
44Q
10th
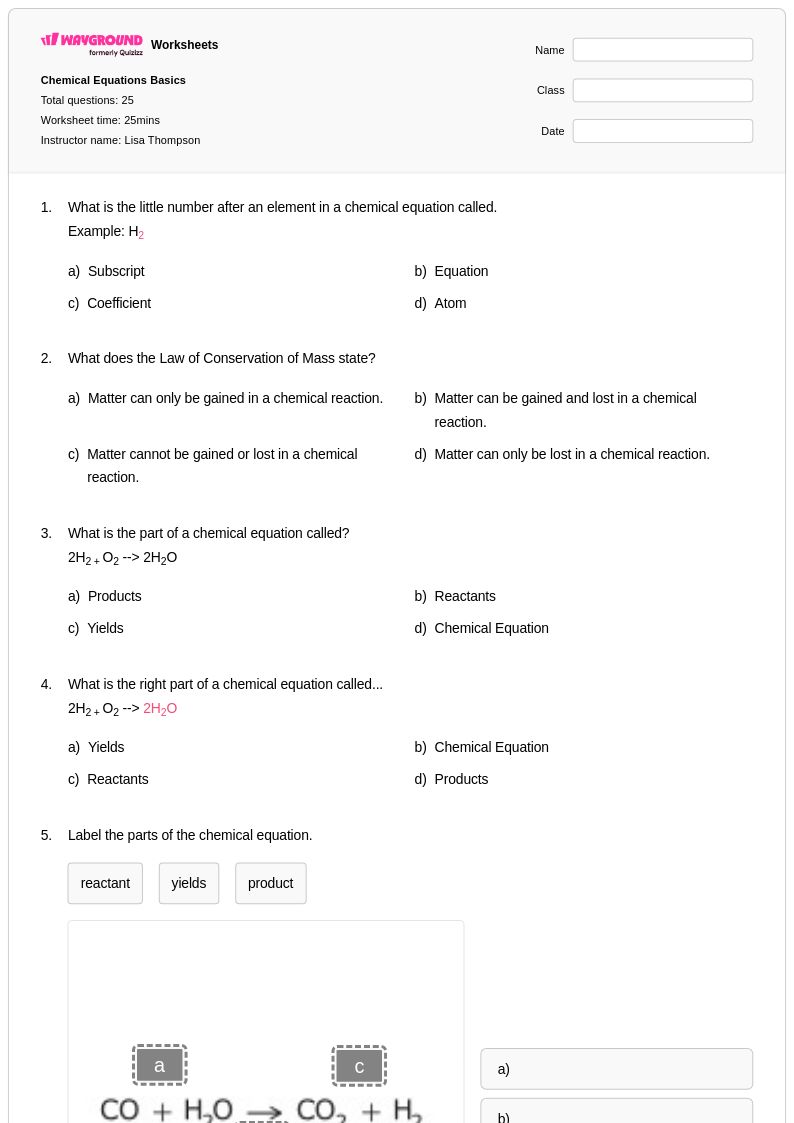
25Q
8th - Uni

25Q
10th - Uni

22Q
9th - 12th

15Q
9th - Uni

20Q
7th - Uni

44Q
9th - 12th
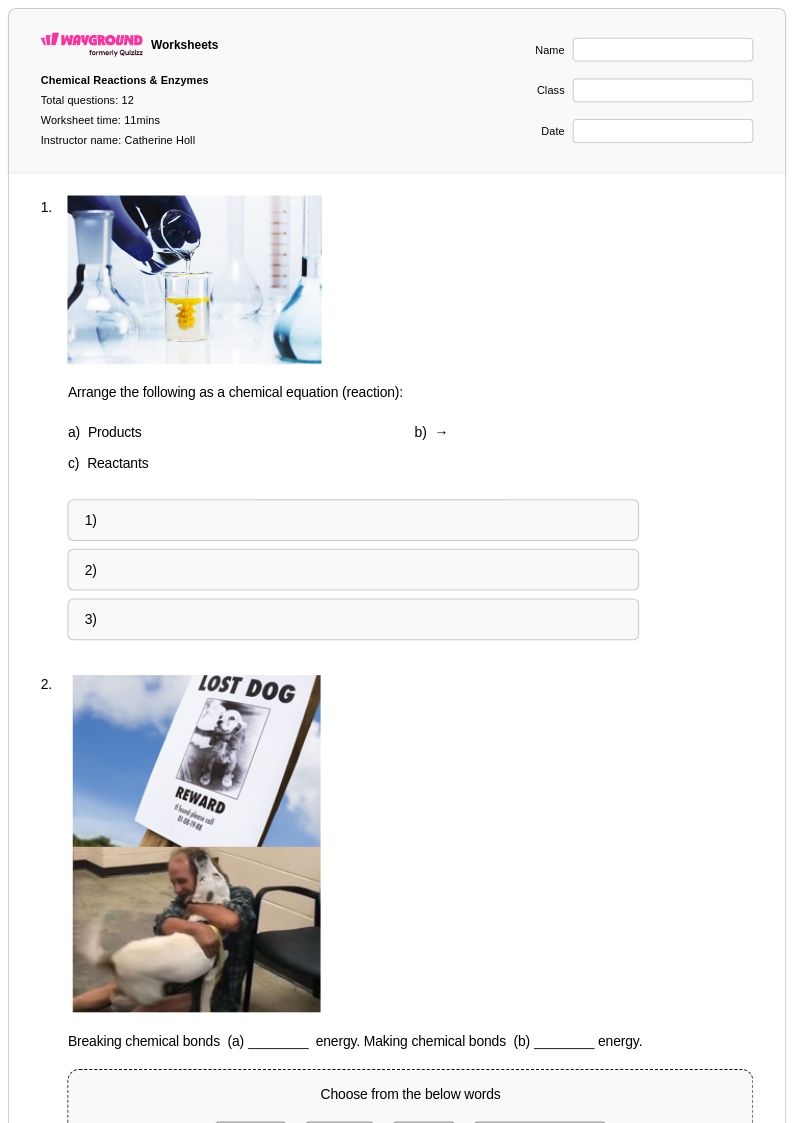
12Q
9th - Uni
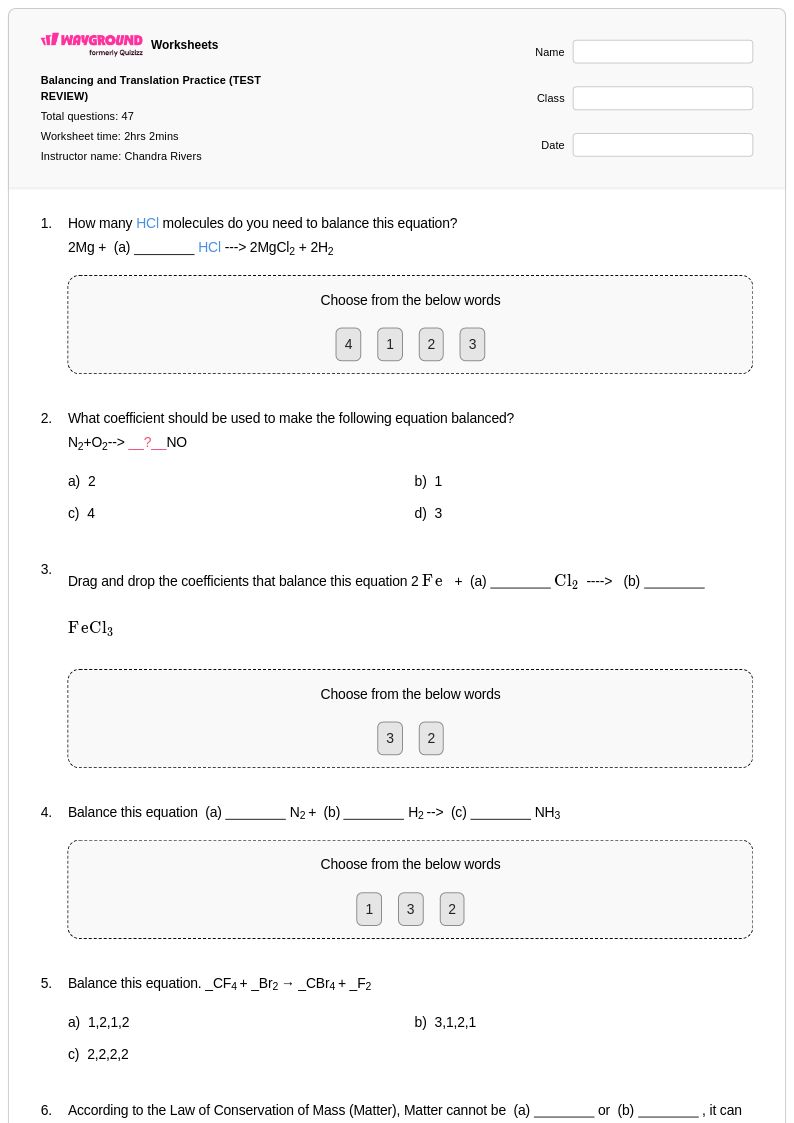
47Q
10th
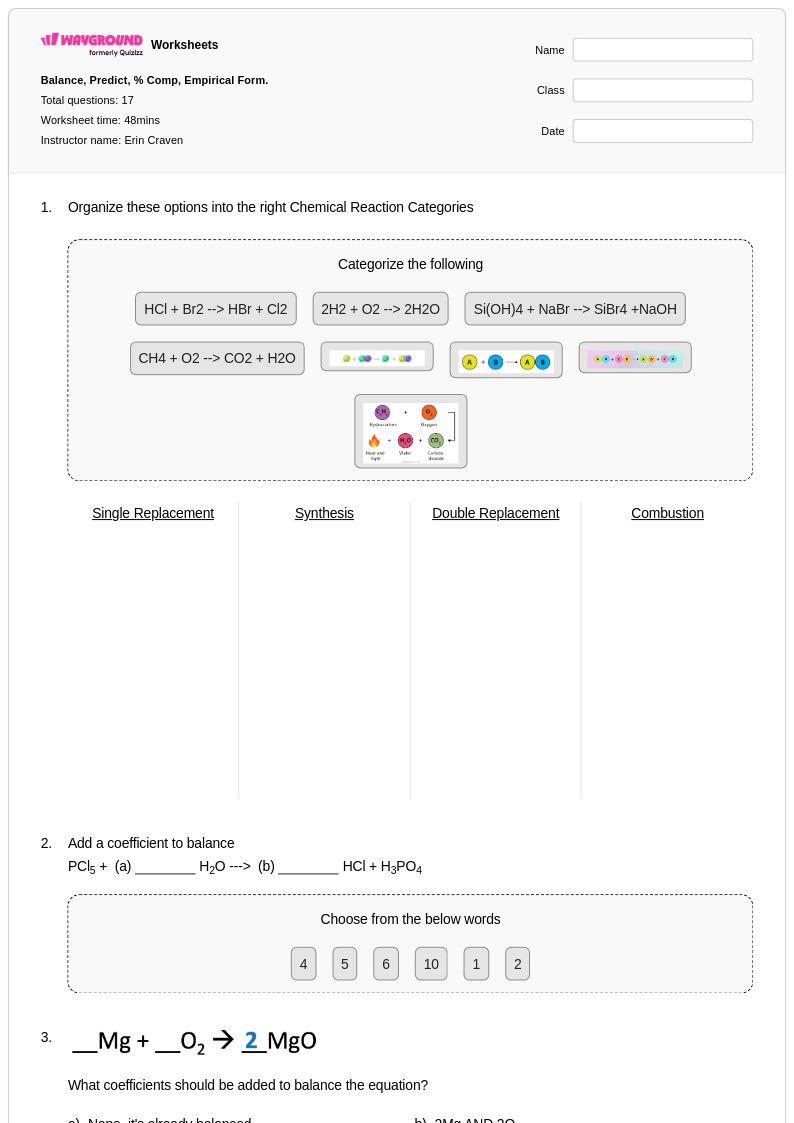
17Q
9th - 12th

25Q
8th - Uni

20Q
10th

22Q
9th - 12th

40Q
9th - 12th

11Q
10th

20Q
8th - Uni

23Q
10th

14Q
10th

21Q
9th - 12th

84Q
10th

75Q
9th - 12th
49Q
9th - 12th

25Q
8th - Uni

80Q
9th - 12th
Explore outras planilhas de assuntos para class 10
Explore printable Balanced Chemical Equations worksheets for Class 10
Balanced chemical equations form the foundation of understanding chemical equilibrium for Class 10 students, requiring mastery of stoichiometric relationships and conservation principles. Wayground's comprehensive collection of balanced chemical equations worksheets provides students with systematic practice in writing, interpreting, and manipulating chemical equations across various reaction types including synthesis, decomposition, single and double replacement reactions. These carefully designed practice problems strengthen essential skills such as identifying reactants and products, applying conservation of mass, determining correct coefficients, and recognizing patterns in chemical behavior. Each worksheet collection includes detailed answer keys and is available as free printable resources in convenient pdf format, allowing students to develop confidence through repeated practice with progressively challenging equations that mirror real-world chemical processes.
Wayground's extensive library draws from millions of teacher-created resources specifically focused on balanced chemical equations and chemical equilibrium concepts, offering educators powerful search and filtering capabilities to locate materials that align with Class 10 science standards and curriculum requirements. The platform's differentiation tools enable teachers to customize worksheets based on individual student needs, from foundational equation balancing for struggling learners to complex multi-step problems for advanced students ready for enrichment activities. Available in both printable and digital formats including downloadable pdfs, these resources support flexible lesson planning whether teachers need quick remediation exercises, comprehensive unit assessments, or supplementary skill practice activities. The robust filtering system allows educators to sort by difficulty level, reaction type, and specific learning objectives, ensuring seamless integration into existing chemistry curricula while providing the scaffolded practice students need to master this critical foundation for advanced chemical equilibrium concepts.
FAQs
How do I teach students to balance chemical equations step by step?
What is the best order to introduce balancing equation practice problems?
What mistakes do students most commonly make when balancing chemical equations?
How can I use balanced chemical equations worksheets to differentiate instruction?
How do I use Wayground's balanced chemical equations worksheets in my classroom?
How do balanced chemical equations connect to other chemistry concepts?
