
11 Q
8th - 12th

10 Q
2nd - Uni
10 Q
9th - 12th

100 Q
12th

16 Q
9th - 12th

14 Q
9th - 12th
28 Q
9th - 12th

20 Q
10th - 12th
23 Q
9th - 12th

49 Q
9th - 12th

27 Q
12th

22 Q
7th - Uni

25 Q
11th - Uni

14 Q
6th - Uni
16 Q
9th - 12th
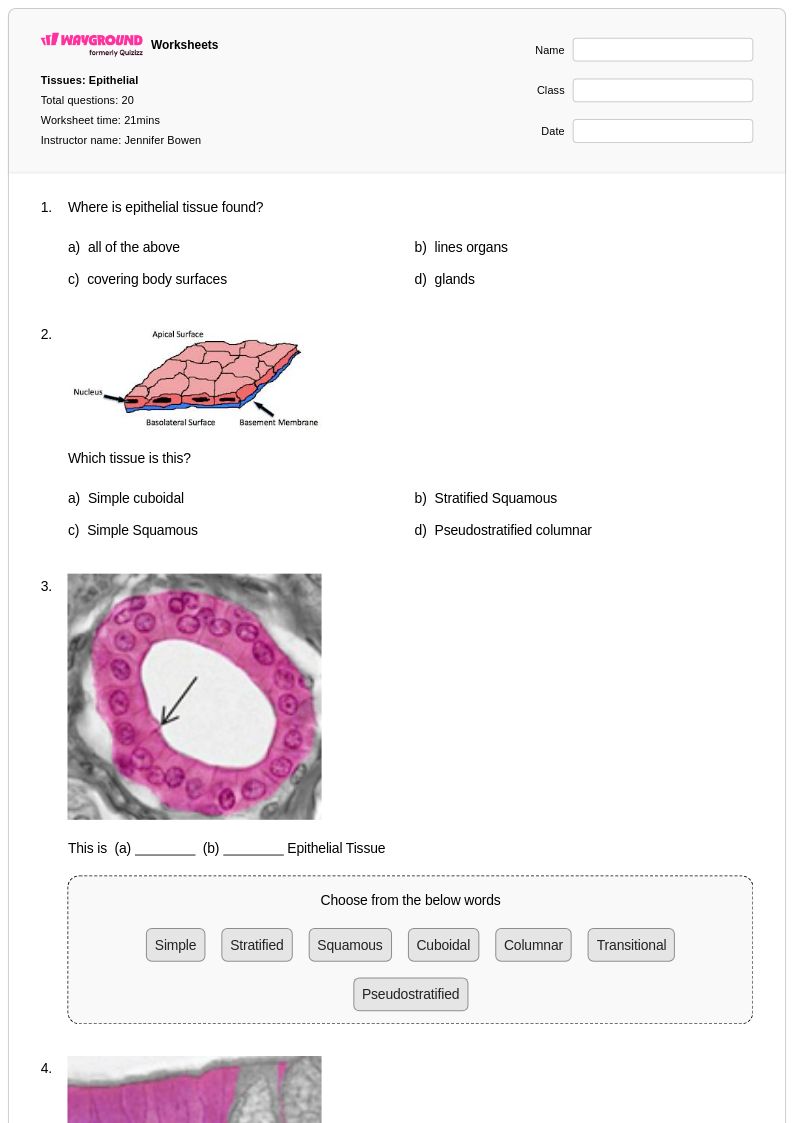
20 Q
12th
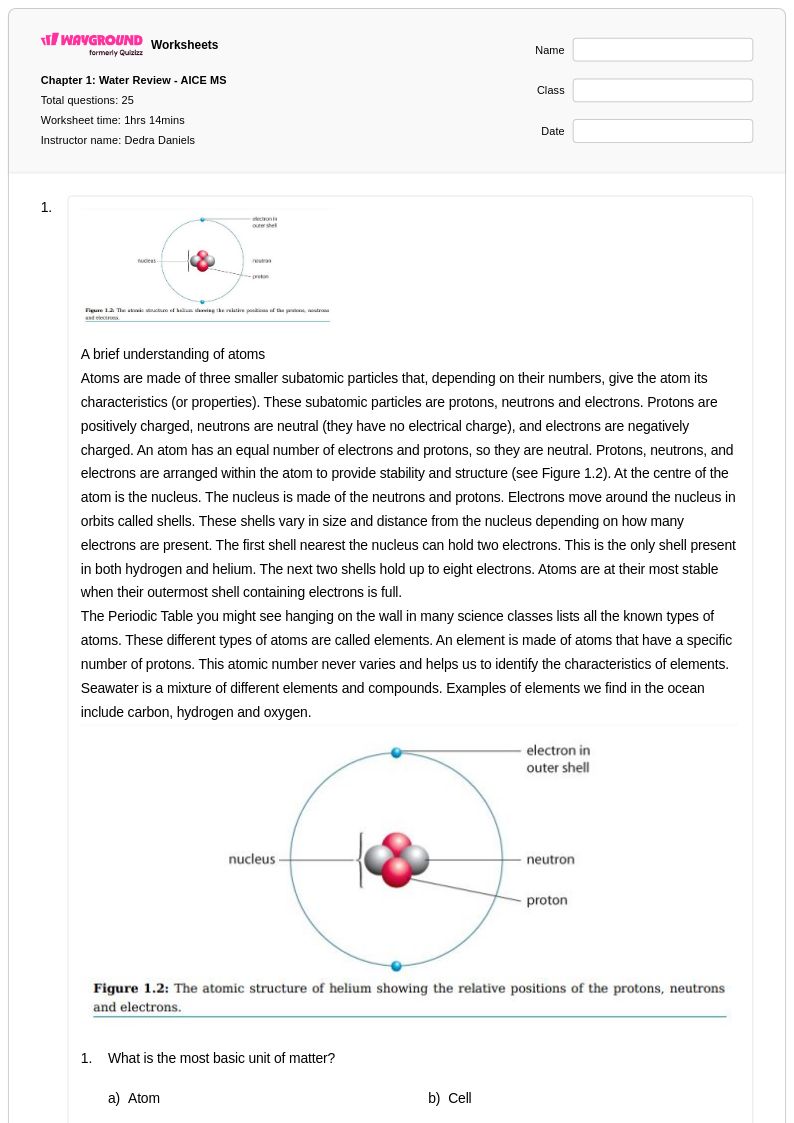
59 Q
9th - 12th

45 Q
12th

25 Q
12th

27 Q
9th - 12th

82 Q
12th
23 Q
9th - 12th
12 Q
12th
25 Q
9th - 12th
Explore Other Subject Worksheets for grade 12
Explore printable Salts worksheets for Grade 12
Salts worksheets for Grade 12 chemistry students available through Wayground (formerly Quizizz) provide comprehensive coverage of ionic compound formation, nomenclature, and properties essential for advanced chemistry mastery. These expertly designed resources strengthen students' understanding of salt formation through acid-base neutralization reactions, precipitation reactions, and the relationship between molecular structure and solubility patterns. Students engage with practice problems that challenge them to predict salt formation, write balanced chemical equations, and analyze the behavior of salts in aqueous solutions. Each worksheet collection includes detailed answer keys that support independent learning and self-assessment, while printable pdf formats ensure accessibility for both classroom instruction and homework assignments. The free resources emphasize critical thinking skills by requiring students to connect theoretical concepts with real-world applications, from understanding ocean chemistry to analyzing industrial salt production processes.
Wayground (formerly Quizizz) empowers educators with millions of teacher-created salt worksheets that streamline lesson planning and enhance student engagement in Grade 12 chemistry courses. The platform's robust search and filtering capabilities allow teachers to quickly locate resources aligned with specific curriculum standards and learning objectives, whether focusing on ionic bonding theory, solubility rules, or quantitative analysis of salt solutions. Differentiation tools enable instructors to customize worksheets for diverse learning needs, supporting both remediation for struggling students and enrichment opportunities for advanced learners. Teachers can seamlessly transition between printable and digital formats, including downloadable pdf versions, to accommodate various classroom environments and teaching preferences. These flexible resources prove invaluable for targeted skill practice, formative assessment, and reinforcing complex concepts that bridge general chemistry principles with more advanced topics in analytical and physical chemistry.
FAQs
How do I teach students the difference between salt types in chemistry?
What practice exercises help students get better at naming ionic compounds and salts?
What common mistakes do students make when learning about salts and ionic compounds?
How do I use salts worksheets to assess whether students understand neutralization reactions?
How can I use Wayground's salts worksheets in my chemistry class?
How do I support struggling students when teaching salt chemistry without slowing down the rest of the class?
