15 Hỏi
11th - Uni

20 Hỏi
9th - 12th
15 Hỏi
11th - Uni
15 Hỏi
11th - Uni
15 Hỏi
11th - Uni

12 Hỏi
9th - 12th
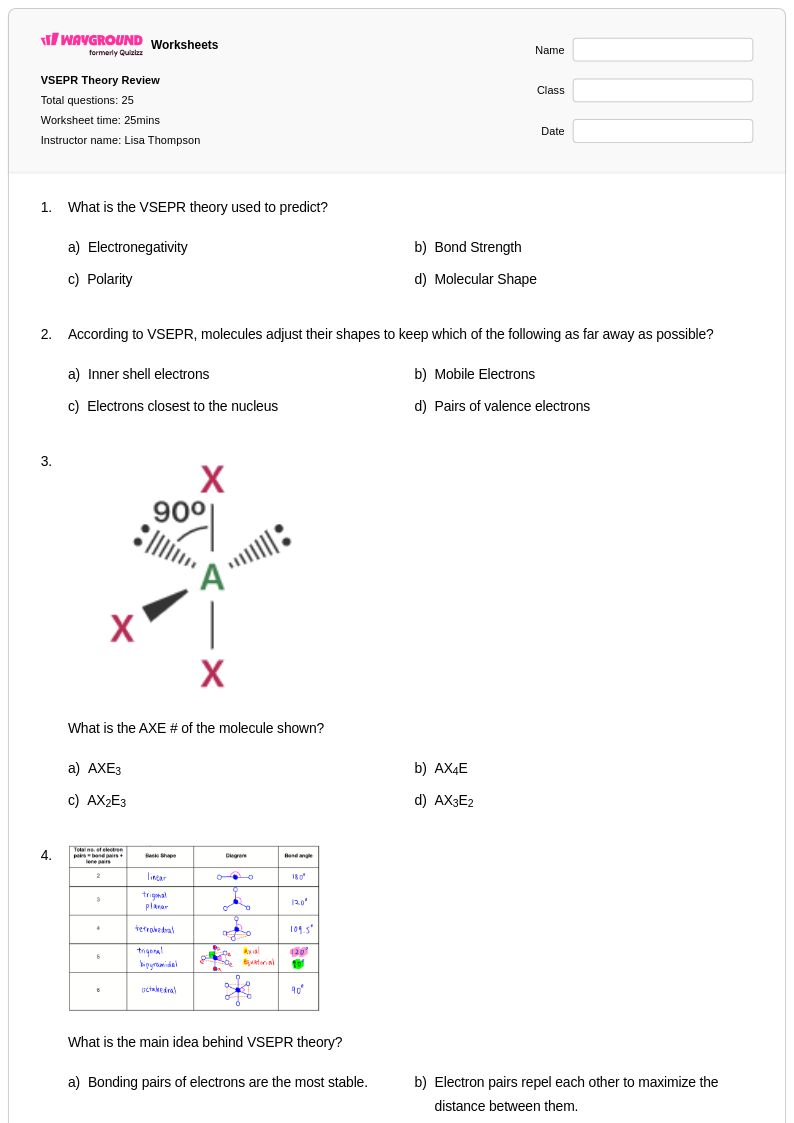
25 Hỏi
11th - Uni
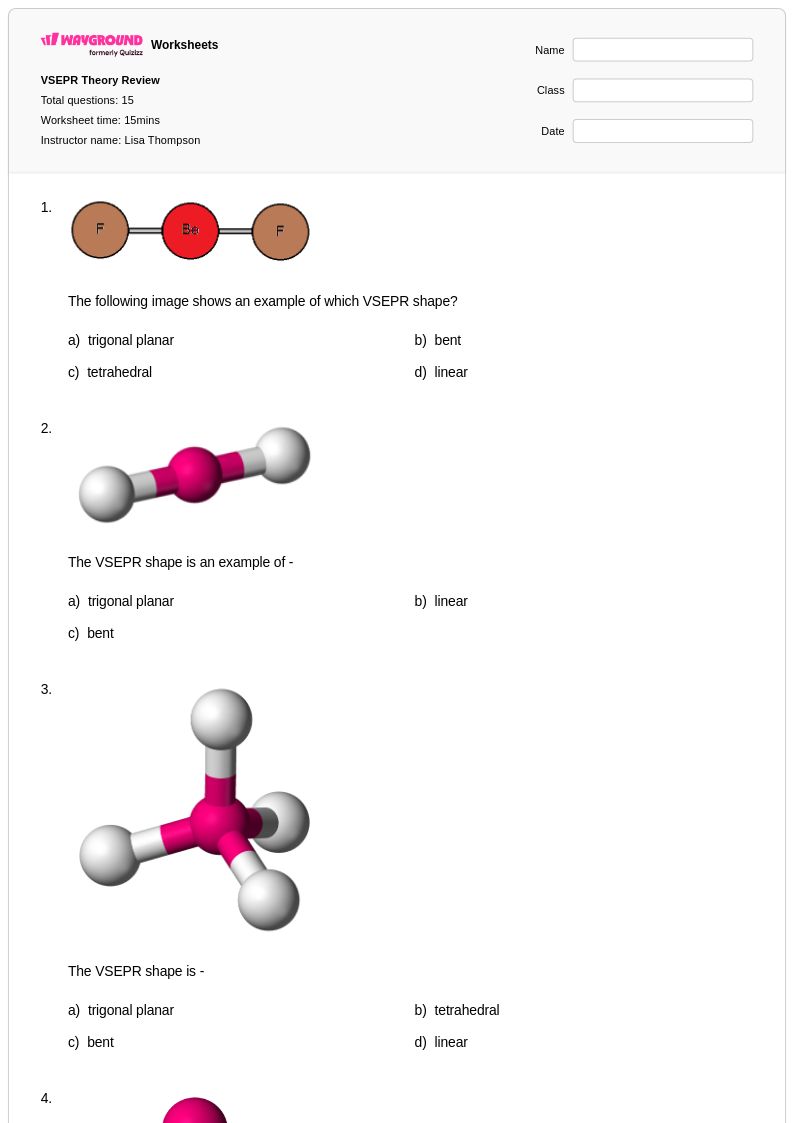
15 Hỏi
11th - Uni

25 Hỏi
11th - Uni

15 Hỏi
10th - Uni
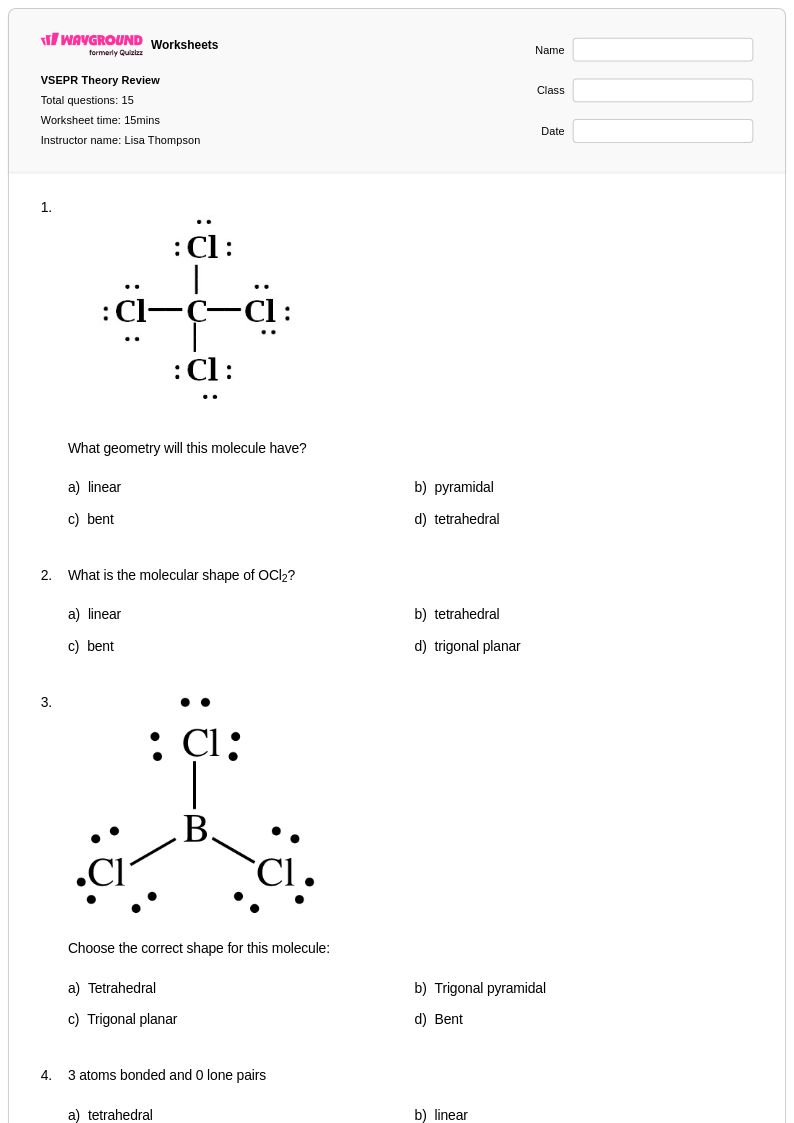
15 Hỏi
11th - Uni
11 Hỏi
11th

25 Hỏi
10th - Uni

10 Hỏi
9th - 12th
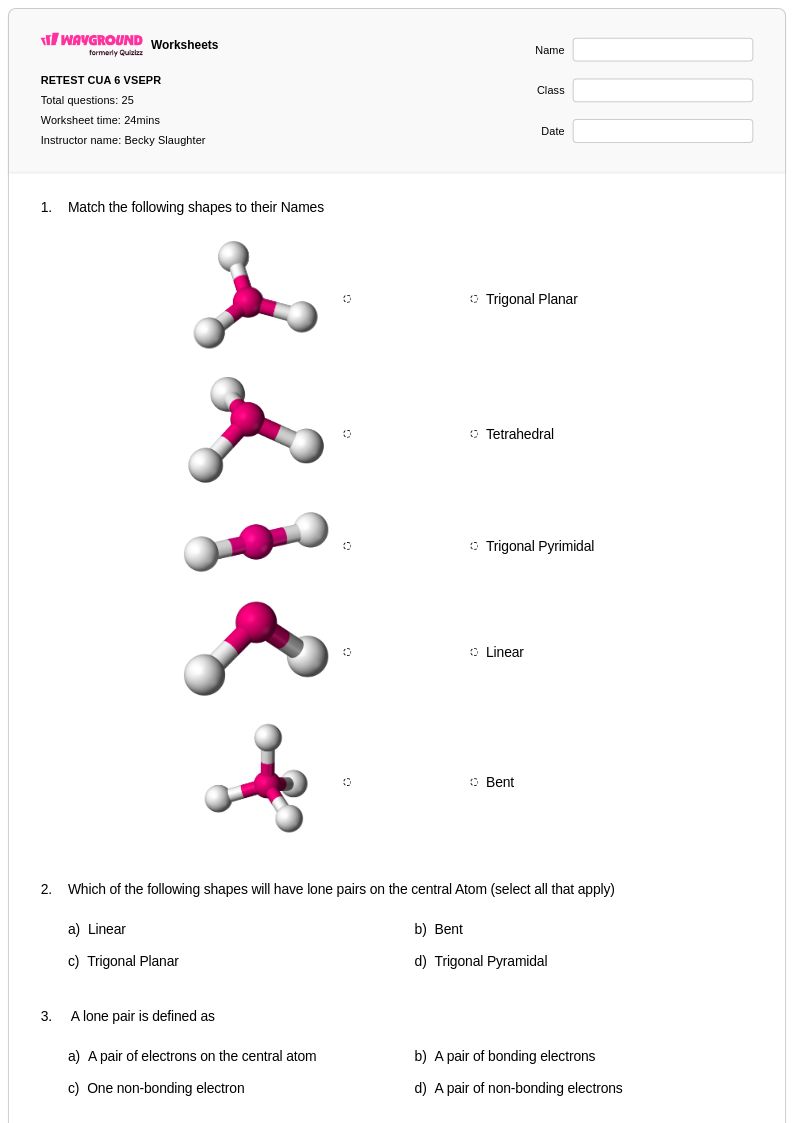
25 Hỏi
9th - 12th

25 Hỏi
11th

15 Hỏi
10th - 11th

25 Hỏi
11th - Uni
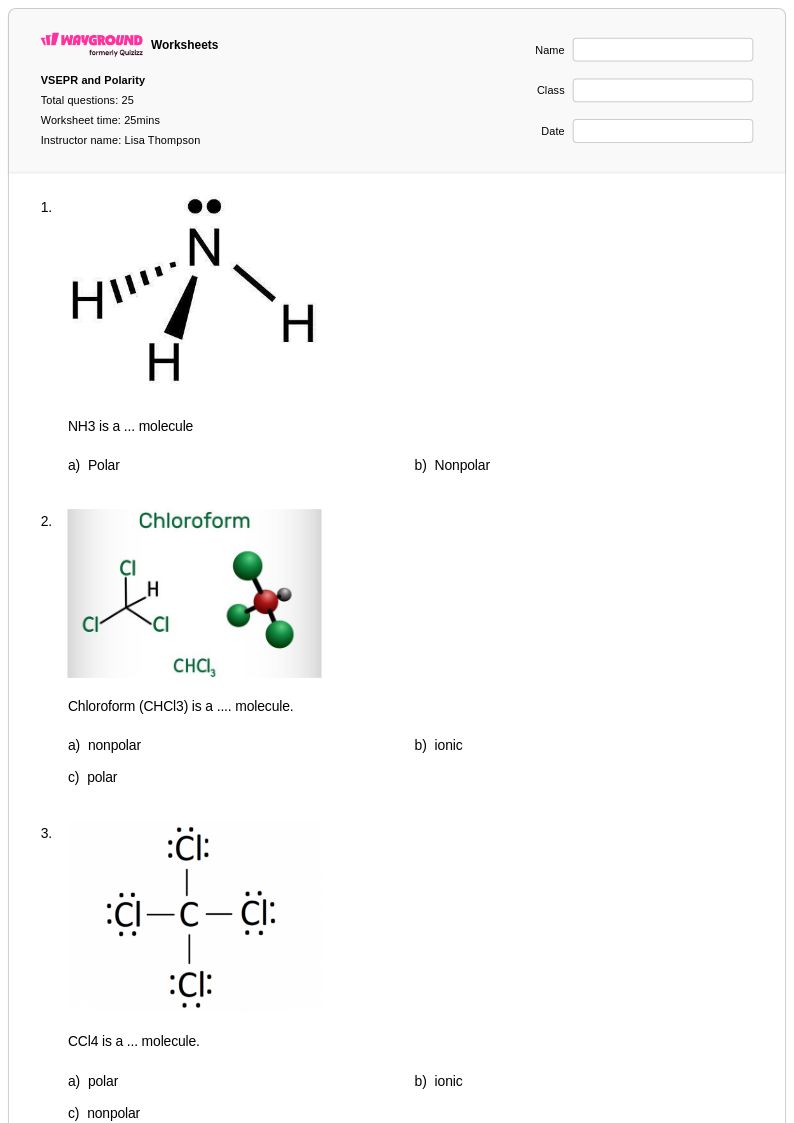
25 Hỏi
11th - Uni

25 Hỏi
11th

35 Hỏi
11th - Uni
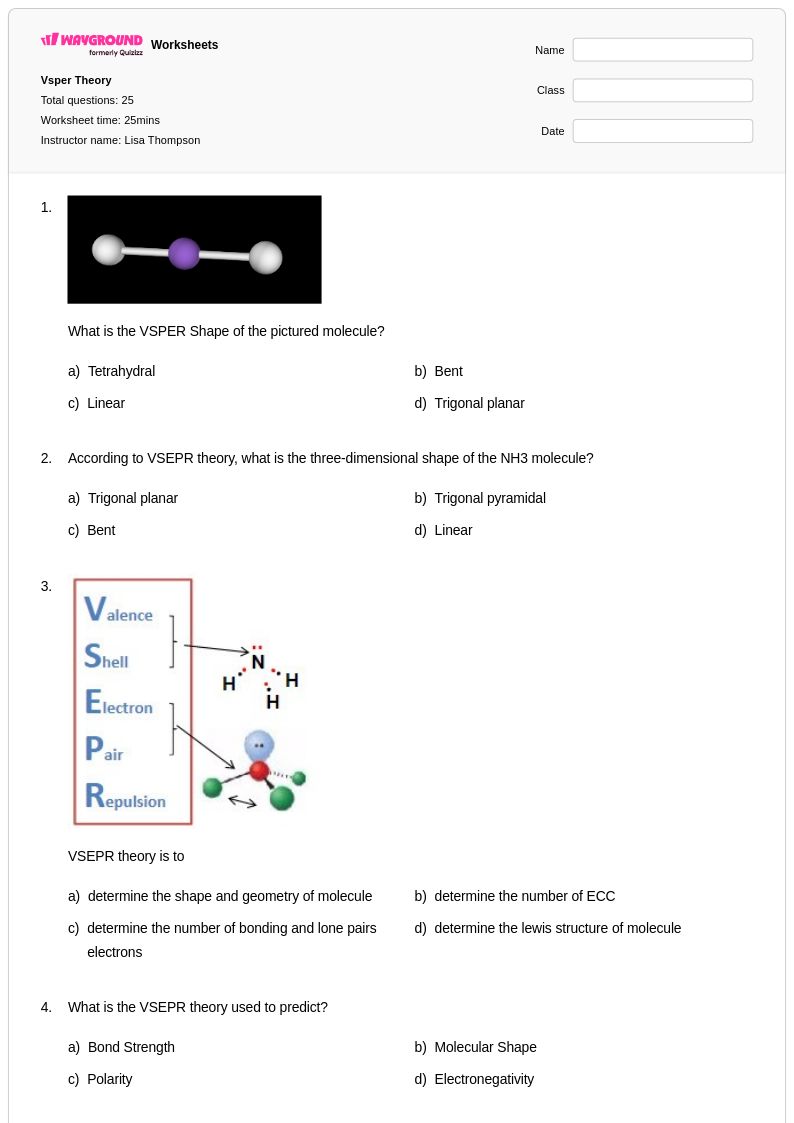
25 Hỏi
10th - Uni
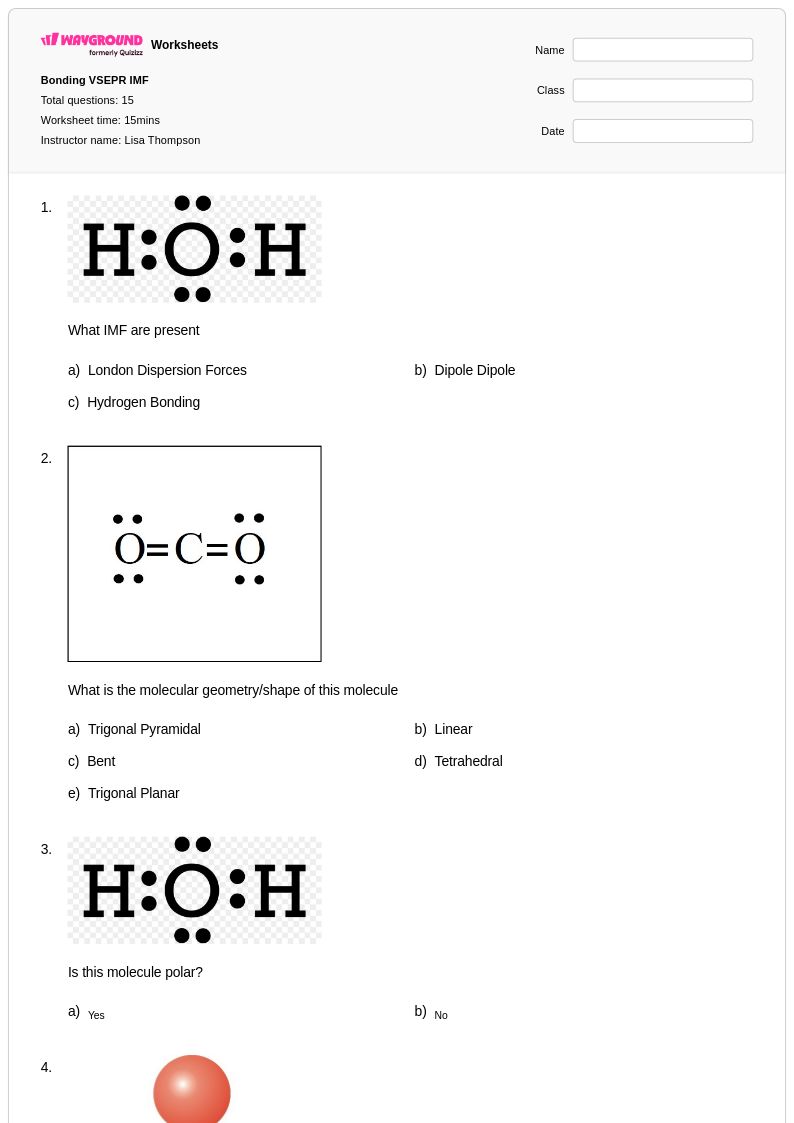
15 Hỏi
11th - Uni
Khám phá các bảng tính chủ đề khác cho class 11
Explore printable VSEPR Theory worksheets for Class 11
VSEPR Theory worksheets for Class 11 students available through Wayground (formerly Quizizz) provide comprehensive practice with molecular geometry prediction and three-dimensional molecular structure analysis. These expertly designed resources strengthen students' understanding of electron pair repulsion principles, enabling them to accurately determine molecular shapes for compounds ranging from simple diatomic molecules to complex polyatomic structures with multiple bonding domains. The worksheets systematically guide students through identifying central atoms, counting electron pairs, distinguishing between bonding and lone pairs, and applying VSEPR rules to predict geometries such as linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral arrangements. Each printable resource includes detailed practice problems that progress from basic molecular geometry identification to advanced applications involving bond angles, molecular polarity, and hybrid orbital theory, with comprehensive answer keys provided in convenient PDF format for immediate feedback and self-assessment.
Wayground (formerly Quizizz) supports chemistry educators with an extensive collection of millions of teacher-created VSEPR Theory resources that streamline lesson planning and enhance student mastery of molecular geometry concepts. The platform's robust search and filtering capabilities allow teachers to quickly locate worksheets aligned with specific curriculum standards and learning objectives, while built-in differentiation tools enable seamless customization for varying skill levels within Class 11 classrooms. Teachers can access these resources in both printable PDF format for traditional worksheet distribution and digital formats for interactive online learning, making them ideal for remediation sessions with struggling students, enrichment activities for advanced learners, and regular skill practice to reinforce three-dimensional thinking in chemistry. The comprehensive answer keys and detailed solution explanations facilitate efficient grading and provide teachers with valuable diagnostic information to identify common misconceptions about electron geometry, molecular geometry relationships, and the impact of lone pairs on molecular shapes.
FAQs
How do I teach VSEPR theory to chemistry students?
What are the most common mistakes students make when predicting molecular geometry using VSEPR?
What practice exercises help students get better at identifying molecular shapes?
How can I differentiate VSEPR theory instruction for students at different ability levels?
How do I use VSEPR theory worksheets from Wayground in my classroom?
How does VSEPR theory connect to other chemistry concepts students need to know?
