

Nuclear Decay & Transmutation
Presentation
•
Science
•
9th - 12th Grade
•
Hard
Standards-aligned
Allison Boggess
FREE Resource
18 Slides • 30 Questions
1
Nuclear Decay & Transmutation
Unit 4: Nuclear Theory
2
Multiple Choice
3

4
Multiple Choice
5


Transmutation
• Changing into another element through
radioactive decay
6

7
Alpha Decay
emits an alpha particle (atom made of 2 protons and 2 neutrons – helium)
Loses energy quickly Least penetrating radiation Can be stopped by a sheet of paper Can cause radiation burns Used in smoke detectors, pacemakers, static eliminators

8
Alpha Decay
Makes atomic number become 2 less and mass number become 4 less

9


Alpha Decay
• Radiation through the loss of 2p + 2n or
(helium)
10

11
Multiple Choice
12
Multiple Choice
Alpha particles (alpha decay) are comprised of
2 Protons & 2 Neutrons
4 Neutrons & 2 Electrons
an electron
an energy wave
13
Multiple Choice
What is the symbol for an alpha particle?
42He
0-1e
0+1 e
10n
14
Multiple Choice
15
Multiple Choice
In alpha decay, the mass number...
increases by 2
decreases by 2
increases by 4
decreases by 4
16
Multiple Choice
In alpha decay, the atomic number...
increases by 2
decreases by 2
increases by 4
decreases by 4
17


Question 3a
Balance the nuclear equation after alpha
decay
18


Question 3a
Balance the nuclear equation after alpha
decay
19
Multiple Choice
20
Multiple Choice
85209At = ___ + 24He
21
Multiple Choice
88226Ra = ___ + 24He
22

23
Beta Decay
neutron decays into a proton and emits an electron (Beta particle)
Caused by a weak force
Much faster and more penetrating
Stopped by sheet of aluminum foil

24
Beta Decay
Can cause radiation burns and some cell damage
Used to treat eye and bone cancer, quality control in paper making
Makes atomic number increase
by 1 and mass number stays the same

25


Beta Decay
• Radiation where a neutron splits, giving off
an electron and becoming a proton in the
new element
26

27
Multiple Choice
Beta particles (Beta decay) are comprised of
2 Protons & 2 Neutrons
4 Neutrons & 2 Electrons
an electron
an energy wave
28
Multiple Choice
In beta decay, the mass number
the mass number decreases by 1
the mass number does not change
the mass number increases by 1
29
Multiple Choice
In beta decay, the atomic number
the atomic number does not change
the atomic number increases by 1
the atomic number decreases by 1
30
Multiple Choice
In the equation, 146C --> 147N + 0-1e, the _______ decay of radioactive carbon-14 results in the creation of a new nitrogen-14 atom.
31



Question 3b
Balance the nuclear equation after beta decay
Remember in beta decay a neutron changes into a proton by giving off an
electron
32



Question 3b
Balance the nuclear equation after beta decay
Remember in beta decay a neutron changes into a proton by giving off an
electron
33
Multiple Choice
614C = ___ + -10e
34
Multiple Choice
2760Co = ___ + -10e
35
Multiple Choice
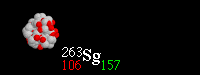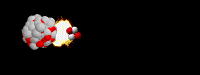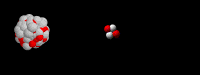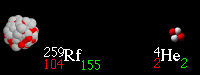
36
Multiple Choice

What type of radioactive decay is happening?
Alpha
Beta
Gamma
37
Multiple Choice

What type of radioactive decay is happening?
Alpha
Beta
Gamma
38
Multiple Choice

What type of radioactive decay is happening?
Alpha
Beta
Gamma
39
Multiple Choice

What type of radioactive decay is happening?
Alpha
Beta
Gamma
40
Multiple Choice
Alpha or beta decay:
146C --> 0-1e + ________
Alpha
Beta
41
Multiple Choice

What type of radioactive decay is happening?
Alpha
Beta
Gamma
42
Multiple Choice
Balance the equation:
146C --> 0-1e + ________
43
Multiple Choice
Alpha or beta decay?
92238U = ___ + 24He
Alpha
Beta
44
Multiple Choice
Balance the equation:
92238U = ___ + 24He
23892U
23490Th
23088Ra
23893Np
45
Draw
Complete the following equation
46
Draw
Complete the following equation
47
Multiple Choice
Which reaction represents fusion?
48
Transmutation Practice
Complete "Decay & Transmutation Practice" in your Guided Notes!
Nuclear Decay & Transmutation
Unit 4: Nuclear Theory
Show answer
Auto Play
Slide 1 / 48
SLIDE
Similar Resources on Wayground

41 questions
Nixon, Ford, Carter
Presentation
•
9th - 12th Grade

42 questions
Introduction to Electricity and Conversion of Units
Presentation
•
9th - 12th Grade

40 questions
Balancing and 5 Types of Chemical Reactions
Presentation
•
9th - 12th Grade

43 questions
Light Waves
Presentation
•
8th - 11th Grade

41 questions
PMA 2 Review-2021/22
Presentation
•
9th - 12th Grade

42 questions
Review - 1st 6wks
Presentation
•
9th - 12th Grade

43 questions
Energy Changes in Chemical Reactions Tutorial
Presentation
•
9th - 12th Grade

41 questions
Nuclear Chemistry
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade