

D Assessement part #1 Review
Presentation
•
Science
•
8th Grade
•
Medium
+8
Standards-aligned
Janet Webster
Used 8+ times
FREE Resource
6 Slides • 54 Questions
1
D Assessement part #1 Review

2

3
Multiple Choice
Which elements have 1 valence electron?
period 1
group 1
group 4
4
Multiple Choice

These elements are in a:
group
period
5
Multiple Choice

These elements are in a:
group
period
6
Multiple Choice
The element is in group 13, and symbol B
Metal
Metalloid
Non Metal
7
Multiple Choice
Group 15 with symbol N
Metal
NOn metal
Metalloid
8
Multiple Choice
Element Ra
Metal
Non metal
Metalloid
9
Multiple Choice
Group 16 with symbol of S
Metal
Non Metal
Metalloid
10

11
Multiple Choice

12
Multiple Choice

13
Multiple Choice

Which of the following formulas would be used to describe molecule 4?
C3HO
C4HO
CH4O
CH3O
14
Multiple Choice

Which elements are in the formula shown above?
Cobalt only
Carbon and Oxygen
Copper only
Carbon and Osmium
15
Multiple Choice

Which of the following accurately describes the chemical formula of the molecule shown above?
3CH8O
C3H7O
C4H8O2
C3H8O
16
Multiple Choice
Which of the following statements accurately describes the number of atoms in a molecule of ammonia (NH3)?
Three atoms of nitrogen and three atoms of hydrogen
One atom of nitrogen and three atoms of hydrogen
Three atoms of nitrogen and one atom of hydrogen
One atom of nitrogen and one atom of hydrogen
17
Multiple Choice
Alanine is an amino acid with formula C3H7NO2. How many atoms in all are in each molecule of alanine?
3
7
12
13
18
Multiple Choice
How many atoms of sulfur are in each molecule of H2SO4?
1
4
2
7
19
Multiple Choice
Which chemical formula has 11 atoms total
C3H8
C2H6
CH6
C4H2
20
Multiple Choice
21
Multiple Choice
An atom has an atomic number of 15 and a mass number of 31. How many protons are there in the atom?
15
31
16
47
22
Multiple Choice
23
Open Ended
Describe the formula
Ba ( OH)3 in words- Explain each elements total atoms.
24

STUDY THIS INFORMATION BEFORE YOU GOT TO NEXT SLIDE. REMEMBER A CHEMICAL REACTION PRODUCES A NEW SUBSTANCE WITH DIFFERENT PROPERTIES
25
Multiple Choice
Which of the following examples shows that a chemical reaction has occurred?
Teeth chop up an apple slice into smaller pieces.
Sugar placed in sugar water sinks to the bottom.
A glass jar shatters into many pieces when dropped.
Bleach gets rid of a ketchup stain on a white shirt.
26
Multiple Choice
27
Multiple Choice
28
Multiple Choice
29
Multiple Choice
30
Multiple Choice
31
Multiple Choice

32
Multiple Choice

Which student correctly recorded all the changes that took place?
33
Multiple Choice
34
Multiple Choice

35

36
Multiple Choice
The Great Rift Valley in Africa and the Mid-Ocean Ridge were all created by this boundary
Divergent
Convergent
Reform
Transform
37
Multiple Choice

Transform boundary
Divergent boundary
Subduction boundary
Convergent boundary
38
Multiple Choice

fossil layers
volcanic islands
deep-sea sediment
seafloor spreading
39
Multiple Choice

The San Andreas Fault in California is an example of a ________.
divergent boundary
convergent boundary
transform boundary
subduction zone
40
Multiple Choice
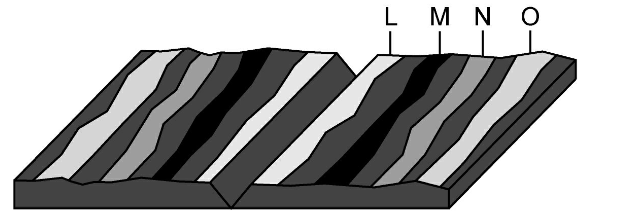
Where would the oldest rock be found?
L
M
N
O
41
Multiple Choice

What plate boundary feature is also shown?
A ridge formed by an inactive convergent boundary
A valley forming at an active divergent boundary
A fault forming at an active transform boundary
A volcano formed by an active transform boundary
42
Multiple Choice

Which student correctly identified the geologic process that formed the Himalayan mountains?
1
2
3
4
43
Multiple Choice
The San Andreas fault is a transform fault. Students were asked to make maps predicting which way land would most likely move over thousands of years. Which one is correct?
44
Multiple Choice
Which diagram correctly models the relative plate motion on each side of a mid-ocean ridge?
45
Multiple Choice

What are the three types of Evidence that supports Alfred Wegener's Theory?
Fossils, Fish, and Land
Sedimentary, Magma, and Climate
Fossils, Land forms, and Climate
Climate, Land forms, and Metamorphic rock
46
Multiple Choice

47
Multiple Choice

One piece of evidence for Plate Tectonics is that--
some continents appear to fit together like a puzzle.
we have video footage of the plates actually moving.
all continents have formed from volcanic eruptions.
the continents are no longer moving.
48
Multiple Choice

Fossil records indicate that the continents were once connected. How do fossil records support this claim?
We have been able to transport the same types of fossils from one continent to another.
Fossils of the same organism that are chemically and physically similar have been found on continents that are now separated by oceans.
Some continents contain fossils of many different types of organisms.
49

50
Multiple Choice
Why didn't Zoe get the ball in the hole on her first try?
right direction but not enough strength
wrong direction but right amount of strength
poor quality of golf club
didn't have proper golf shoes
51
Multiple Choice
Why is the egg NOT moving before Zoe hits the pan?
forces are balanced
forces are unbalanced
there are no forces
none of these
52
Multiple Choice
Which of these involves balanced and unbalanced forces?
golf
pool (billiards)
hoverboarding
all of these
53
Multiple Choice
When Izzy and Zoe pull on the rope with the same amount of force, the forces are _____.
increased
unbalanced
balanced
decreased
54
Multiple Choice
When Izzy and Zoe pull on the rope with the same amount of force, the forces are _____.
increased
unbalanced
balanced
decreased
55
Multiple Choice
A car traveling at constant speed?
Balance
Unbalance
56
Multiple Select

This block will _____.
change its motion
accelerate
change its velocity
keep on doing what it is already doing
57
Fill in the Blanks

Type answer...
58
Multiple Choice

The net force acting on the object is acting _____.
to the right
to the left
up
down
59
Multiple Choice
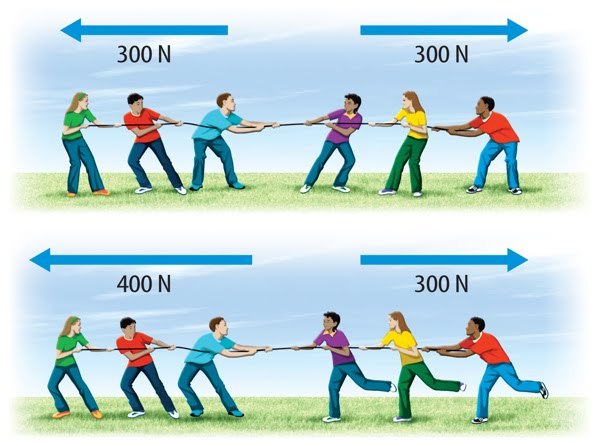
60
Multiple Choice
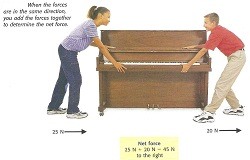
D Assessement part #1 Review

Show answer
Auto Play
Slide 1 / 60
SLIDE
Similar Resources on Wayground

57 questions
Synthetic fibers and Plastic
Presentation
•
8th Grade

53 questions
MASA PENDUDUKAN JEPANG
Presentation
•
8th Grade

55 questions
Earth Science Review Lesson
Presentation
•
8th Grade

55 questions
Science TCAP Practice
Presentation
•
8th Grade

58 questions
Petroski - Physical Science LEAP Review
Presentation
•
8th Grade

55 questions
S8GU1L3 - The life cycle of stars
Presentation
•
8th Grade

55 questions
Teach .Ed. Periodic table/Mixtures
Presentation
•
8th Grade

60 questions
Ecosystems & Cycles
Presentation
•
8th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

12 questions
PS.8.1.2, PS.8.1.3, PS.8.1.5 Review
Quiz
•
8th Grade

20 questions
Moon Phases and Eclipses
Quiz
•
6th - 8th Grade

22 questions
6th & 8th Grade Science Material SOL Review
Quiz
•
6th - 8th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade

48 questions
8th Grade NC Science EOG Review 2
Quiz
•
8th Grade

19 questions
Renewable and Nonrenewable Resources (CK)
Quiz
•
8th Grade

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade