

3.1 Enzymes
Presentation
•
Biology
•
10th - 12th Grade
•
Medium
+1
Standards-aligned
Lloyd Cascabel
Used 5+ times
FREE Resource
58 Slides • 43 Questions
1

2
Macromolecules Review
Living things are composed of 4 macromolecules:

• amino acids for polypeptides (proteins)
• fatty acids for lipids
• monosaccharides such as glucose for polysaccharides
• nucleotides form polynucleotides (DNA & RNA)
3

Macromolecules have simple and complex forms
4

5
The next slides will test whether you remember the details of the slides you just read.
If you need to review the previous slides again, feel free to do so.

6
Multiple Choice

7
Multiple Choice
8
Multiple Choice
9
Multiple Choice
10
Multiple Choice
11
Multiple Choice

Simple sugars like glucose can be used to make larger organic molecules. Identify a larger molecule made from simple sugars.
12
Multiple Choice

13
Multiple Choice
14
Multiple Choice
15
Multiple Choice

16
Multiple Choice
Enzymes are an example of
Lipids
Carbohydrate
Protein
Nucleic Acid
17
Multiple Choice
18
Enzymes
Enzymes are functional proteins (quarternary structure) that act as biological catalysts.
Catalysts are substances that increase the rate of a chemical reaction without becoming part of being changed in the process.

19
Enzymes
Enzymes can either break complex substances into simpler forms (catabolic), or build complex substances from simple molecules (anabolic).

20
Key Terms
Catalyst - a substance that increases the rate of chemical reaction and is not changed by the reaction
Enzymes - proteins that function as biological catalysts
Chemical Reactions - joining or breaking down of substrates; can be anabolic or catabolic
Substrates - molecule/s that undergo reactions
Product - the result of a chemical reaction.
21
Naming Enzymes
Enzymes are named according to the reaction that they catalyze. For example enzymes which catalyse the breakdown of carbohydrates are called Carbohydrases.
- breaks down lipids = Lipases
- breaks down proteins = Proteases
- breaks down amylose (starch) = amylase
- breaks down maltose = maltase
- synthesizes ATP = ATP Synthase
22
Multiple Choice
23
Multiple Choice
Enzymes are an example of
Lipids
Carbohydrate
Protein
Nucleic Acid
24
Multiple Choice
25
Multiple Choice
26
Multiple Choice
27
Multiple Choice
This enzyme breaks down lactose.
fructase
amylase
lactase
maltase
28
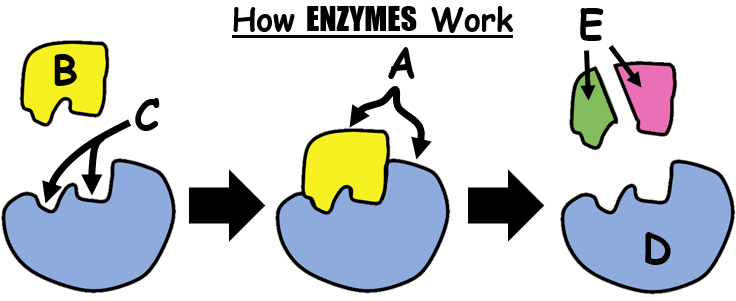
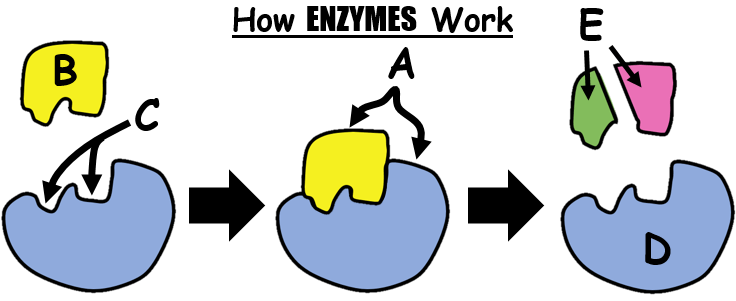
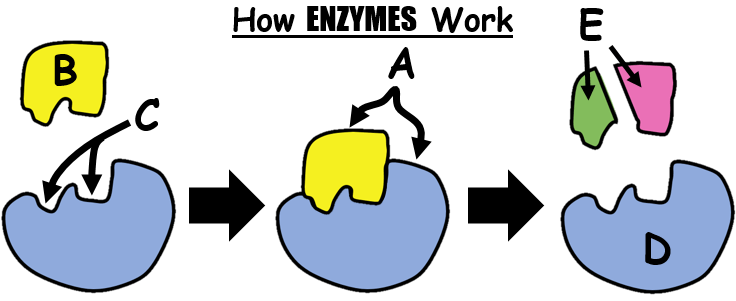
Lock and Key Mechanism
A belief on how Enzymes catalyze reactions is the lock and key mechanism. This principle explains that in order for an enzyme to work it must perfectly fit to a molecule. Enzymes are therefore highly SPECIFIC.
Lactase will only catalyze the breakdown of lactose into simple sugars but it will not work on any other disaccharide. It will certainly not work too on proteins of any kind.
The fit has to be perfect much like a key and a lock.
29
Lock and Key Mechanism
An area or a side of the enzyme have a particular shape (called Active Site) which lets in a very specific substrate just like a key to a lock.
When they join together, they form an Enzyme-Substrate Complex where the substrate may either be broken down or merged with another substrate.

30
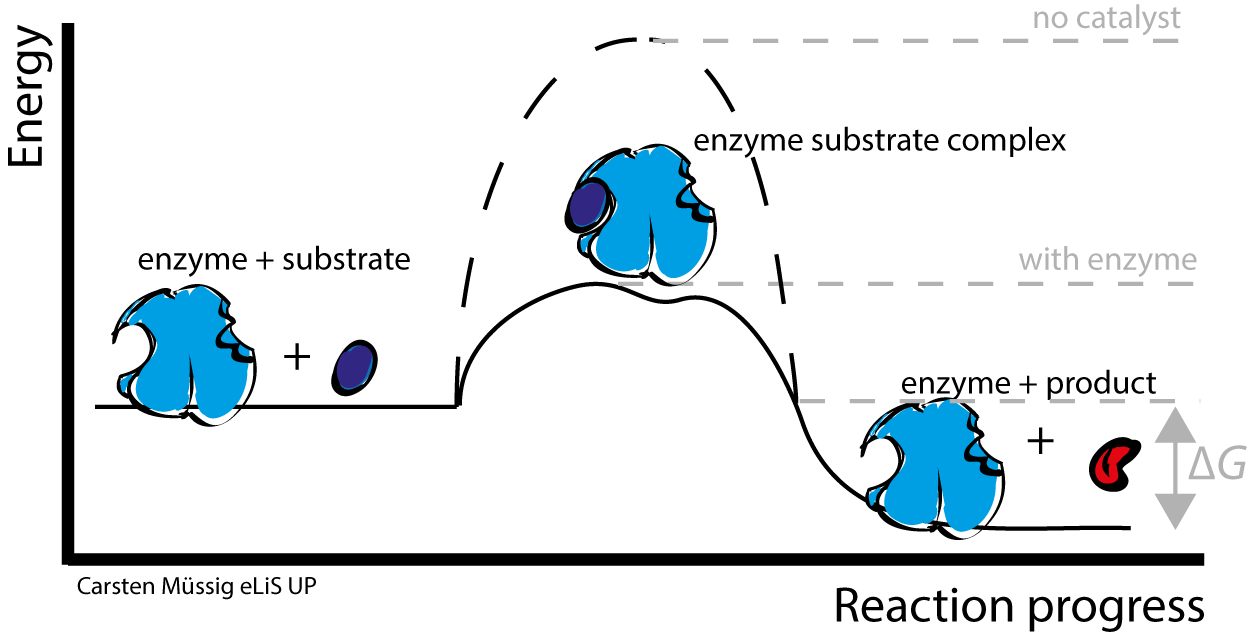
Lowering the Activation Energy
Enzymes generally lower activation energy by reducing the energy needed for reactants to come together and react. For example: Enzymes bring reactants together so they don't have to expend energy moving about until they collide at random.

31
Active Site
Substrate is a term used for a substance before a reaction. It later on can change and if so, it is then called a product. Example when eating bread, the starch (or amylum) is the substrate and if the enzyme amylase binds with it, starch will be broken down into maltose. Maltose is therefore the product.

32
Multiple Choice
33
Multiple Choice
34
Multiple Choice
35
Multiple Choice

36
Multiple Choice
37

38

39

40

41

42

43

44

45

46

47

48

49

50

51

52

53
Multiple Choice
54
Multiple Choice
55
Multiple Choice
Carbonic anhydrase is an enzyme involved in the reaction of carbon dioxide with water to form a molecule that dissolves well in the liquid part of blood. How does carbonic anhydrase affect the reaction of carbon dioxide with water?
A. by making the reaction reversible
B. by changing chemical products of the reaction
C. by increasing the time needed for the reaction to occur
D. by decreasing the amount of energy needed to complete the reaction
56
Multiple Choice
Each cell in the human body contains thousands of different enzymes responsible for regulating reactions within the cell. Why is a single enzyme unable to regulate a variety of reactions?
A. The binding of a product and an enzyme is specific.
B. The binding of a substrate and an enzyme is specific.
C. The enzyme is consumed by the products of the reactions.
D. The enzyme is consumed by the substrates of the reactions.
57
Multiple Choice
58

59

60

61

62

63

64

65

66

67
Multiple Choice
Collisions are the result of _______ of the substrate and enzyme. Successful collisions are ones in which the substrate and active site correctly ______.
random movements, align
repulsion, repel
cooling, attract
denaturation, match up
68
Multiple Choice
Low temperatures result in _______ thermal energy for the activation of an enzyme-catalyzed reaction.
insufficient
overabundant
69
Multiple Choice
At temperatures above the optimum temperature, enzyme stability will _______.
decrease
increase
70
Multiple Choice
The loss of enzyme shape is called
denaturation.
stress.
random movement.
alignment.
71
Multiple Choice
Changing the pH alters the ______ of the enzyme, which then affects protein solubility and may change the shape of the molecule.
charge
stress
speed
alignment
72
Multiple Choice
T/F: All enzymes have the same optimum pH range.
true
false
73
Multiple Choice

Use the graph to answer the following question.
The graph shows how the activity of an enzyme changes at different temperatures. Which statement best describes what happens to the enzyme when the temperature of the reaction increases to 63°C?
The enzyme is used up and the reaction stops.
The enzyme begins to decrease the rate of the reaction.
The enzyme continues to increase the rate of the reaction.
The enzyme changes shape and can no longer speed up the reaction.
74

75

76

77

78

79

80

81

82

83

84

85

86

87

88

89

90

91

92

93

94
Multiple Choice
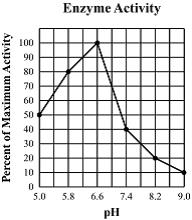
95
Multiple Choice
Allosteric inhibitors block the active site
true
false
96
Multiple Choice
97
Multiple Choice
What is an inhibitor?
the region of an enzyme that attaches to a substrate
a change in the shape of a protein
substance that can bind to an enzyme and prevent the substrate from binding
macromolecules that contain carbon, hydrogen, oxygen, and nitrogen
98
Multiple Choice
99
Multiple Choice
What are vitamins and minerals?
Part of our food, give us energy
activators
inhibitors
enzymes
100
Multiple Choice
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following?
the need for a coenzyme
allosteric inhibition
insufficient cofactors
competitive inhibition
101
Match
Match the following
Binds to active site to slow reaction
Binds at a different spot on enzyme to change shape
Binds to active site
Speeds up a chemical reaction without being used up
Part of enzyme where reactants bind to
Competitive
Allosteric
Substrate
Catalyst
Active Site
Competitive
Allosteric
Substrate
Catalyst
Active Site

Show answer
Auto Play
Slide 1 / 101
SLIDE
Similar Resources on Wayground

95 questions
DNA 1
Presentation
•
12th Grade

99 questions
Developmental Psych
Presentation
•
10th - 11th Grade

93 questions
STAAR Biology Cells
Presentation
•
9th - 12th Grade

97 questions
Adjectives andAdverbs; Comparison
Presentation
•
9th Grade - University

100 questions
Unit 3: Ecosystems
Presentation
•
9th - 12th Grade

98 questions
The Muscular System PPT
Presentation
•
9th - 12th Grade

97 questions
Day 5 - Solving & Graph Linear Inequalities
Presentation
•
9th - 12th Grade

97 questions
Holidays at the White House 2025
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Biology

20 questions
Food Chains and Food Webs
Quiz
•
7th - 12th Grade

20 questions
Genetics 2026
Quiz
•
10th Grade

10 questions
Exploring Evidence of Evolution
Interactive video
•
6th - 10th Grade

15 questions
STAAR Review Quizziz 3
Quiz
•
10th Grade

16 questions
AP Biology: Unit 2 Review (CED)
Quiz
•
9th - 12th Grade

20 questions
Codominance and Incomplete Dominance
Quiz
•
9th - 10th Grade

15 questions
Photosynthesis and Cellular Respiration
Quiz
•
10th Grade

10 questions
Evolutionary Fitness and Natural Selection Worksheet
Quiz
•
10th Grade