

Elemental Carbon and Simple Organic Compounds
Presentation
•
Science
•
10th Grade
•
Hard
Joseph Anderson
FREE Resource
69 Slides • 50 Questions
1
Organic Chemistry
2

3
let's talk groups!
Groups (also called families) are the vertical (up and down) columns on the periodic table
There are 18 groups on the periodic table BUT only groups 1 and 2 and 13-18 follow any sort of pattern.
The group number can help you quickly figure out the number of valence electrons.

4
The Group Pattern
The bolded number at the top of the group tell the number of valence electrons the group has.
Group 1 has 1 valence electron
Group 2 has 2 valence electrons
Group 13 has 3 valence electrons
Group 14 = 4, 15 = 5, 16 = 6, 17 = 7
Group 18 has 8 valence electrons (a full outer shell, except for Helium)

5

6
Multiple Choice
Valence electrons are
the electrons in the outer most energy level
all the electrons in the atom
7
Multiple Choice

What are the vertical columns on the Periodic Table called?
groups
periods
nonmetals
metals
metalloids
8
Multiple Choice

Which elements have a full set of valence electrons?
period 1
group 1
period 18
group 18
9
Multiple Choice

Which elements have 5 valence electrons?
period 5
group 5
period 15
group 15
10
Multiple Choice

What do the vertical columns on the periodic table tell us?
The number of orbitals
The number of valence electrons (electrons on the outer most shell)
11

12
Electron Shells
Each element in each period have the same number of electron shells.
Hydrogen (1e-) and Helium (2e-) are in period 1 and have 1 electron shell.
Lithium (3e-), Carbon (6e-), Oxygen (8e-), and Neon (10e-) are all in period 2 and have 2 electron shells.

13
Multiple Choice
What are the columns referred to on the Periodic Table of Elements?
Families or groups
Periods
14
Multiple Choice
What are the rows referred to on the Periodic Table of Elements?
Families or groups
Periods
15

16
Multiple Choice
Atomic Mass equals the combined number of
Protons and Electrons
Electrons and Neutrons
Protons and Neutrons
17
Multiple Choice

Which elements have 7 valence electrons?
period 7
group 17
period 17
group 7
18
Multiple Choice

The chemical symbol for an element is always represented by only 1 letter
True
False
19
Organic Chemistry- The study of carbon & carbon compounds.
Organic compounds are the primary constituents of all living organisms.

20
What is organic chemistry?
The study of carbon compounds.
Carbon
the backbone of biological molecules


21

22
Why is carbon essential to life on Earth?
6th element on the periodic table
A compound found mainly in living things is known as an organic compound.
Carbon is the main element in organic compounds

23
Significance of Carbon


24

25
Multiple Choice
Carbon is essential to life because it is the main element in inorganic compounds.
True
False
26
Carbon makes organic compounds, so what?
A compound is a substance that consists of two or more elements.
The smallest particle of a compound is called a molecule.
Consider water: two hydrogen atoms and one oxygen. Held together by chemical bonds.
A chemical bond is a force that holds molecules together; they are formed from reactions.

27
Multiple Choice
What is the an important element for living things.
Neon
Hydrogen
Helium
Carbon
28

29
Multiple Choice

Which of these element(s) will C form bonds with?
C
H
O
N
all of these
30
Multiple Choice

What type of compounds have at least 1C atom bonded to at least 1H atom?bon
inorganic
organic
ionic
31
Multiple Choice
Which of the following is an organic compound that makes up living things?
carbohydrates
salts
oxides
hydrocarbons
32
Carbon and Covalent bonds

Carbon has 4 valence electrons (outer shell electrons)
Allows for several types of covalent bonds.
33
Covalent Bonds
Covalent bonds exist between NONMETAL and NONMETAL.
Example: H2, H2O, NO3, CH4
Covalent bonds SHARE electrons.
Shared electrons don’t belong to either atom.

34
Multiple Choice

How many bonds will a C atom form?
1
2
3
4
35
Multiple Choice

2 Nonmetals
1 Nonmetal and 1 Metal
2 Metals
2 Noble Gases
36
Multiple Choice

When one atom takes the other atom's electron
When the atom shares an electron with an another atom
When the two nucleus merge
When the neutrons leave the nucleus
37
Organic Compounds
Covalently bonded compounds containing carbon, excluding carbonates and oxides.
Few exceptions, such as Na2CO3, CO, and CO2, that are considered inorganic.
All organic compounds contain carbon atoms.
Aspirin, polyethylene in plastic bags, citric acid in the fruit, and amino acids in animals are all examples of organic compounds.

38
Carbon and Covalent bonds

able to form chains
able to form rings

39
Carbon atom is unique
Ability to form long chains and rings of covalently bonded atoms.
Catenation, the covalent bonding of an element to itself to form chains or rings.
Carbon atoms bind readily to elements with similar electronegativities.

40

41
Carbon atom is unique
Hydrocarbons are composed of only carbon and hydrogen; they are the simplest organic compounds
Other organic compounds contain hydrocarbon backbones to which other elements, primarily O, N, S, and the halogens, are attached.

42
Multiple Choice

nitrogen
oxygen
carbon
sodium
43
Carbon atom is unique
Carbon is able to form 4 covalent bonds (4 valence electrons) with other carbon or other elements.
The bonding capabilities of carbon also allow for different arrangements of atoms

44
Fill in the Blanks
45

46
Multiple Choice
cat
calcium
carbon
crypton
47

48
Multiple Choice

How many chemical bonds can each carbon atom form?
one
two
three
four
49

50
Macromolecule Formation
Monomers
small units that can join together to form polymers
Polymers
large compounds formed from combinations of many monomers

51
Multiple Choice
Which of the following is NOT a unique nature of carbon?
Forms 4 strong covalent bonds
It can form only one type of bond
It has the ability to form stable bonds with itself
It can form multiple bonds
52
Multiple Choice
Which type of bonding is present within organic molecules?
Covalent bond
Ionic bond
Metallic bond
Hydrogen bond
53
Multiple Choice
How many unpaired electrons does carbon have when it is excited?
2
3
4
5
54
Multiple Choice
This unique ability of carbon is defined as the ability of atoms to form stable bonds.
Polar Bonding
Carbonation
Covalent Bonding
Catenation
55
Multiple Choice
Which two elements do most organic compounds contain?
Carbon and hydrogen
Carbon and oxygen
Oxygen and hydrogen
Carbon and nitrogen
56
Worksheet
57
Characteristics of Organic Compounds
Mostly built up of only three elements- carbon, hydrogen and oxygen. Other elements like halogen, nitrogen as well as phosphorous are also present but to a lesser extent.

58
Characteristics of Organic Compounds
Possess complex structures and high molecular weights
Their properties are decided by certain active atom or group of atoms known as the functional group.
They are mostly insoluble in water but soluble in organic solvents.

59
4 types of polymers
Carbohydrates
Lipids
Proteins
Nucleic acids

60
Characteristics of Organic Compounds
They are combustible in nature
They have low melting points – due to weak intermolecular forces.
Chemical reactions involving organic compounds proceed at slower rates.
They react slower than ionic compounds – due to strong covalent bonds between atoms.

61
Multiple Choice
Carbohydrates, Lipids, Nucleic Acids, and Oxygen
Carbohydrates, Lipids, Proteins, and Carbon
Carbohydrates, Lipids, Proteins, Nucleic Acids
Carbon, Lipids, Proteins, Nucleic Acids
62
Open Ended
Give at least four (4) characteristics of organic compounds.
63

64
Representing Organic Molecules
Simply shows the number of each type of atom present. It tells you nothing about the bonding within the compound

65
Macromolecule Chart Carbohydrates
Elements
C
H
O
Monomer
C, H, O in a 1-2-1 ratio

66
Representing Organic Molecules
Gives the simplest possible whole number ratio of the different types of atom within the compound.

67
Macromolecule Chart
Carbohydrates
Function(s)
energy
storage
structure
Examples
glucose
starch
glycogen

68
Representing Organic Molecules
A text-based, each carbon atom is listed separately, with atoms attached to it following. An exception is cyclic parts of molecules, e.g. benzene, where the carbons are grouped.

69
Macromolecule Chart Carbohydrates
Function(s)
energy
storage
structure

70
Representing Organic Molecules
Shows all of the atoms and all of the bonds present in an organic compound. The bonds are represented as lines.

71
Macromolecule Chart Carbohydrates
Examples
glucose
starch
glycogen
sucrose

72
Representing Organic Molecules
Similar to the displayed formula-not all bonds are shown, although all atoms are still indicated using subscript numbers. Carbon-hydrogen bonds are often simplified.

73
Macromolecule Chart: Carbohydrates
Unusual Information
Monosaccharide
simple sugar
glucose

74
Representing Organic Molecules
In a skeletal formula, most hydrogen atoms are omitted, and line ends or vertices represent carbons. Functional groups and atoms other than carbon or hydrogen are still shown. Easiest to draw & commonly used.

75
Macromolecule Chart: Carbohydrates
Unusual Information
Disaccharide
2 simple sugars
sucrose

76
Multiple Choice

The formula is an example of ...
Skeletal formula
Structural formula
Displayed formula
Condensed formula
77
Macromolecule Chart: Carbohydrates
Unusual Information
Polysaccharide
many simple sugars
starch/glycoge

78
Multiple Choice

This represents a...
Condensed formula
Structural formula
Displayed formula
Skeletal formula
79
Multiple Choice
The monomer Glucose/Monosaccharides make up the polymer....
Proteins
Lipids
Nucleic Acids
Carbohydrates
80
Multiple Choice

This is an example of...
Molecular formula
Empirical formula
Condensed formula
Displayed formula
81
Multiple Choice
82
Fill in the Blanks

Type answer...
83
Multiple Choice

This molecule could be the building block of...
nucleic acids
proteins
lipids
polysaccharides
waxes
84
Open Ended
What have you learned from today's lesson? List as many as you can.
85

86
Macromolecule Chart Lipids
Elements
C
H
Monomer
Glycerol
3 Carbons
1 alcohol
3 fatty acid chain

87
Macromolecule Chart Lipids
Function(s)
store energy
biological membranes
waterproof covering
hormones
structural support

88
Macromolecule Chart Lipids
Examples
fats
oils
waxes
steroids
corn oil
hormones

89
Macromolecule Chart Lipids
Unusual Information
Saturated
Each C in fatty acid chain has single bond with another C

90
Macromolecule Chart Lipids
Unusual Information
Monounsaturated
Have 1 C in fatty acid chain with a double bond to another C

91
Macromolecule Chart Lipids
Unusual Information
Polyunsaturated
2 or more C in fatty acid chain have double bond with another C

92
Multiple Choice

93
Multiple Choice
The monomer Fatty acids make up the polymer....
Proteins
Lipids
Nucleic Acids
Carbohydrates
94
Multiple Choice
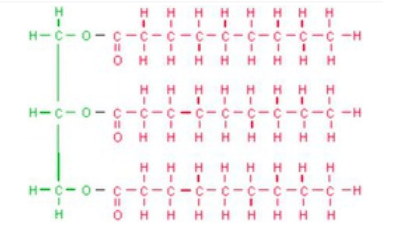
95

96
Macromolecule Chart: Nucleic Acids
Elements
C
H
N
O
Phosphate (PO4)

97
Macromolecule Chart: Nucleic Acids
Monomer
Nucleotides
5 carbon sugar
phosphate group
nitrogenous base

98
Macromolecule Chart: Nucleic Acids
Function(s)
store and transmit genetic information
biological membranes
blueprint for making proteins

99
Macromolecule Chart: Nucleic Acids
Examples
Deoxyribonucleic acid
DNA
RIbonucleic Acid
RNA


100
Macromolecule Chart: Nucleic Acids
Unusual Information
in charge of characteristics of life
at least 2 nucleotides combined by a covalent bonds

101
Multiple Choice
102
Multiple Choice

103
Multiple Choice
The monomer Nucleotides make up the polymer....
Proteins
Lipids
Nucleic Acids
Carbohydrates
104

105
Macromolecule Chart: Proteins
Elements
C
H
N
O

106
Macromolecule Chart: Proteins
Monomer
Amino Acids
Amino group (NH)
Carboxyl group (COOH)
H off central C
R-group

107
Macromolecule Chart: Proteins
Function(s)
controls rate of reactions for life processes
forms bones and muscles

108
Macromolecule Chart: Proteins
Function(s)
transports substances in / out of cells
helps fight diseases

109
Macromolecule Chart: Proteins
Examples
Collagen
Carotene
Enzymes
Hemoglobin

110
Macromolecule Chart: Proteins
Unusual Information
more than 20 amino acids (aa) formed in nature
bind to other aa by binding amino group to carboxyl group

111
Macromolecule Chart: Proteins
Unusual Information
form poly peptide chains
Fold into complex shapes

112
Multiple Choice
113
Multiple Choice

114
Multiple Choice

115

116
Multiple Choice
117
Multiple Choice
118
Multiple Choice
119
Multiple Choice
Organic Chemistry
Show answer
Auto Play
Slide 1 / 119
SLIDE
Similar Resources on Wayground

121 questions
F4 Mathematics Statistics Lesson
Presentation
•
9th - 10th Grade

105 questions
Cells
Presentation
•
10th Grade

110 questions
End of Year Review Physical Science
Presentation
•
8th Grade

114 questions
2025 Cell Transport ORG PPT
Presentation
•
9th Grade

120 questions
PIT_Chapter 6 Office Application Features
Presentation
•
9th - 12th Grade

106 questions
-AR Verb Lesson
Presentation
•
8th Grade

112 questions
4. Quarter 2 - Module 2 Lesson 1-Quantitative Research Designs
Presentation
•
12th Grade

121 questions
PhySci Final Review 3: Thermal and Electrical
Presentation
•
9th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

40 questions
NCFE Earth and Environmental Science Released Test
Quiz
•
9th - 12th Grade

10 questions
Exploring Weather Influences and Map Reading
Interactive video
•
6th - 10th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade

20 questions
Final Review Thermochemistry
Quiz
•
9th - 12th Grade

10 questions
Exploring Animal Adaptations: Physical, Behavioral, and Life Cycle Changes
Interactive video
•
6th - 10th Grade

11 questions
Explore Homeostasis and Feedback Mechanisms
Quiz
•
9th - 12th Grade

99 questions
PS Semester 2 Review
Quiz
•
9th - 12th Grade

49 questions
Florida Biology EOC Practice Test
Quiz
•
9th - 12th Grade