

Biochemistry Pre-test
Presentation
•
Biology
•
9th - 12th Grade
•
Medium
+7
Standards-aligned
David Jones
Used 6+ times
FREE Resource
0 Slides • 50 Questions
1
Multiple Choice

Which subatomic particle is not in the nucleus?
proton
neutron
electron
2
Multiple Choice
Which is not a particle in an atom?
electron
megatron
proton
neutron
3
Multiple Choice
The amino acid tryptophan has a chemical formula of C11H12N2O2. Which element isn't present in this molecule?
hydrogen
calcium
oxygen
nitrogen
4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
7
Multiple Choice

8
Multiple Choice
What is the purpose of an enzyme?
give permission for the reaction
raise activation energy
speed up reactions
control how many reactions occur
9
Multiple Choice
10
Multiple Choice
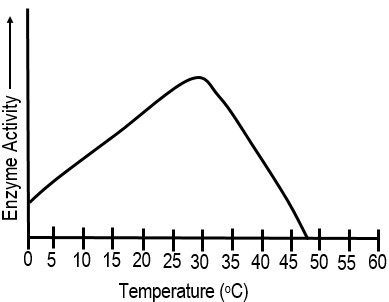
11
Multiple Choice
Most enzymes are composed of .....
Lipids
Carbohydrates
Proteins
Nucleic Acids
12
Multiple Choice
13
Multiple Choice
What type of bond is formed when atoms share electrons?
Covalent bond
Ionic bond
Hydrogen bond
van der Waals force
14
Multiple Choice
Imagine you're a scientist working on a new experiment. What would you call the energy needed to kickstart your chemical reaction?
Activation energy
Enzyme
Catalyst
Substrate
15
Multiple Choice
What is the starting material in a chemical reaction called?
Reactant
Product
Activation energy
Catalyst
16
Multiple Choice
Imagine you are in a chemistry lab. You observe a reaction where electrons are transferred from one atom to another. What type of bond is being formed in this scenario?
Ionic bond
Covalent bond
Hydrogen bond
van der Waals force
17
Multiple Choice
Imagine you are a scientist studying substances. How would you define an element in your research?
A substance that cannot be broken down into simpler substances by chemical means
A substance composed of two or more different elements chemically combined
Atoms of the same element that have different numbers of neutrons
Substances formed as a result of a chemical reaction
18
Multiple Choice
What are substances that release hydrogen ions (H+) in water called?
Acids
Bases
pH
Buffers
19
Multiple Choice
You are making a cup of tea. You add sugar and stir until it completely dissolves. What would you call this type of homogeneous mixture where sugar (one substance) is dissolved in tea (another substance)?
Solution
Mixture
Solute
Solvent
20
Multiple Choice

21
Multiple Choice
22
Multiple Choice

23
Multiple Choice

24
Multiple Choice

25
Multiple Choice

26
Multiple Choice

27
Multiple Choice

28
Multiple Choice

29
Multiple Choice

30
Multiple Choice

31
Multiple Choice

32
Multiple Choice

33
Multiple Choice
34
Multiple Choice

35
Multiple Choice
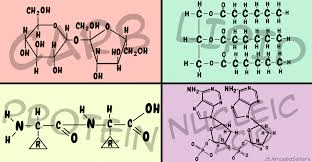
36
Multiple Choice
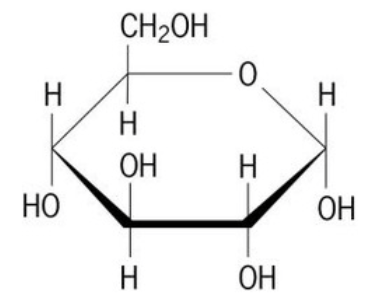
37
Multiple Choice
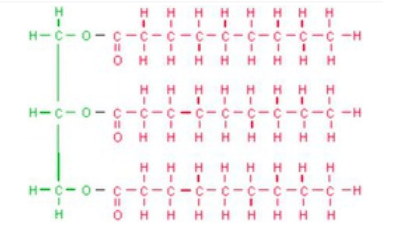
38
Multiple Choice
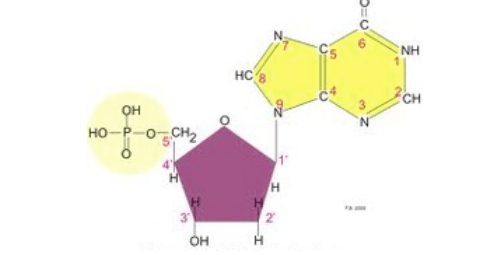
39
Multiple Choice
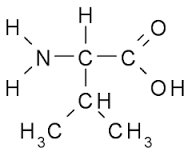
40
Multiple Choice
What is the monomer of a Lipid?
41
Multiple Choice

What is the monomer of a protein?
42
Multiple Choice

What is the monomer of a nucleic acid?
43
Multiple Choice
What is the monomer of a carbohydrate?
44
Multiple Choice
What elements are in a nucleic acid?
CHO
CHOP
CHONP
CHON
45
Multiple Choice

What elements are in a protein?
CHO
CHOP
CHON
CHONP
46
Multiple Choice
What elements are in a Lipid
47
Multiple Choice
What elements are in a carbohydrate
CHO
CHON
CHOP
CHONP
48
Multiple Choice
Bases release which ions ?
hydroxide ions
no ions
hydrogen ions
hydroxide and hydrogen ions
49
Multiple Choice

50
Multiple Choice

Which subatomic particle is not in the nucleus?
proton
neutron
electron
Show answer
Auto Play
Slide 1 / 50
MULTIPLE CHOICE
Similar Resources on Wayground

41 questions
Cell's Organelles
Presentation
•
9th - 12th Grade

41 questions
Air Pollution Lesson
Presentation
•
9th - 12th Grade

45 questions
Unit 4B Pt2
Presentation
•
9th - 12th Grade

42 questions
Six Kingdoms EOC Review
Presentation
•
9th - 12th Grade

43 questions
Macromolecules
Presentation
•
9th - 12th Grade

45 questions
Building Amino Acid
Presentation
•
9th - 12th Grade

43 questions
Evidence of Evolution
Presentation
•
9th - 12th Grade

44 questions
Chromosomes, Biological Technology, and Mutation
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Biology

14 questions
Ecological Succession: Primary and Secondary
Quiz
•
9th Grade

20 questions
Structure & DNA Replication Quiz
Quiz
•
9th Grade

50 questions
Biology EOC Review
Quiz
•
9th Grade

33 questions
Module C - Virus vs Cells, Biomolecules & Enzymes
Quiz
•
9th Grade

25 questions
photosynthesis and cellular respiration
Quiz
•
9th Grade

25 questions
STAAR Evolution Vocabulary
Quiz
•
9th Grade

20 questions
Food Chains and Food Webs
Quiz
•
7th - 12th Grade

20 questions
Genetics 2026
Quiz
•
10th Grade