

*Topic 3 Polarity
Presentation
•
Science
•
11th Grade
•
Easy
Standards-aligned
Marisa Macrides
Used 1+ times
FREE Resource
21 Slides • 18 Questions
1

As you work through this presentation, please fill out your notes sheet and answer the questions as you go.
2
Recall: The position of an element on the periodic table determines its electronegativity.

3

4
Multiple Select
A molecule is polar when.... (Choose all that apply)
When the central atom has no lone pairs
When the central atom has 1 or more lone pairs
When the molecule has 1 or more polar bonds
5

6
Multiple Choice
What is the reason the shared electrons are held closer to fluorine in the polar molecule H-F?
Fluorine has a lower electronegativity
Hydrogen has a higher electronegativity
Fluorine has a higher electronegativity
Both atoms share electrons equally
7

8
Multiple Choice

9
Multiple Choice
True or False: All diatomic molecules are nonpolar.
True
False
10

11
Multiple Choice
What is the primary factor that determines whether a molecule is polar or nonpolar?
The size of the molecule
The molecular weight
The shape of the molecule and the distribution of charge
The number of electrons
12

13
Multiple Choice
What are the general rules to determine if a molecule is polar or nonpolar?
Asymmetrical molecules are always nonpolar
Asymmetrical molecules are always polar
14
Take your time working through these examples! They will take you step by step on how to determine the polarity of molecules.
15

Draw the Lewis Diagram on your notes sheet on your own first, then click next to check if you drew them correctly!
16

17
Multiple Choice
Based on the two Lewis Structures you drew, which molecule is Polar?
HF
H2
18

19

Draw the Lewis Diagram on your notes sheet on your own first, then click next to check if you drew them correctly!
20

21
Multiple Choice
Based on the two Lewis Structures you drew, which molecule is Polar?
CO2
COS
22

23

Draw the Lewis Diagram on your notes sheet on your own first, then click next to check if you drew them correctly!
24

Recall: Did having a lone pair of electrons on the central atom make the molecule polar or nonpolar?
25
Multiple Choice
H2O and NH3 have at least one lone pair of electrons on the central atom, so the molecules must be...
Polar
Nonpolar
26

27

Draw the Lewis Diagram on your notes sheet on your own first, then click next to check if you drew them correctly!
28

29
Multiple Choice
Which molecule is asymmetrical and therefore polar?
CH4
CH3Cl
30

31
Review Questions
32
Labelling
Label the molecule accordingly.
Higher electronegativity
Lower electronegativity
33
Multiple Choice
The electrons in a polar covalent molecule are shared...
Evenly
Unevenly
Electrons are not shared
None of the Above
34
Multiple Choice
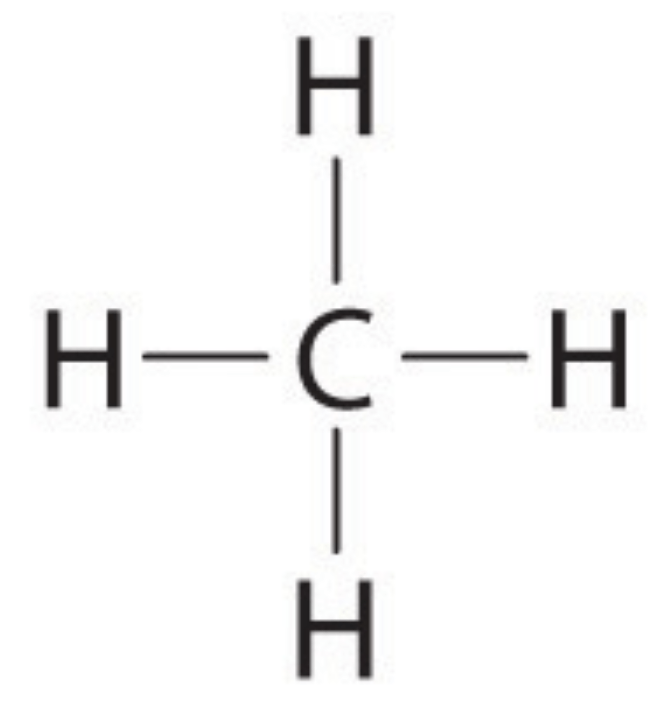
Classify the following molecule.
polar
nonpolar
35
Multiple Choice

Classify the following molecule.
polar
nonpolar
36
Multiple Choice

Why is the molecule nonpolar?
There are lone pairs on the central atom.
There are different types of elements bonded to the central atom.
There are no lone pairs on the central atom and all of the atoms bonded to the central atom are the same. (symmetrical)
37
Multiple Choice

Classify the following molecule.
polar
nonpolar
38
Multiple Choice

Why is the molecule polar?
There is a lone pair on the central atom, creating a negative side to the molecule.
There are different types of elements bonded to the central atom.
There are no lone pairs on the central atom and all of the atoms bonded to the central atom are the same.
39
Poll
How confident do you feel about this topic now?

As you work through this presentation, please fill out your notes sheet and answer the questions as you go.
Show answer
Auto Play
Slide 1 / 39
SLIDE
Similar Resources on Wayground

31 questions
Impressions: Footwear, Tire Tracks, & Soil
Presentation
•
11th - 12th Grade

31 questions
14.2 Urinary System
Presentation
•
11th - 12th Grade

33 questions
Chapter 5 - Morality
Presentation
•
11th Grade

34 questions
Phy vs Chem depth
Presentation
•
11th Grade

33 questions
Acids, Bases, and Covalent Compound Names
Presentation
•
11th Grade

34 questions
Collision Theory & Reaction Rate Experiment
Presentation
•
KG

33 questions
EAPP Module 1
Presentation
•
11th Grade

35 questions
1-2 PRE-LAB Using BP to Identify Unknown Liquids
Presentation
•
11th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade

49 questions
AP Environmental Science Final Exam Review
Quiz
•
10th Grade - University

16 questions
Taxonomy and Classification
Quiz
•
6th - 12th Grade

50 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

28 questions
Greenhouse effect
Quiz
•
9th - 12th Grade