

Nuclear Fission & Fusion
Presentation
•
Science
•
9th - 12th Grade
•
Medium
+1
Standards-aligned
Allison Boggess
Used 2+ times
FREE Resource
15 Slides • 29 Questions
1
Nuclear Fission & Fusion
Unit 4: Nuclear Theory
2
Isotope Recap
Isotopes are elements with the same number of protons and different numbers of neutrons
Different numbers of neutrons means different mass
Mass = Protons + Neutrons
Isotopes are named for their different mass number
Ex. Carbon-12 has a mass of 12 and Carbon-13 has a mass of 13
3
Multiple Choice
Isotopes are atoms of the same element with different ____________.
Number of Neutrons & Mass Number
Number of Protons & Atomic Number
Number of Protons, Neutrons, and Electrons
Number of Electrons & Mass Number
4
Multiple Choice

What is the ATOMIC MASS of the isotope Carbon-14?
14
6
12
20
5
Multiple Choice

Carbon-14 has ____ protons, ____ electrons, and _____ neutrons.
6, 6, 8
6, 6, 6
6, 6, 14
6, 10, 14
6
Multiple Choice
Isotopes of the same element have the same number of ________ but a different number of _______.
protons, neutrons
neutrons, protons
protons, electrons
electrons, neutrons
7
Multiple Choice

By studying the picture, you can determine that Carbon-13 has ____ protons.
6
13
12
14
8
Multiple Choice

Using the picture, you can determine that Carbon-13 has ____ neutrons.
13
6
12
7
9

A radioactive isotope:
• Has an unstable nucleus
• Spontaneously emits a particle and
decays into another element (to become
more stable)
10
Unstable Nuclei = Radiation
Atoms with more than 84 protons are unstable
Radioactivity is the emission of particles/energy from the nucleus
The nucleus is too big
This gradually makes more stable isotopes

11


When are nuclei unstable? (naturally radioactive)
a.
Large nuclei (Z (atomic mass) > 82) – electrical forces of repulsion are greater than strong forces of attraction
b.
Wrong neutron : proton ratio
12


When are nuclei unstable?
Bigger atoms require more neutrons per
proton to keep the atom stable
13
Multiple Select
What are two reasons that a nucleus might be unstable?
Too many electrons
Too large - mass greater than 82
Ratio of protons and neutrons not correct
Too few protons
14
Multiple Select
Which of these atoms is most likely to undergo some form of radiation? (you can choose more than 1)
Uranium
Oxygen
Plutonium
Calcium
Platinum
15
16

17


18

19

20
Multiple Choice
Breaking up the nucleus of an atom is called _________?
Fusion
Nuclear Reaction
Fission
Particle Accelerator
21
Multiple Choice
Fission is --
fusing together
division
22
Multiple Select
Which of the following correctly display nuclear fission?
23
Multiple Choice
24

25
The Sun
a massive ball of plasma.
It is constantly burning so hot that hydrogen is fused together to form helium.
This happens at such a rate that a huge amount of energy is released.

26
Multiple Choice
The process of combining the nuclei of atoms to make different atoms is called _________?
Fission
Nuclear Reaction
Fusion
Particle Accelerator
27
Multiple Choice
Fusion is --
fusing together
division
28
Multiple Choice
29
Multiple Select
Which of the following correctly display nuclear fusion?
30
Multiple Choice
31
Multiple Choice
In the sun
32
Fusion or Fission?
33
Multiple Choice
When small nuclei are exposed to intense heat and pressure, they join to create a larger nucleus, releasing huge amounts of energy
Nuclear Fusion
Nuclear Fission
34
Multiple Choice
In which type of reaction do two lighter nuclei combine to form one heavier nucleus?
combustion
oxidation
nuclear fission
nuclear fusion
35
Multiple Choice
Two smaller atoms combine and form a larger, heavier atom.
Fusion
Fission
36
Multiple Choice

Scenario A shows--
Nuclear Fusion
Nuclear Fission
37
Multiple Choice

Scenario B shows--
Nuclear Fusion
Nuclear Fission
38
Multiple Choice

Which type of reaction does the diagram illustrate?
Fission
Fusion
Alpha Decay
Beta Decay
39
Multiple Choice
In which process does a heavy nucleus split into two lighter nuclei?
freezing
condensation
fission
fusion
40
Multiple Choice
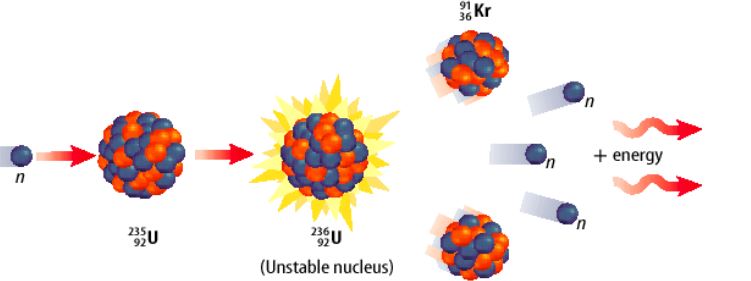
41
Multiple Choice
42
Multiple Choice
What occurs in both fusion and fission reactions?
Small amounts of energy are converted into large amounts of matter.
Small amounts of matter are converted into large amounts of energy.
Heavy nuclei are split into lighter nuclei.
Light nuclei are combined into heavier nuclei.
43
Categorize
Deals with unstable nuclei
Means come together
Creates two smaller nuclei
Creates the most energy
Creates radioactive waste
Creates helium
Is what causes half-life
Requires very high temperatures
Happens in the sun
Means to split
No radioactive waste
Happens in nuclear power plants




Separate into fission or fusion
44
Complete Fission & Fusion Notes
Complete pages 3 & 4 in your Guided Notes!
Nuclear Fission & Fusion
Unit 4: Nuclear Theory
Show answer
Auto Play
Slide 1 / 44
SLIDE
Similar Resources on Wayground

35 questions
Nuclear fission vs fusion
Presentation
•
9th - 12th Grade

39 questions
PSU4.A Pg 3-4
Presentation
•
9th - 12th Grade

38 questions
History of the Atomic Structure
Presentation
•
9th - 12th Grade

37 questions
Forensic Science Lesson 09: Hair Evidence Part 2
Presentation
•
9th - 12th Grade

42 questions
Cell Division Day 3: Meiosis
Presentation
•
9th - 12th Grade

39 questions
CCU5 Test Prep
Presentation
•
9th Grade - University

43 questions
Atoms Lesson
Presentation
•
KG

36 questions
APES FRQ; How to answer the question that is asked?
Presentation
•
10th - 12th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade