

PS.Chem 2.1 Coloring the PT's Groups
Presentation
•
Science
•
9th - 12th Grade
•
Medium
+11
Standards-aligned
Julie Combs
Used 2+ times
FREE Resource
23 Slides • 31 Questions
1

Coloring & Labeling the Periodic Table
2
SPDF Blocks
Please use your new periodic table to:
Outline
S block
P block
D block
F block
Re-number the "floors"

Please do NOT color or mark on the arrows!
3

Questions Ahead...
4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
7
Multiple Choice
Which of the following elements is NOT a metalloid?
CARBON (C)
BORON (B)
ARSENIC (As)
8
Multiple Choice
How many valence electrons do metalloids have?
1–2
2–4
3–6
9
Multiple Choice
Which element is NOT a metalloid?
ANTIMONY (Sb)
TELLURIUM (Te)
ALUMINUM (Al)
10
Multiple Choice
Which of the following is a nonmetal?
CALCIUM (Ca)
SULFUR (S)
POTASSIUM (K)
11
12


Make sure you are taking notes in your packet!
Please follow instructions carefully.
You will go between notes and coloring on your Periodic Table.
13
Alkali Metals
Located in Group 1 (except Hydrogen)
Extremely reactive--they want to lose 1 electron to become "noble gas" like

14
15
The color does not matter, however make sure you ONLY color the Alkali Metals. Make sure to fill in your color key!
Color the Alkali Metals

16
Alkaline Earth Metals
Located in Group 2
Also very reactive
Both Group 1 & 2 occur naturally as compounds
not individual elements
Needs to lose 2 electrons to become "noble gas like"

17
18
The color does not matter, however make sure you ONLY color the Alkaline Earth Metals. DO NOT color inside the arrow! Make sure to fill in your color key!
Color the Alkaline Earth Metals

19
Halogens
Located in Group 7
Very active nonmetals
Want to gain 1 electron to become more like a noble gas

20
21
The color does not matter, however make sure you ONLY color the Halogens. Make sure to fill in your color key!
Color the Halogens

22
Noble Gases
Group 8
Sometimes called "inter gases" since generally they NEVER react
Mainly, but not always true, Kr & Xe will sometimes react
Have a full valence shell (8 electrons
Look at you, taking notes!

23
The color does not matter, however make sure you ONLY color the Noble Gases. Make sure to fill in your color key!
Color the Noble Gases

24
Located in the center of the Periodic Table
10 elements wide (d block)
Semi-reactive, very valuable, crucial to many life processes
Transition Metals

The Yellow color only!
25
26
The color does not matter, however make sure you ONLY color the Transition Metals. DO NOT color inside the arrow! Make sure to fill in your color key!
Color the Transition Metals

27
Located at the bottom of the Periodic Table
14 elements wide (f block)
Some are radioactive, but NOT all
Lanthanides = Period 6(4f)
Actinides = Period 7 (5f)
Lanthanides & Actinides


28
The color does not matter, however make sure you ONLY color the Lanthanides & Actinides. Make sure to fill in your color key!
Color the Lanthanides & Actinides

29

Knowledge check ahead...
30
Multiple Choice
Which element is similar to Li?
Be
K
Fe
I
31
Multiple Choice
Which element is similar to Mg?
Ca
B
O
Cl
32
Multiple Choice
Which element is similar to B?
C
Al
Cl
Na
33
Multiple Choice
Which element is similar to He?
H
Ar
Au
Hg
34
Multiple Choice

35
Multiple Choice
36
Multiple Choice
37
Multiple Choice
What is the name of group number 1?
halogens
noble gases
alkali metals
alkaline earth metals
38
Multiple Choice
What is the name of group number 2?
transition metals
halogens
metaloids
alkaline earth metals
39
Multiple Choice
What group number are the halogens found?
18 (8A)
1
2
17 (7A)
40
Multiple Choice
Periodic law states that the elements are arranged according to their atomic ________ so that elements with similar chemical properties are in the same ________ and properties repeat periodically.
numbers, rows
masses, rows
masses, column
numbers, column
41
Multiple Choice
42
Multiple Choice
Name this group: These metals contain familiar metals such as gold, iron, and copper.
43
Multiple Choice

Which element is found in group 2, period 6?
Oxygen (O)
Barium (Ba)
Selenium (Se)
Carbon (C)
44
Multiple Choice
Which halogen is found in period 4?
Krypton (Kr)
Xenon (Xe)
Bromine (Br)
Iodine (I)
45
Multiple Choice
Which element has similar chemical properties as sodium (Na) but has 7 energy levels?
Francium (Fr)
Cesium (Cs)
Rubidium (Rb)
Potassium (K)
46
Multiple Choice
47
Multiple Choice
48
Multiple Choice
How many valence electrons does Phosphorus have?
31
5
15
4
49
Multiple Choice
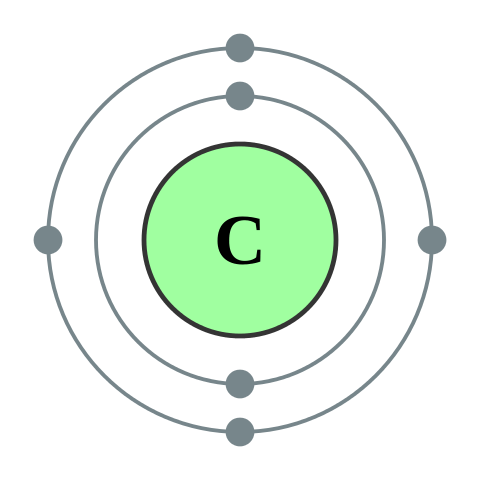
How many valence electrons does carbon have?
4
5
6
7
50
Multiple Choice

How many valence electrons?
51
Multiple Choice
52
Multiple Choice
53
Multiple Choice
How many valence electrons are in the following configuration?
1s22s22p63s2
6
8
2
12
54

Does your PT look like this?

Coloring & Labeling the Periodic Table
Show answer
Auto Play
Slide 1 / 54
SLIDE
Similar Resources on Wayground

46 questions
Biomolecule terms - photosynthesis and respiration
Presentation
•
9th - 12th Grade

49 questions
Pedigrees
Presentation
•
9th - 12th Grade

47 questions
LAB SAFETY - SPRING 2022
Presentation
•
9th - 12th Grade

48 questions
Biogeochemical Cycles: Nutrient Cycles
Presentation
•
9th - 12th Grade

44 questions
Properties of Water
Presentation
•
9th - 12th Grade

50 questions
Electron Configuration Notes
Presentation
•
9th - 12th Grade

48 questions
Particle Theory and Bonding
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

9 questions
EOC Domain 4: Evolution
Presentation
•
9th - 10th Grade

14 questions
EOC Domain 2: Genetics
Presentation
•
9th - 10th Grade

10 questions
EOC Domain 3: Taxonomy
Presentation
•
9th - 10th Grade