

CA Remediation AKS 1a/b & 4
Presentation
•
Science
•
9th - 12th Grade
•
Medium
Standards-aligned
Marissa Brown
Used 4+ times
FREE Resource
15 Slides • 29 Questions
1
AKS 1a/b & 4a-c Remediation
2
Chapter 10.1 "Radioactivity" Learning Objectives
Explain what happens during nuclear decay
Identify and explain the three main types of nuclear radiation
Describe how the different types of nuclear radiation affect atoms
3
Nuclear Decay
Radioactivity: Natural process where an unstable atomic nucleus emits charged particles and energy
Radioisotope: Atom with an unstable nucleus
Nuclear Decay: Atoms (radioisotopes) of one element spontaneously change into atoms of a different element (e.g., U-238 decays to Th-234)
4
Multiple Choice
Radioactivity
Any atom containing an unstable nucleus
Process where an unstable atomic nucleus emits charged particles
Atoms of one element change into atoms of a different element or elements
Total protonic reversal (shout out to "Ghost Busters")
5
Multiple Choice
Radioisotope
Any atom containing an unstable nucleus
Process where an unstable atomic nucleus emits charged particles
Atoms of one element change into atoms of a different element or elements
Total protonic reversal (shout out to "Ghost Busters")
6

7
Multiple Choice
Nuclear Decay
Any atom containing an unstable nucleus
Process where an unstable atomic nucleus emits charged particles
Atoms of one element change into atoms of a different element or elements
Total protonic reversal (shout out to "Ghost Busters")
8
Types of Nuclear Radiation
Nuclear Radiation: Charged particles and energy emitted from nuclei of radioisotopes
Three Types of Nuclear Radiation:
Alpha Particles
Beta Particles
Gamma Rays
9
Multiple Choice
Nuclear Radiation is best defined as
Charged particles and energy emitted from nuclei of radioisotopes
Non-charged particles and energy emitted from nuclei of radioisotopes
Charged particles and energy retained in nuclei of radioisotopes
Non-charged particles and energy emitted from nuclei of radioisotopes
10
Multiple Choice
Which is NOT a type of Nuclear Radiation?
Alpha Particles
Beta Particles
Gamma Rays
C Rays
11
Alpha Decay
Alpha Particle: Positively charged atomic particle with two protons and two neutrons (e.g., like Helium without the two electrons)
Alpha Decay: Nuclear decay that releases alpha particles = a nuclear reaction (expressed similarly to a balanced chemical reaction)
Alpha particles are the least penetrating type of nuclear radiation

12
Multiple Choice
Alpha Particles can be stopped by a single piece of paper.
True
False
13
Multiple Choice
Alpha particles are weak, but they travel long distances.
True
False
14
Beta Decay
Beta Decay: Thorium-234 nucleus experiences Beta Decay, where one of its neutrons decomposes into one proton and one electron, creating
The new proton stays trapped in Thorium's nucleus and the electron is released as negatively charged radiation called Beta Particles
Beta Particle: An electron emitted by an unstable nucleus
Electrons negatively charged assigned a -1 Atomic Number
Electrons have negligible mass compared to Protons = 0 Mass Number
Beta Particles are more penetrating than Alpha Particles (smaller mass and faster speed)
15

16
Multiple Choice
Beta Particles cannot be stopped even by a thick sheet of metal.
True
False
17
Gamma Decay
Nuclear Radiation emitted from an unstable nucleus in the form of Gamma Rays (not charged particles like Alpha and Beta Particles)
Gamma Radiation has no mass and no charge
Gamma Rays are energy waves that travel through space at the speed of light (see Electromagnetic Spectrum)
During Gamma Decay, mass of the atom stays the same, but the energy of the nucleus decreases.
Often happens during/alongside Alpha or Beta Decay
18

19
Multiple Choice
Gamma Rays are much more penetrating than Alpha or Beta Particles.
True
False
20
Multiple Choice
In Gamma Decay, the nucleus _____________ energy.
increases
decreases
maintains the same energy
it's the electrons that decrease in energy not the nucleus
21
Multiple Choice
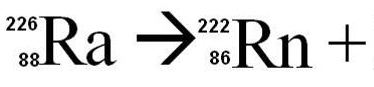
22
Multiple Choice

23
Multiple Choice

24
Multiple Choice
146C --> 0-1e + ________
25
Multiple Choice

26
Multiple Choice
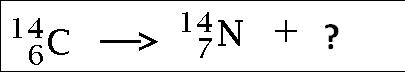
27
Multiple Choice

Identify the missing variable in the following reaction.
28
Multiple Choice

Identify the missing variable in the following reaction.
29

Radiometric Dating and Half-life
The time it takes for a radioactive isotope to decay is measured using a term called half-life.
The half-life of a radioactive substance is the time it takes for half of its unstable atoms to break
down into daughter products.
Isotopes that are extremely unstable decay quickly

30
Multiple Choice
The half-life of Carbon-14 is 5,730 years. Carbon-14 decays into Nitrogen-14.
After 5,730 years, how much of the sample would become Nitrogen-14?
Hint: Half-life represents the time required for half of the substance to decay.
25%
50%
75%
100%
31

The half-life of the radioactive isotope is a critical factor. It determines the timescale over which decay occurs and helps determine the appropriate dating method.
Half-Life
32
What is a Half Life?
The rate that an element decays never changes
Half Life: the amount of time it takes for half of a radioactive element to decay
Ohio Grade 8 | Lesson 6.3

33
Fill in the Blanks

Type answer...
34
Multiple Choice
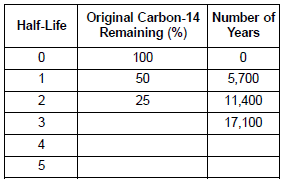
After 22,800 years, approximately what percentage of the original carbon-14 remains?
15%
12.5%
6.25%
3.125%
35
Multiple Choice
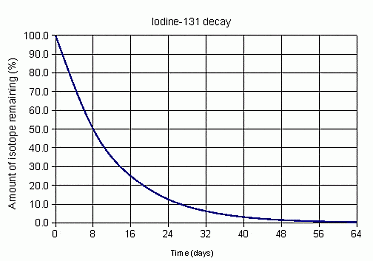
36
Multiple Choice
The % of the parent isotope remaining after 1 Half Life.
50%
25%
12.5%
6.25%
37


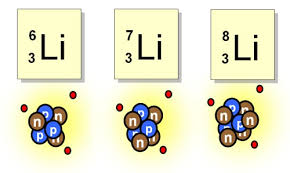
ISOTOPES
Atoms with the same number of protons and a
different number of neutrons
38


ISOTOPE NOTATION:
39
Multiple Choice

40
Multiple Choice
41
Multiple Choice
42
Multiple Choice

43
Multiple Select
Atomic mass = ______ + _______
Electrons
Neutrons
Protons
Nucleus
44
Multiple Choice
Isotopes of the same element have the same number of
Electrons
Protons
Neutrons
Elements
AKS 1a/b & 4a-c Remediation
Show answer
Auto Play
Slide 1 / 44
SLIDE
Similar Resources on Wayground

38 questions
History of the Atomic Structure
Presentation
•
9th - 12th Grade

37 questions
Forensic Science Lesson 09: Hair Evidence Part 2
Presentation
•
9th - 12th Grade

35 questions
Nuclear fission vs fusion
Presentation
•
9th - 12th Grade

40 questions
Crash Course A&P #36-37 Metabolism and Nutrition
Presentation
•
9th - 12th Grade

39 questions
PSU4.A Pg 3-4
Presentation
•
9th - 12th Grade

39 questions
Active in Cell Membrane
Presentation
•
9th - 12th Grade

41 questions
Biological Relationship Scenarios
Presentation
•
9th - 12th Grade

37 questions
Water Usage and Management
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

11 questions
EOC Domain 1: Cells
Presentation
•
9th - 10th Grade

14 questions
EOC Domain 2: Genetics
Presentation
•
9th - 10th Grade

10 questions
Exploring the Human Respiratory System
Interactive video
•
6th - 10th Grade