

Semester 1 Exam Review
Presentation
•
Science
•
10th Grade
•
Medium
John Oglesby
Used 1+ times
FREE Resource
48 Slides • 69 Questions
1
Review for Sem. 1 Exam
Some text here about the topic of discussion
2
What's on the exam?
Metric System - 5 Questions (3 pts each)
Energy - 5 Questions (3 pts each)
Simple Machines - 5 Questions (3 pts each)
Waves - 5 Questions (4 pts each)
Thermal Energy - 5 Questions (3 pts each)
Essay Questions - 2 Questions (5 pts each)
27 Questions Total - 100 pts
3
Metric System Review
Some text here about the topic of discussion
4
Base Units
Volume = Liters = L
Mass = Grams = g
Length = meters = m

5
Prefixes
The Metric system is based on multiples of tens.

6
Remember
“King Henry Doesn’t (Usually) Drink Chocolate Milk”

7
Multiple Select
Which units below are NOT part of the metric system? Check all that apply.
Inches
Feet
Meters
Grams
8
Multiple Select
Which units are the BASE units in the metric system? Check all that apply.
Meters
Liters
Grams
Inches
Miles
9
Multiple Choice
Liter
Meter
gram
inch
10
Multiple Choice
Gram
Liter
Pound
Meter
11
Multiple Choice
Gram
Meter
Liter
Ounce
12
Converting Units
Changing a Metric Units to another Metric Unit
Moving BIG unit to small Multiply
Moving small to BIG Divide

13
Multiple Choice
42 L = ? mL
4,200
.0042
4.20
42,000
14
Multiple Choice
3 cm = ? mm
30
300
3000
0.3
15
Multiple Choice
75.2 cm = ? m
0.752
7.52
75.2
752
16
Multiple Choice
Convert:
3 kg = ? g
3
30
300
3,000
17
Multiple Choice
6,000 mL = ? L
60
600
6
6,000
18
Multiple Choice
What is 3 litres in ml?
30
300
3000
0.3
19
Multiple Choice
What is 0.9km in meters?
0.9
9
90
900
20
Energy Review
Some text here about the topic of discussion
21
Potential Energy
The energy you possess by virtue of height above the ground
GPE = mgh
22
Multiple Choice
what is acceleration due to gravity for earth?
9.81m/s2
1.34m/s2
10m/s2
8.91m/s2
23
Multiple Choice
what 3 factors effect gravitational potential energy
- Mass (m)
-Weight (N)
- speed (m/s)
- Mass (N)
- Height (km)
- Acceleration due to gravity (g)
- Mass (kg)
- Height (m)
- Acceleration due to gravity (g)
- Mass
- Height
- Acceleration due to gravity
24
Multiple Choice
An object with a mass of 10 kg is at a point 20 m above the ground. What is the gravitational potential energy of the object?
200 J
1960 J
2 J
1000 J
25
Multiple Choice
__________ energy is stored energy.
Potential
Kinetic
Force
Gravity
26
Multiple Choice
A 20 kg box is lifted 6 m above the ground. How much GPE does it have?
120 J
200 J
1176 J
60 J
27
Multiple Choice
Which of the following statements about a skier skiing down a steep slope is true?
His kinetic energy decreases and his potential energy increases
His potential energy decreases and his kinetic energy increases
His chemical energy increases
Both his potential and kinetic energy increases
28
Multiple Choice
Which of the following statements is implied by the law of conservation of energy?
Energy is static and unchanging.
Energy only ever transforms or transfers.
Energy will eventually run out.
None of these.
29
Multiple Choice

A 6kg cat misjudges a jump and falls 5 meters. How much gravitational potential energy does the cat have? (The cat is fine by the way.)
294 J
180000 J
1225 J
3920000 J
30
Kinetic Energy
The energy of motion
KE = 1/2 mv2
An object has KE whenever it is moving
Some text here about the topic of discussion
31
Multiple Choice
A scientific law stating that energy cannot be created or destroyed.
The Law of Potential Energy
The Law of Kinetic Energy
The Law of Conservation of Energy
32
Multiple Choice
If a 4 kg cat is running at 3 m/s what is its kinetic energy?
18 J
18
12
12 J
33
Multiple Choice
The faster an object moves, the ________ kinetic energy it has.
more
less
34
Multiple Choice
Which has a greater impact on KE?
mass
speed
35
Multiple Choice

A 50kg (100lb) person is running at 7m/s (about 16 mph). How much kinetic energy do they have?
1225 J
180000 J
294 J
3920000 J
36
Multiple Choice
What is the unit of measure
ment for KE
Newton
Watt
Joule
37
Multiple Choice

A 1 kg bird flies south at 4 m/s. What is its KE?
2 m/s
8 J
2 J
4 J
38
Multiple Choice
According to the law of conservation of energy, the total amount of energy in the universe ____.
remains constant
changes constantly
increases
decreases
39
Simple Machines - Review
Some text here about the topic of discussion
40
SIMPLE MACHINES
a device that increases or changes the direction of a force
41
Levers
Levers are probably the most common simple machine.
The fulcrum is the point in which a lever rests (pivots);
The load is what you are trying to move;
Effort is the force you apply.

42
First Class Levers
The fulcrum is between the effort and the load.

43
Second Class Levers
The load is between the effort and the fulcrum.

44
Third Class Levers
The effort is between the fulcrum and the load.

45
Leavers and Loads
A lever might be able to...
decrease the effort required to move a load;
change the direction that a load moves;
increase the distance that load moves;
Increase the speed with which a load moves.

46
Multiple Choice
FOOT, EFFORT, REPORT
FULCRUM, STRUCTURE, EFFORT
FULCRUM, EFFORT, LOAD
47
Multiple Choice

TRUE
FALSE
48
Multiple Choice

Which part is the 'load'?
Arrow
Yellow box
Blue triangle
49
Fill in the Blanks
Type answer...
50
Multiple Choice

At what distance should a 2N load placed from the pivot so that the see saw won't move?
0.2m
0.5m
0.45m
0.42m
51
Multiple Choice
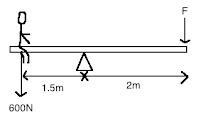
A boy of weight 600N sits on the see-saw as shown at a distance of 1.5m from the pivot. What is the force F required at the other end to balance the see-saw?
450 N
350 N
4.5 N
200 N
52
Multiple Choice

A ramp is an example of a(n) _______.
inclined plane
fulcrum
force
lever
53
Multiple Choice
Newtons
Joules
Watts
Meters
54
Multiple Choice

Pulley
Wheel and Axle
Lever
Wedge
55
Multiple Choice
wheel and axle
lever
inclined plane
pulley
56
Mechanical advantage of simple Machines
By Ms. Taylor
57
Mechanical Advantage
What is it?
A Ratio of Forces
MA = Output Force / Input Force
A machine that has a MA above 1, means that the force out is greater than the force you put in.
Some text here about the topic of discussion
58
Mechanical Advantage, MA
How to interpret MA
A machine that gives a MA 1 only causes a change in direction.
A machine that gives a MA less than 1 usually increases the distance over which the work is done.
A machine that gives a MA greater than 1 usually increases the output force (less force needed)
Some text here about the topic of discussion
59
Multiple Choice
The amount that input is magnified by a simple machine is known as the :
mechanical advantage
output force
resistance
none of these
60
Multiple Choice
The Mechanical Advantage (MA) of a simple machine makes work
Easier
Harder
No difference to the work done
None Of the above
61
Multiple Choice
What is effort (input) force?
the force one exerts on a machine
the force which an effort force must overcome in order to do work on an object via a simple machine.
the force the machine exerts on a object
the output work times the input distance
62

63

Straight, slanted surface
Does not decrease amount of work needed but increases the distance.
Same work is done but over a
greater distance.
longer ramp, more MA
MA=length/height of ramp
Inclined plane
64
Multiple Select
Is a staircase an inclined plane?
yes
no
65
Examples of a 1st Class Lever


66
Examples of a 2nd Class Lever

67
Examples of a 3rd Class Lever


68
Waves - Review
Some text here about the topic of discussion
69

70
Multiple Choice
Longitudinal Compression waves move in the direction of the wave.(They are parallel to the direction of the wave)
True
False
71
Multiple Choice
A wave that has the vibration perpendicular to the direction of the wave is called:
Longitudnal Wave
Transverse Wave
72

73

74
Multiple Choice

What is B?
crest
trough
rarefaction
compression
75
Multiple Choice

What is A?
crest
trough
rarefaction
compression
76

77
Multiple Choice

What does point C represent?
Crest
Trough
Amplitude
Wavelength
78
Multiple Select

The wavelength can be measured from ( check all that apply)
Distance from Crest to Crest
Distance from Crest to Trough
Distance from Trough toTrough
Distance from Crest to midpoint
79
Multiple Choice

What does point A represent?
Crest
Trough
Amplitude
Wavelength
80
Multiple Choice

What does the arrow in the Wave represent?
Crest
Trough
Amplitude
Wavelength
81
Multiple Choice

If the amplitude increases, the energy of the wave:
Increases
Decreases
Stays the same
82
Multiple Choice
vibration
compression
medium
crest
83
Multiple Choice
The frequency of a wave is measured in
meters
hertz
periods
eras
meters per second
84
Multiple Choice
The distance between 2 corresponding parts of a wave (example- crest to crest) is its
trough
frequency
amplitude
wavelength
85
Multiple Choice
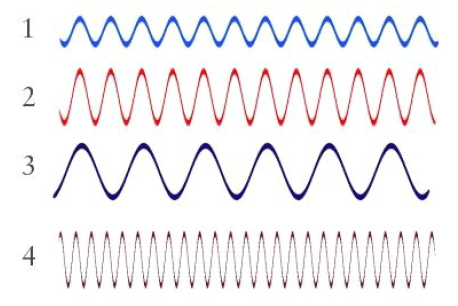
Which wave in the diagram has the greatest frequency?
1
2
3
4
86
Multiple Choice
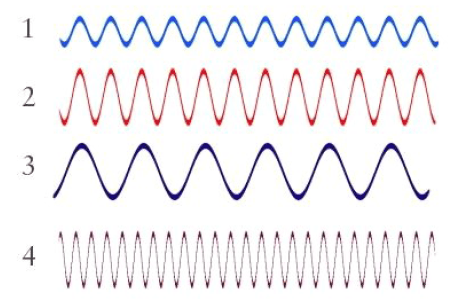
Which wave in the diagram has the longest wavelength?
1
2
3
4
87
Multiple Choice

You can't hear sounds in outer space because there is no
medium
light
energy
heat
88
Thermal Energy
Review
Some text here about the topic of discussion
89
Thermal Energy
The flow of energy from hot to cold.
The atoms(molecules) of something hot are moving faster than those of something cold.
Temperature is the measure of the speed of those particles and is equal to the avg kinetic energy of all the particles in a sample.
Subject | Subject
Some text here about the topic of discussion
90
Thermal Energy Transfer
Conduction → Transfer of thermal energy by collisions between particles in matter. Occurs in solids, liquids, & gases. Metals are the best conductors.
Convection → Transfer of thermal energy in a fluid by the movement of warmer and cooler fluid from place to place. Occurs in liquids & gases.
Radiation → Transfer of energy by electromagnetic waves. Occurs in gases only.
91
Conduction
Transfer of thermal energy that occurs in solids, liquids, and gases when two substances of different temperatures touch.

92
Convection
Heat transfer caused by the rising of hotter, less dense fluids and the falling of cooler, more dense fluids.

93
Radiation
The transfer of energy by the movement of electromagnetic waves or subatomic particles.

94
Conductor
A substance that allows the flow of electrical charge or transfers thermal energy through matter.

95
Insulator
A material that does not conduct heat or electric current

96
Multiple Choice
Heat is the transfer of _____ from one object to another due to a difference in temperature.
Time
Force
Energy
Velocity
97
Multiple Choice

The man's hand will immediately receive heat from the metal as the candle gradually warms it. That is called____?
Conduction
Convection
Radiation
98
Multiple Choice

"Metals" are the best____?
Insulators of heat
Conductors of heat
Conductors of light
Insulators of electricity
99
Multiple Choice
How does heat move?
from a warmer to a cooler object
from a cooler to a warmer object
toward a hot object
away from a cold object
100
Multiple Choice
Energy transmitted by electromagnetic waves
conduction
convection
radiation
101
Multiple Choice

When you touch hot sand, heat is transferred by _________ to your skin.
conduction
convection
radiation
102
Multiple Choice

conduction
convection
radiation
103
So, What is TEMPERATURE?

104
Temperature - The average kinetic energy of all the molecules of the sample

105
What are the temperature scales based on?
Celcius & Fahrenheit are based on the freezing pt and boiling point of water.
Kelvin is based on absolute zero. At absolute zero atoms have zero kinetic energy.
106
°C = 5∕9 (°F - 32)
°F = 9∕5 °C + 32
Celcius / Fahrenheit Conversion
107
Convert 40 deg C to F
°F = 9/5 °C + 32
= 9(40)/5 + 32
= 104 F
108
Fill in the Blanks
Type answer...
109
Let's convert 68 deg F to Celcius
⁰C = 5/9 (⁰F - 32)
⁰C = 5/9 (68 - 32)
⁰C = 5/9 (36)
⁰C = 20⁰F
Remember to do what is in the parenthases first
110

Temperature scale conversion
To convert a temperature from Celsius to Kelvin, just add 273
to the Celsius temperature.
For example, 10 °C equals 283 Kelvin.
111

C to F
Converting between Celsius and Fahrenheit is more
complicated.
Celsius → Fahrenheit: (°C × 1.8) + 32 = °F
Fahrenheit → Celsius: (°F - 32) ÷ 1.8 = °C
112
Multiple Choice
80 C = F
176
44.4
12.4
144
113
Multiple Choice
76 F = C
44
24.4
12
349
114

Absolute Zero
The lowest temperature that is theoretically possible, at which
the motion of particles that constitutes heat would be
minimal.
It is zero on the Kelvin scale, equivalent to –273.15°C or
–459.67°F.
115
Multiple Choice
Anna pours herself some room-temperature soda from a bottle and adds four ice cubes. In a few minutes the ice cubes are smaller and the soda is much colder. Which of the following best explains the change in temperature of the soda?
Heat flowed from the soda to the ice cube and caused it to partially melt.
Coldness flowed from the ice cube to the soda, making the soda colder.
The cold water from the melting ice went into the soda, replacing the warmer water that was in the soda.
The water from the melting ice makes the soda more dilute, and the lower concentration makes the temperature lower.
116
Multiple Choice
1. David drinks a glass of water in his kitchen. The water is at room temperature, and David decides to make it colder by adding ice. Which of the following describes why the ice cubes will melt?
Liquid water is reacting with solid water to absorb
energy.
Water is evaporating from the surface of the water.
Slow-moving molecules move out of the ice.
Heat from the water moves into the ice.
117
Temperature Conversions
F⁰ to C⁰
C = 5/9(F⁰-32)
Ex. 75⁰ F⁰ is what in C
C ⁰= 5/9(75-32)
C⁰ = 24⁰
note order of operation ( parenthesis first!)
Review for Sem. 1 Exam
Some text here about the topic of discussion
Show answer
Auto Play
Slide 1 / 117
SLIDE
Similar Resources on Wayground

105 questions
F4 Core 3 Unit 3.2
Presentation
•
10th Grade

111 questions
ULANGKAJI MATEMATIK FORM 1 (Cikgu Afiqah)
Presentation
•
9th - 12th Grade

110 questions
Lesson 5 & 6 - Taxonomy and Dichotomous Keys
Presentation
•
9th - 12th Grade

106 questions
OSMANLI DÜNYA GÜCÜ GENEL TEKRAR
Presentation
•
9th Grade

108 questions
Chapter 14: Interactions in the Ecosystem
Presentation
•
10th Grade

110 questions
Systems TEST
Presentation
•
11th Grade

116 questions
Sequence Lesson
Presentation
•
9th Grade

116 questions
Organism Reproductive System
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

11 questions
EOC Domain 1: Cells
Presentation
•
9th - 10th Grade

14 questions
EOC Domain 2: Genetics
Presentation
•
9th - 10th Grade

10 questions
Exploring the Human Respiratory System
Interactive video
•
6th - 10th Grade

10 questions
Exploring Newton's Laws of Motion
Interactive video
•
6th - 10th Grade

10 questions
Exploring the Formation of the Solar System
Interactive video
•
6th - 10th Grade