquid,
Gas, Freezing,
Melting,
Vaporization,
and
condensation.
2

Copyright © 2011 MsRazz ChemClass
Video
• https://www.bing.com/videos/search?q=tyler+de
witt+phase+changes&&view=detail&mid=C6B29
A29C817C2B5C584C6B29A29C817C2B5C584&&F
ORM=VRDGAR&ru=%2Fvideos%2Fsearch%3Fq%3D
tyler%2520dewitt%2520phase%2520changes%26q
s%3DMM%26form%3DQBVR%26sp%3D1%26pq%3D
tyler%2520dewitt%2520phases%26sc%3D1-
19%26cvid%3DAD19A0212B3F4AB4AA40C6EB2446
4693
3

Copyright © 2011 MsRazz ChemClass
Essential
Question
Using your picture of the heating curve,
answer what happens to the
temperature of the material when it is
undergoing a phase change?
4

Copyright © 2011 MsRazz ChemClass
Standard
SC.912.P.10.7
DISTINGUISH BETWEEN ENDOTHERMIC AND EXOTHERMIC
CHEMICAL PROCESSES.
5

Copyright © 2011 MsRazz ChemClass
After today, you should be able to…
Explain what thermochemistry is
List different types of energy
Explain what happens to particle
motion when heat energy is added
Convert between units of calories
(cal), Joules (J) , and kilo-Joules (kJ)
6

Copyright © 2011 MsRazz ChemClass
After today you should be able to…
• Explain what occurs on each
point of a heating curve
•Explain the phase changes as
endothermic or exothermic
7
Copyright © 2011 MsRazz ChemClass
Heating
Curves
When a solid substance is
heated (A), its temperature
will increase until it reaches
its melting point (m.p.).
• Temperature will then stay
constant during the melting
process. (C = liquid) (phase
change=constant
temperature)
8

Copyright © 2011 MsRazz ChemClass
Heating Curves
A heating curve is a graph of
temperature versus time.
•It describes the enthalpy
changes that take place
during phase changes.
9

Copyright © 2011 MsRazz ChemClass
10

Copyright © 2011 MsRazz ChemClass
Endothermic and
Exothermic & Phase
Changes
•Sublimation, when a solid
transforms into a gas, is
an endothermic phrase change,
•Deposition, when a gas transforms
to a solid, is an exothermic phase
change.
•There are a total of
six phase changes in matter, and
three are endothermic and three
are exothermic
11

Copyright © 2011 MsRazz ChemClass
Heating
Curves
When a substance is
completely melted, its
temperature will again
increase until it has
reached its boiling point
(b.p.).
• Temperature stays
constant during boiling.
• Once completely
vaporized, the
temperature will again
increase. (E=gas)
12
Multiple Choice

Which letter indicates where water is in both the solid and liquid phase at the same time?
A
B
C
D
E
13
Multiple Choice
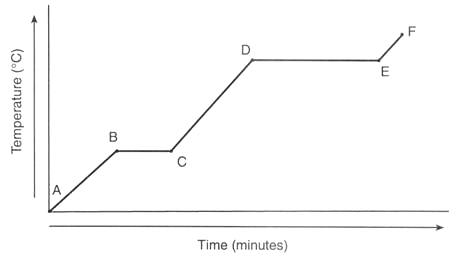
Between which points is the temperature of the substance remaining constant?
A-B only
A-B, C-D, and E-F
B-C only
B-C and D-E
14
Multiple Choice
A measure of the average kinetic energy of the particles in an object is called
Heat
Temperature
Kinetic energy
Enthalpy
15
Multiple Choice

In which region(s) does temperature increase?
Region C only
Regions B and D
Regions A, C, and E
Regions A and B
16
Multiple Choice
The energy transferred between objects that are at different temperatures is called
Heat
Temperature
Energy
Friction
17
Multiple Choice

Describe the substance at letter A.
Solid
Liquid
Melting
Evaporating
18
Multiple Choice

At which letter is the substance changing its state of matter?
A only
A, C, and E
D and E
B and D
19
Multiple Choice

The melting point of the sample is
-60 ºC
20 ºC
60 ºC
100 ºC
20
Multiple Choice
__________ show the change in state of matter as heat is added.
Phase diagrams
Heating curves
Density graphs
Temperature plots

Copyright © 2011 MsRazz ChemClass
Bell Ringer
Complete the handout
while watching the video.
Use the word bank to help
with labeling the heating
curve
Word Bank:
Solid, Liquid,
Gas, Freezing,
Melting,
Vaporization,
and
condensation.
Show answer
Auto Play
Slide 1 / 20
SLIDE
Similar Resources on Wayground

13 questions
3.4.d Eclipses Review
Presentation
•
9th - 12th Grade

20 questions
Evolution of Plant Transport System
Presentation
•
9th - 12th Grade

18 questions
Introduction to Membrane Structure (Review of Water)
Presentation
•
9th - 12th Grade

14 questions
HUMAN CENTERED CASE STUDY
Presentation
•
KG - University

18 questions
Experience Chemistry Lesson 01-2.2
Presentation
•
10th - 12th Grade
![5.1 How Populations Grow [Pt 2]](https://cf.quizizz.com/image/image-loader.svg)
19 questions
5.1 How Populations Grow [Pt 2]
Presentation
•
10th - 12th Grade

20 questions
Chapter 3: Matter and Change
Presentation
•
9th - 12th Grade

12 questions
🌊 Day 5 of Waves
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade