

AAP Final Reivew
Presentation
•
Science
•
9th - 12th Grade
•
Medium
Ms. noble
Used 1+ times
FREE Resource
24 Slides • 87 Questions
1
Kinetic and Potential
2
3
Multiple Choice
Conservation of energy means that
energy can be created but not destroyed
energy can be destroyed but not created
energy can be both created and destroyed
energy can be neither created nor destroyed
4
Explanation Slide...
Conservation of energy states that energy cannot be created or destroyed.
5
Multiple Choice
Kinetic energy can be described as
energy in motion
stored energy
a chemical reaction
connected energy
6
Explanation Slide...
7
Multiple Choice
A bicycle rider is traveling up a hill. When the rider reaches the top of the hill, she stops to rest. Then she travels down the hill. Which statement correctly describes the potential energy and the kinetic energy of the bicycle rider?
As the rider moves up the hill, her kinetic energy increases, and her potential energy decreases.
As the rider reaches the bottom of the hill, her kinetic energy and her potential energy decrease.
When the rider is at the top of the hill, her potential energy is the greatest, and her kinetic energy is the least.
When the rider goes down the hill, her potential energy increases, and her kinetic energy decreases.
8
Explanation Slide...
9
Multiple Choice
As a boulder rolls from the top of a hill to the bottom, how does its potential and kinetic energy change?
Potential and kinetic energy both increase.
Potential energy increases and kinetic energy decreases.
Potential energy decreases and kinetic energy increases.
Potential and kinetic energy both increase.
10
Explanation Slide...
11
Multiple Choice
Sammy studied kinetic and potential energy by observing apples on a tree during a field investigation. What type of energy do the apples on the tree have?
elastic potential energy
mechanical energy
gravitational potential energy
sound energy
12
Explanation Slide...
13
Multiple Choice
If a green golf ball is dropped from a height of 7 feet and a yellow golf ball is dropped from a height of 12 feet, which ball has greater potential energy before it is released?
yellow
green
Both have the same amount of potential energy.
Neither will have more potential energy than the other.
14
Explanation Slide...
15
Multiple Choice
When a tennis ball is dropped, potential energy is destroyed and kinetic energy is created.
True
False
16
Explanation Slide...
17
Multiple Choice

Describe the energy in the picture.
Gravitational Potential
Kinetic
Elastic Potential
No energy.
18
Explanation Slide...
19
Multiple Choice
What word best describes Kinetic Energy?
Stored
Motion
Elastic
Gravity
20
Explanation Slide...
21
Multiple Choice
What word best describes potential energy?
Stored
Motion
22
Explanation Slide...
23
Multiple Choice
Gravitational Potential energy increases as _________ increases.
Speed
Elasticity
Height
Movement
24
Explanation Slide...
25
Multiple Choice

A ball is released from rest at position 1. The diagram shows the ball in four positions as it rolls along a track from left to right. In which position does the ball have its minimum gravitational potential energy and maximum kinetic energy?
1
2
3
4
26
Explanation Slide...
27
Multiple Choice

The illustration shows a roller coaster and indicates four different positions the car might be at as it moves along the track.
At which point in the roller coaster's journey would it's potential energy be changing to kinetic energy
Z-Y
Y-X
W-X
28
Explanation Slide...
29
Multiple Choice

At which point will the roller coaster have the greatest amount of potential energy?
A
B
C
D
30
Explanation Slide...
31
Multiple Choice

The kinetic energy of the pendulum is increasing, and its potential energy is decreasing when it moves from point–
1 to 2.
3 to 4.
4 to 5.
32
Explanation Slide...
33
Multiple Choice

At what point in the pendulum swing would potential energy be the greatest?
1
2
3
34
Explanation Slide...
The correct answer is point 1. It's the highest point of the swing and will therefore have the most potential energy.
35
Multiple Choice

At what point in the pendulum swing would kinetic energy be at its greatest?
1
2
3
36
Explanation Slide...
The correct answer is point 3. We're at the lowest point of the swing and all of the energy is now being used in the motion of the pendulum.
37
Multiple Choice

The kinetic energy of the pendulum is decreasing, and its potential energy is increasing when it moves from point–
1 to 2.
2 to 3.
4 to 5.
38
Explanation Slide...
39
Multiple Choice
Stretching a rubber band, pulling back on a bow string, or pulling a back on a spring are examples of what kind of potential energy?
elastic potential energy
gravitational potential energy
chemical potential energy
40
Explanation Slide...
41
Multiple Choice
Energy stored in food, fuel, or batteries is an example of what type of potential energy?
elastic potential energy
gravitational potential energy
chemical potential energy
42
Explanation Slide...
43
Specific Heat
44
Multiple Choice

According to the ESRT, which material would require the most heat energy to increase the temperature of 1 gram of the material one Celsius degree?
Ice
Water
Basalt
Granite
45
Multiple Choice

Which will heat up faster?
copper
granite
iron
basalt
46
Multiple Choice

Which material would require the greatest amount of heat energy to raise its temperature from 5oC to 10oC? [Refer to the Earth Science Reference Tables.]
Iron
Lead
Granite
Dry Air
47
Multiple Choice

Which will heat up slower?
water
lead
granite
iron
48
Multiple Choice

If the same amount of heat is added to each sample, the smallest change in temperature will occur in the
Iron
Dry Air
Granite
Water
49
Multiple Choice
Which of the following best explains why the sand at the beach is hotter than the water?
Sand has a higher specific heat than water.
Sand has a lower specific heat than water.
There is more water than sand at the beach.
There is more sand than water at the beach.
50
Multiple Choice

What would heat up the fastest if left out in the sunlight on a clear, summer day?
Liquid Water
Dry Air
Lead
Granite
51
Multiple Choice
The amount of energy required to raise the temperature 1ºC for every gram is called____?
Thermal Energy
Specific Heat
Temperature
Kinetic Energy
52
Multiple Choice
Why does water heat heat up and cool down VERY SLOWLY?
High Specific Heat
Low Specific Heat
It's solid
It's always cold
53
Multiple Choice
54
Multiple Choice

55
Multiple Choice
56
Multiple Choice
57
Multiple Choice

58
Multiple Choice
59
Multiple Choice
60
Multiple Choice
Heat ALWAYS moves from
Low to High Temperature
High to Low Temperature
It doesn't move
High to Low Pressure
61
Multiple Choice

62
Multiple Choice
Specific Heat is....
The speed of the particles in a substance
The temperature of the substance
How much energy it takes to heat something up
Heat
63
Multiple Choice
The SI units J/g°C measures
Specific heat
mass
temperature
heat energy
64
Multiple Choice
When heat is absorbed by a substance, what happens to the temperature of the substance?
it increases
it stays the same
it decreases
it depends on the substance
65
Multiple Choice

Some 1kg of water and 1kg of diamond are heated to the same temperature. Which is true?
They both require the same amount of energy
The diamond needs around 8 times more energy
The water needs about 8 times more energy
The diamond heats up slower than the water
The water ends up colder
66
Multiple Choice
When heat is released by a substance, what happens to its temperature?
it increases
it stays the same
it decreases
it depends on the substance
67
Multiple Choice

2kg of water and 4 kg of water are heated to the same temperature. Which is true
They both require the same amount of energy
2kg needs twice as much energy than the 4kg
4kg needs twice the energy the 2kg needs
4kg needs 4 times the energy the 2kg needs
The 2kg is hotter
68
Multiple Choice
If Q=mcΔT , what is ΔT
ΔT=mcQ
ΔT=Qmc
ΔT=Qmc
ΔT=Qcm
c=mΔTQ
69
Multiple Choice
70
Multiple Choice
Which one will have the SMALLEST change in temperature?
71
Conduction, Convection, and Radiation
72
Dropdown

73
Multiple Choice
On a hot summer day, a student dropped an ice cube onto a park bench. Within minutes, the ice cube melted. Which best describes the process the student observed?
radiation
combustion
conduction
convection
74
Drag and Drop

75
Multiple Choice

A cold metal spoon is placed in a bowl of hot soup. Which best describes how the spoon is affected?
The heat from the soup does not affect the temperature of the spoon.
The heat from the soup is conducted through the spoon, making the spoon warmer.
The cold air in the room radiates into the soup, cooling it, and keeping the spoon cool.
The cold from the spoon is conducted into the soup, causing the soup to cool to the same temperature as the spoon.T
76
Drag and Drop

77
Multiple Choice

Mary stands in a swimming pool and notices that the water around her feet is a lot cooler than the water near the surface. Which process causes this difference in temperature?
convection
evaporation
radiation
conduction
78
Dropdown

79
Multiple Choice

Which best explains why a hot air balloon floats?
The heated air expands to fill the balloon. The air in the balloon is now lighter than the surrounding air, causing the balloon to rise.
The heated air expands to fill the balloon. The air in the balloon is now heavier than the surrounding air, causing the balloon to rise.
The heated air contracts to fill the balloon. The air in the balloon is now denser than the surrounding air, causing the balloon to rise.
The heated air contracts to fill the balloon. The air in the balloon is now lighter than the surrounding air, causing the balloon to rise.
80
Drag and Drop

81
Multiple Choice
Which is an example of conduction?
Steam is rising from a cup of hot coffee.
A hot biscuit is warming the plate it is on.
The sun is shining through a window and warming a table.
82
Drag and Drop

83
Multiple Choice
Which best describes the process of convection?
Convection is the primary way heat travels through rays.
Convection is the primary way heat travels through metals.
Convection is the primary way heat travels through appliances.
Convection is the primary way heat travels through liquids and gasses.
84
Dropdown

85
Multiple Choice
James measured the temperature outside his house everyday for 2 months. He observed that on days the sky was mostly cloudy, the temperature was an average of 9 degrees cooler than days the sky was mostly sunny. Which best describes the reason for this pattern?
The clouds blocked radiation from the sun, causing cooler temperatures.
The clouds blocked conduction from the sun, causing cooler temperatures.
The clouds allowed more radiation from the sun to reach the Earth’s surface.
The clouds allowed more conduction from the sun to reach the Earth’s surface.
86
Dropdown

87
Multiple Choice

A small ice cube at a temperature of 0°C is dropped into a glass of water at 28°C and melts. What is the temperature of the water in the glass just after the ice cube melts?
0°C
between 0°C and 28°C
28°C
greater than 28°C
88
Multiple Select

Cold Pack on Person
Heat is transferred from the cold pack to the person.
Heat is transferred from the person to the cold pack
The cold pack is getting warmer. The person is getting colder.
The cold pack is getting colder. The person is getting warmer.
89
Multiple Choice
Which best explains how convected heat causes a hot air balloon to stay in the air?
The heat is falling and replacing cooler air.
The heat is produced from the rising of air currents.
The heat is absorbed through the balloon from the sun.
The heat is rising and circulating, causing the balloon to stay up in the air.
90
Drag and Drop
91
Multiple Choice
Which best explains how ice melts in a person’s hand?
The heat from a person’s hand transfers by radiation to the ice, causing it to melt.
The heat from a person’s hand transfers by conduction to the ice, causing it to melt.
The cool temperature of the ice transfers by radiation to a person’s hand, cooling it off.
The cool temperature of the ice transfers by conduction to a person’s hand, cooling it off.
92
Drag and Drop
93
Multiple Choice

Tina is boiling water in a metal pot for a weather experiment. What is most likely the reason she would need to use a pot holder for moving the metal pot?
Radiation causes the pot to be hot.
Conduction causes the pot to be hot.
Convection causes the pot to be hot.
Transpiration causes the pot to be hot.
94
Drag and Drop
95
Multiple Choice
A pot of water is placed on a stove. The water begins to boil rapidly. Which explains why the water is boiling faster?
The cooler water is rising in the pot to replace the warmer water on top through the process of radiation.
The warm water is rising in the pot to replace the cooler water on top through the process of radiation.
The warm water is rising in the pot to replace the cooler water on top through the process of convection.
The cooler water is rising in the pot to replace the warmer water on top through the process of convection.
96
Dropdown
97
Multiple Choice

A boy touches a stove and gets burned. What transfer of heat caused his hand to get burned?
radiation
conduction
convection
combustion
98
Fill in the Blanks
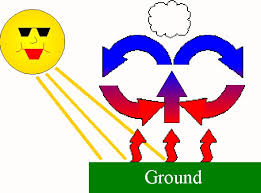
Type answer...
99
Multiple Choice
What is the original source of most of the energy that people use?
Energy that reaches Earth’s surface by radiation from the Sun.
Energy that reaches Earth’s surface by convection from the Sun.
Energy that reaches Earth’s surface by radiation from Earth’s core.
Energy that reaches Earth’s surface by conduction from Earth’s core.
100
Drag and Drop

101
Multiple Choice

Which is the best choice for the handle of a frying pan?
Metal, because it conducts heat.
Wood, because it conducts heat.
Metal, because it does not conduct heat.
Wood, because it does not conduct heat.
102
Dropdown

103
Multiple Choice
Which substance will conduct the least amount of heat?
glass
copper
plastic
aluminum
104
Dropdown

105
Multiple Choice

Which part of the diagram shows conduction?
Part A
Part B
Part C
106
Multiple Choice

107
Multiple Choice
Ice cubes are placed in a glass of water that is at room temperature. Which statement best describes the temperature of the water after 10 minutes?
The temperature decreases because some of the water’s heat is lost to the ice.
The temperature decreases because some of the ice’s heat is lost to the water.
The temperature increases because some of the coolness of the water is absorbed by the ice.
The temperature increases because some of the coolness of the ice is absorbed by the water.
108
Match
conduction
convection
radiation
ironing a shirt
a radiator heating a cold room
a microwave heating food
ironing a shirt
a radiator heating a cold room
a microwave heating food
109
Multiple Choice
Which best describes the flow of heat?
from a larger object to a smaller object
from a warmer object to a cooler object
from a cooler object to a warmer object
110
Multiple Choice
Heat transfer through waves is called___
conduction
convection
radiation
111
Multiple Choice
Which is an example of heat being transferred by conduction?
a furnace heating the air in a school
an ice cube melting in a person’s hand
a pot of water on a stove boiling
the sun heating water in a swimming pool
Kinetic and Potential
Show answer
Auto Play
Slide 1 / 111
SLIDE
Similar Resources on Wayground

107 questions
higiena cyfrowa
Presentation
•
KG - University

100 questions
Unit 2 Notes Alterations of Ecosystems
Presentation
•
9th - 12th Grade

104 questions
Chemistry Semester 1 Credit Recovery
Presentation
•
10th - 12th Grade

108 questions
Chapter 14: Interactions in the Ecosystem
Presentation
•
10th Grade

110 questions
Lesson 5 & 6 - Taxonomy and Dichotomous Keys
Presentation
•
9th - 12th Grade

106 questions
All subject quiz
Presentation
•
8th Grade - University

101 questions
WAVES
Presentation
•
9th - 12th Grade

103 questions
Evolution EOC Review
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

11 questions
EOC Domain 1: Cells
Presentation
•
9th - 10th Grade

14 questions
EOC Domain 2: Genetics
Presentation
•
9th - 10th Grade

10 questions
Exploring the Human Respiratory System
Interactive video
•
6th - 10th Grade