

PhySci Final Review 3: Thermal and Electrical
Presentation
•
Science, Physics
•
9th Grade
•
Medium
William Jared Lovering
Used 3+ times
FREE Resource
62 Slides • 59 Questions
1
PhySci Final Review 3: Thermal and Electrical
By William Lovering
2
Thermal Energy
Thermal Energy → sum of the kinetic and potential energy of all the particles in an object; thermal energy of an object increases as temperature increases
Heat → thermal energy that flows from something at a higher temperature to something at a lower temperature
3
Temperature
Average kinetic energy of all the particles in a material; measured by a thermometer in degrees (usually degrees Celsius or degrees Fahrenheit)

4

5
Thermal Energy Transfer
Conduction → Transfer of thermal energy by collisions between particles in matter. Occurs in solids, liquids, & gases. Metals are the best conductors.
Convection → Transfer of thermal energy in a fluid by the movement of warmer and cooler fluid from place to place. Occurs in liquids & gases.
Radiation → Transfer of energy by electromagnetic waves. Occurs in gases only.
6

7

8
Heat Transfer
The thermal energy exchange between two objects of different temperatures; energy moves in a predictable pattern from warmer sites to cooler sites until all sites have reached the same temperature

9
Conduction
Transfer of thermal energy that occurs in solids, liquids, and gases when two substances of different temperatures touch.

10
Convection
Heat transfer caused by the rising of hotter, less dense fluids and the falling of cooler, more dense fluids.

11
Radiation
The transfer of energy by the movement of electromagnetic waves or subatomic particles.

12
Conductor
A substance that allows the flow of electrical charge or transfers thermal energy through matter.

13
Insulator
A material that does not conduct heat or electric current

14

15

16
17

18

19

20

21
Electric Fields
An electric field is a region around a charged object where the object's electric force is exerted on other charged objects.
Because positive charges are repelled by positive charges.. and attracted to negative charges..field lines point away from positive charges and towards negative charges
22

23
How Does Charge Build Up?
Most atoms are uncharged, or neutral because the protons equal the electrons.
Uncharged objects become charged by gaining or losing electrons
Static electricity is the buildup of charges on an object
In static electricity, charges build up on an object, but they do not flow in a current
24
Charging Objects
Charges are neither created nor destroyed. This is known as the law of conservation of charge.
If one object loses electrons, another object must pick them up
There are four methods by which charges can redistribute themselves: Friction, conduction, induction, polarization
25

26

27
Charging by Friction
Charging by friction is the transfer of electrons from one uncharged object to another by rubbing objects together

28
Charging by Conduction
When a charged object touches another object, electrons can be transferred
This is the process of transferring electrons by direct contact

29
Charging by Induction and by Polarization
Electrons can react to the electric field of a charged object without touching the object itself. This is induction
Polarization occurs when electrons move only within their own atoms. In this case, the electrons create charged ends of the atoms
30
31

32
electric currents

33
What is an electric current?
The flow of electric charges through a conductor.
34
The difference between static electricity and electric currents.
Electric charges that build up on static electricity do not flow. Instead the charges jump between two objects.
electric currents is a flow of electrons like a stream of water through a conductor like copper or metal.
35
What is a direct current?
An electric current in which charges flow in one directly only.
The current produces by electrochemical cells is direct current. The chemical energy is being changed into electrical energy.
When the switch is on the electric current produced by the dry cells keeps the light bulb on

36
Alternating current:
type of current that does not flow in one direction only.
constantly changes direction at a regular rate.
can be transported more easily over long distance
37
What is a Circuit Made of?
Objects that use electricity contain circuits
All electric circuits have these basic features: devices that run on electrical energy, sources of electrical energy, and conducting wires
38

39
Circuit Symbols
Battery
Bulb
Switch
Resistor

40
Circuit Energy Sources
Power plants and batteries are examples of energy sources
These supply the voltage that causes current to flow
When the energy source is a battery, current flows from the positive end to the negative end
41

42
Types of Circuits
Series Circuits
Parallel circuits

43
Series circuits
All elements in the circuit are in one loop
Every electron flow through every element
If one bulb burned out, the rest would also go out

44
Parallel circuits
There are several pathways or loops through the circuit
Electrons flow through just one of those pathways
If one bulb burned out, the rest of the bulbs would still glow

45
Ohm's Law
Georg Ohm found that conductors and most other devices have a constant resistance regardless of the applied voltage
Ohm's law says that resistance in a circuit is equal to voltage divided by current
Resistance = voltage / current
Units for resistance (Ohms); voltage (volts - V), Current is amps (A)
46
Volts, Amps, and Resistance
V = I * R
Voltage = current multiplied by the resistance
Series:
R = R1+R2+R3+R4 ...
Parallel:
R = 1/R1 + 1/R2 + 1/R3 ...
47

48

49

50

51
52

53

54

55

56
Voltage in Series Circuits
The voltage drops each time it passes through a resistor (like a lightbulb)
Total voltage equals the sum of all the voltage drops.
V total = V1 + V2 + V3 + V4 ...
Total Voltage also equals the voltages of all batteries placed in series

57
Voltage in Parallel
The voltage is the same no matter which branch of the parallel circuit you're on.
VTotal = V1 = V2 = V3 = ...
Like a platform with several water slides (paths down), no matter which one you choose, everyone ends back up at the bottom.

58
Current in Series
The same current is shared by the whole circuit, since there's only one path for the electrons to go, so all the electrons go that way
ITotal = I1 = I2 = I3 = I4 = I5 = ...

59
Current in Parallel
In parallel, the electrons will split and go down different paths, so only some will go down any given path
This decreases the current through each path.
Current will be faster where there's less resistance.
Electrons regroup at the end, though, so... IT = I1 + I2 + I3 + ...

60
Resistance in Series
The more resistors you add, the more resistance overall.
Adding more lightbulbs to this circuit would make each bulb dimmer and dimmer.
RTotal = R1 + R2 + R3 + R4 ...

61
Resistance in Parallel
Total resistance decreases the more resistors you add in parallel
It's like on Black Friday when the checkout line gets long and they add another checkout. There won't be as many people going through each line, but each person won't have the line holding them back as much.
1/RT = 1/R1 + 1/R2 + 1/R3 ...
62
63
Multiple Choice
64
Multiple Choice
65
Multiple Choice
66
Multiple Choice
67
Multiple Choice
68
Multiple Choice
What is resistance measured in?
Ω (Ohms)
V (Volts)
A (Amps)
W (Watts)
69
Multiple Choice
70
Multiple Choice
71
Multiple Choice
72
Multiple Choice
73
Multiple Choice
If you added a can of Coke (20oC) to a fridge (5oC), at what temperature would heat stop moving?
when the coke is at 10 degrees and the fridge is at 5 degrees.
When the coke is at at 6 degrees and the fridge is at 6 degrees
When the coke is at at 5 degrees and the fridge is at 15 degrees
When the coke is at at 20 degrees and the fridge is at 5 degrees
74
Multiple Select
Electricity happens with the flow of
electrons.
water
sand
75
Multiple Choice
76
Multiple Choice
77
Multiple Choice

78
Multiple Choice

What happens to the temperature if I add ice cubes to a glass of soda
The heat transfers from the coke to the ice
The heat transfers from the ice to the coke
79
Multiple Choice
80
Multiple Choice
81
Multiple Choice

82
Multiple Choice
83
Multiple Choice
84
Multiple Choice
Two objects have different temperatures. Object A has a temperature of 42 degrees and Object B is 37 degrees. Which direction should the energy transfer between Objects A and B?
From Object A to Object B
From Object B to Object A
Energy will not transfer
Energy will transfer both directions
85
Multiple Choice
Ohms
Watts
86
Multiple Choice
87
Multiple Choice
88
Multiple Choice
89
Multiple Choice
90
Multiple Choice
91
Multiple Choice
92
Multiple Choice
93
Multiple Choice
94
Multiple Choice
95
Multiple Choice
96
Multiple Choice
The accumulation of excess electric charge on an object is called
Static electricity
Electric discharge
Resistance
Current electricity
97
Multiple Choice

98
Multiple Choice
This type of thermal energy transfer can take place in fluids (liquids & gases). Rising of warmer fluid and sinking of cooler fluid forms a current.
conduction
convection
radiation
99
Multiple Choice
If you added ice (0oC) to a glass of water (15oC) how would the heat move between the ice & water?
from the ice to the water
from the water to the ice
100
Multiple Choice
101
Multiple Choice
Specific heat is
The amount of thermal energy needed to raise the temperature of 1 kg of a substance by 1 degree celsius
The amount of kinetic energy needed to raise the temperature of 4 kg of a substance by 3 degrees Fahrenheit
The amount of heat required to raise 10 mg of a substance by 40 degrees celsius
The amount of temperature to raise 1 kg of a substance by 1 degree Kelvin
102
Multiple Choice
If a substance needed a lot of energy to raise the temperature by 1 degree Celsius, its specific heat is
Non-existant
Low
Undeterminable
High
103
Multiple Choice

104
Multiple Choice
105
Multiple Choice

106
Multiple Choice

107
Multiple Choice
108
Multiple Choice
109
Multiple Choice
110
Multiple Choice
111
Multiple Choice
What letter is used to represent specific heat capacity?
H
P
C
T
112
Multiple Choice
113
Multiple Choice
The specific heat of platinum is 0.133 J/g°C. How much heat(Q) is released when a 10 g piece of platinum cools from 100°C to 50°C?
66.5 J
665 J
0.0266 J
0.665 J
114
Multiple Choice
Determine the heat energy needed to heat the water in the pot to boiling.
115
Multiple Choice
116
Multiple Choice

117
Multiple Choice
The specific heat(c) of copper is 0.39 J/g °C. What is the temperature change(∆t) when 100 Joules of heat(Q) is added to 20 grams?
12.82 °C
24.12°C
1.95 °C
5128 °C
118
Multiple Choice
The condition where two substances in physical contact with each other exchange no heat energy. They are said to be the same temperature.
Thermal Energy
Cooler to Warmer
Energy Transfer
Thermal Equilibrium
119
Multiple Choice
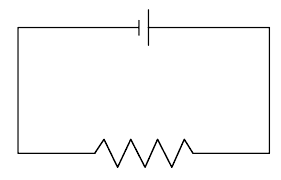
120
Multiple Choice
121
Multiple Choice
PhySci Final Review 3: Thermal and Electrical
By William Lovering
Show answer
Auto Play
Slide 1 / 121
SLIDE
Similar Resources on Wayground

112 questions
contries all
Presentation
•
8th Grade
![G7- Final Review Chapter 2 [Term1]](https://cf.quizizz.com/image/image-loader.svg)
117 questions
G7- Final Review Chapter 2 [Term1]
Presentation
•
7th Grade

115 questions
AC review
Presentation
•
8th Grade

117 questions
Semester 1 Exam Review
Presentation
•
10th Grade

115 questions
Unit 5 Topic 11-12 Q & A + Unit Revie
Presentation
•
9th Grade

120 questions
Trig lesson
Presentation
•
9th Grade

121 questions
NMMS 2021 WRITING CAMP - The Conventions Pyramid!
Presentation
•
8th Grade

117 questions
F5 Life's Great Mysteries L13 Review
Presentation
•
10th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

9 questions
EOC Domain 4: Evolution
Presentation
•
9th - 10th Grade

14 questions
EOC Domain 2: Genetics
Presentation
•
9th - 10th Grade

10 questions
EOC Domain 3: Taxonomy
Presentation
•
9th - 10th Grade

12 questions
EOC Domain 5: Ecology
Presentation
•
9th - 10th Grade