
5 Q
10th - Uni

21 Q
9th - 12th

23 Q
11th
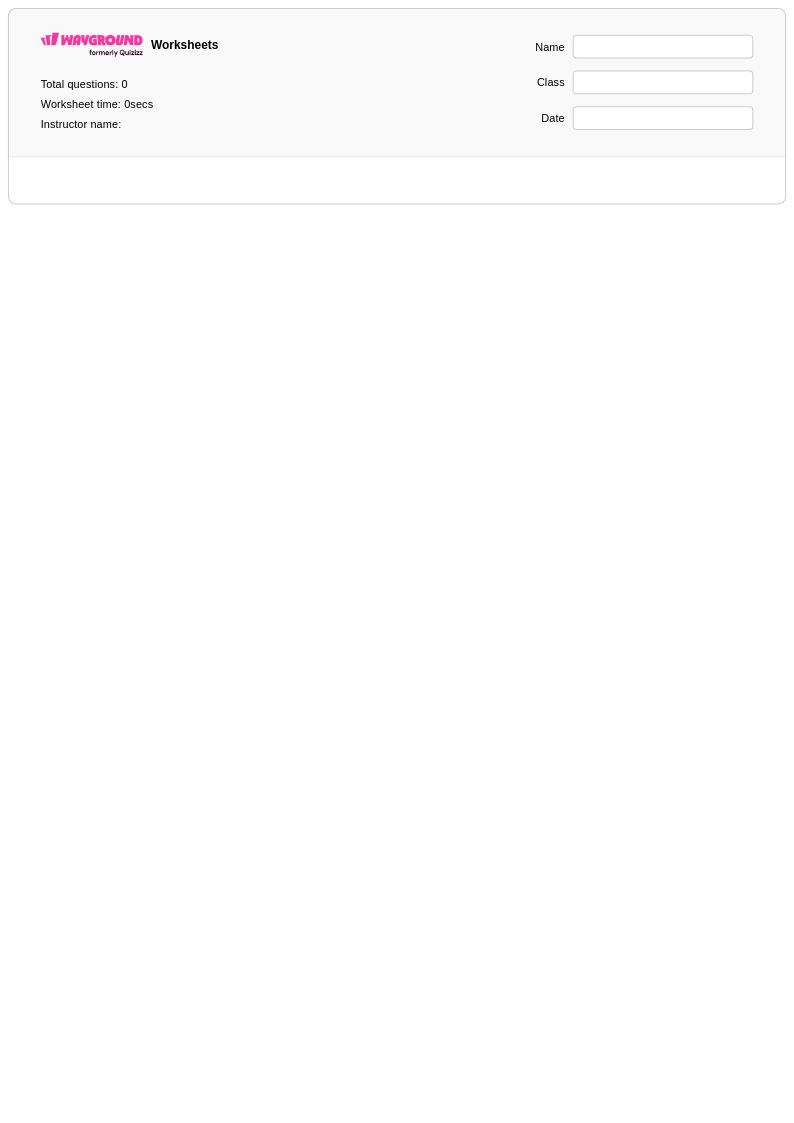
26 Q
11th - Uni

10 Q
11th

42 Q
9th - 12th

37 Q
9th - 12th

25 Q
10th - 11th

30 Q
11th

10 Q
11th

15 Q
9th - 12th
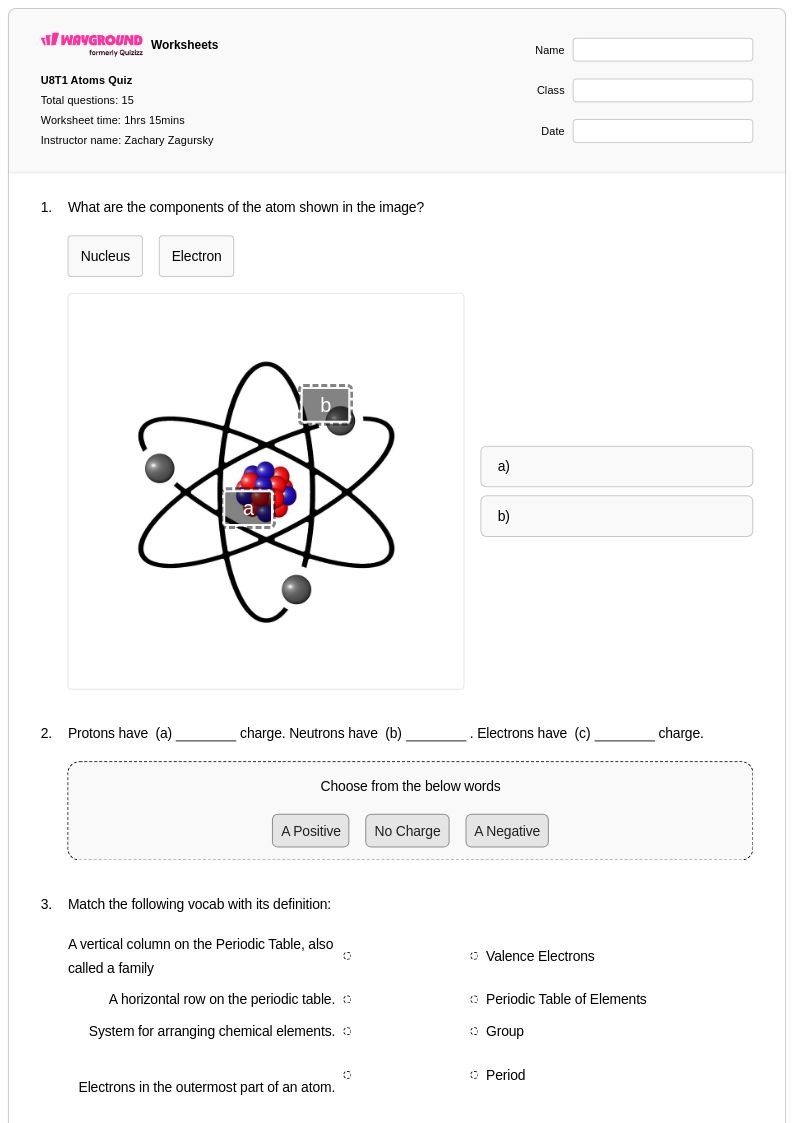
16 Q
9th - 12th

35 Q
10th - Uni

23 Q
9th - 12th

22 Q
9th - 12th
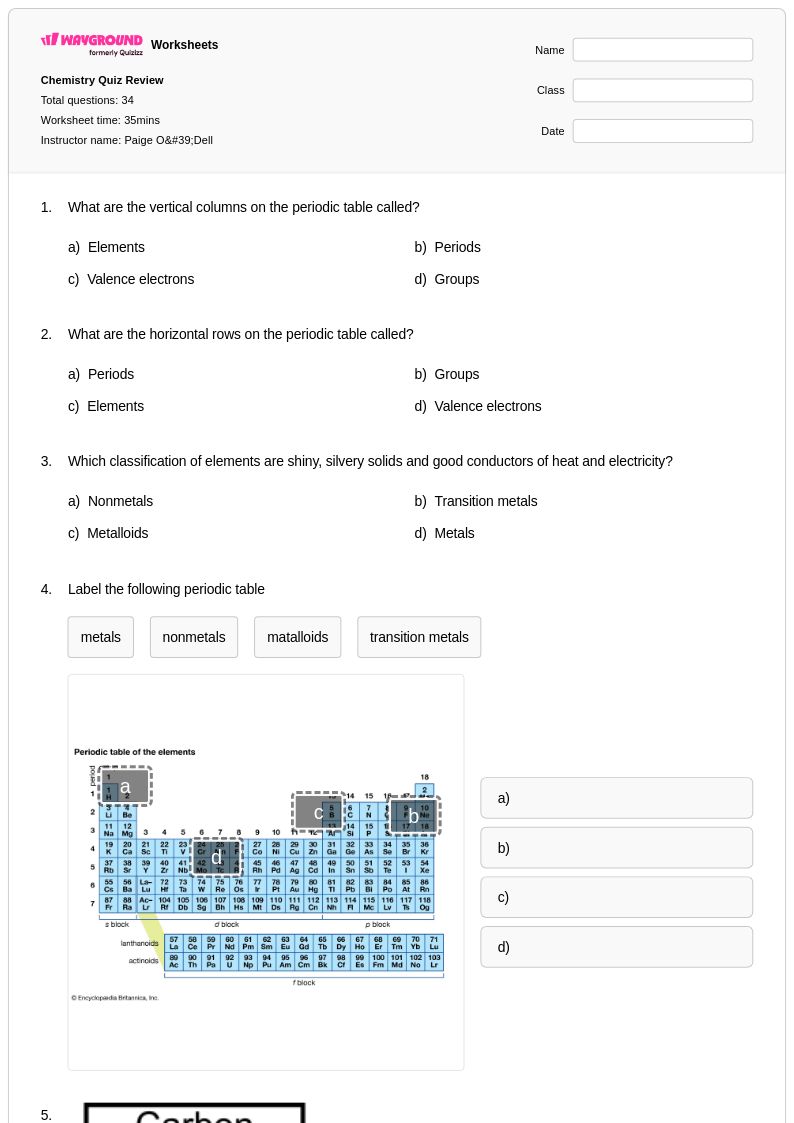
34 Q
9th - 12th

20 Q
9th - 12th

25 Q
11th

17 Q
11th

25 Q
9th - 12th

14 Q
9th - 12th
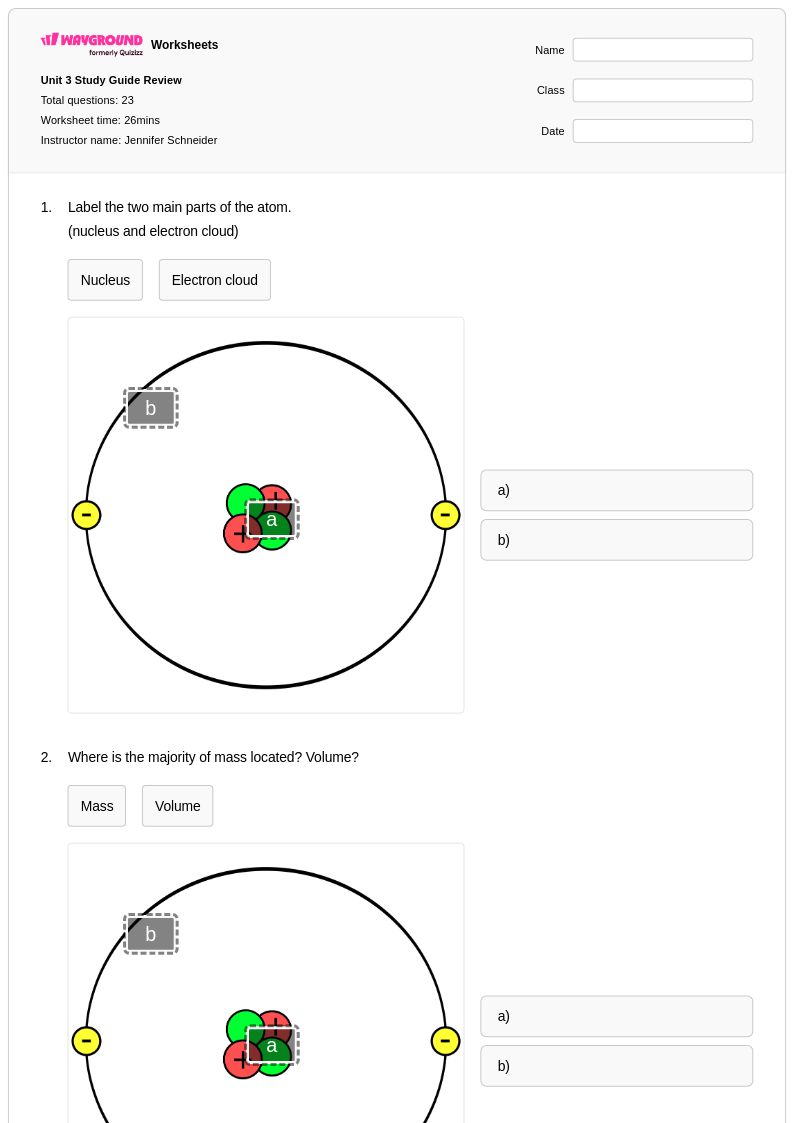
23 Q
9th - 12th
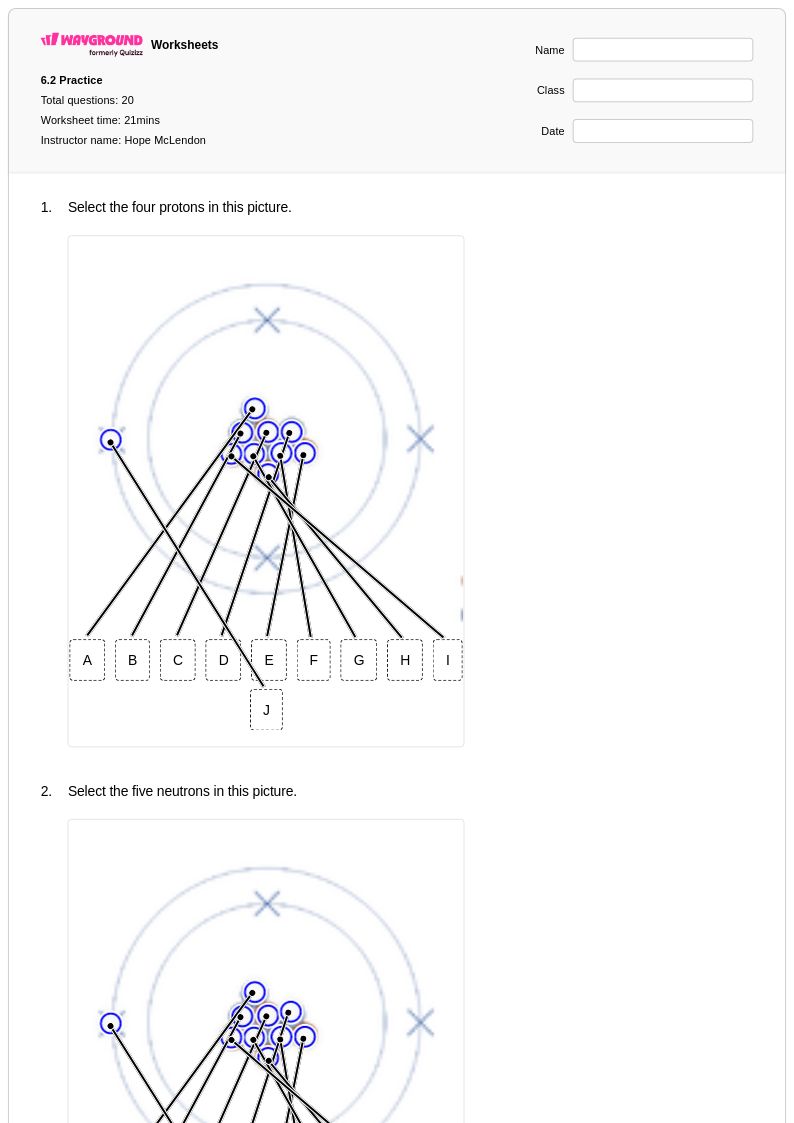
20 Q
11th
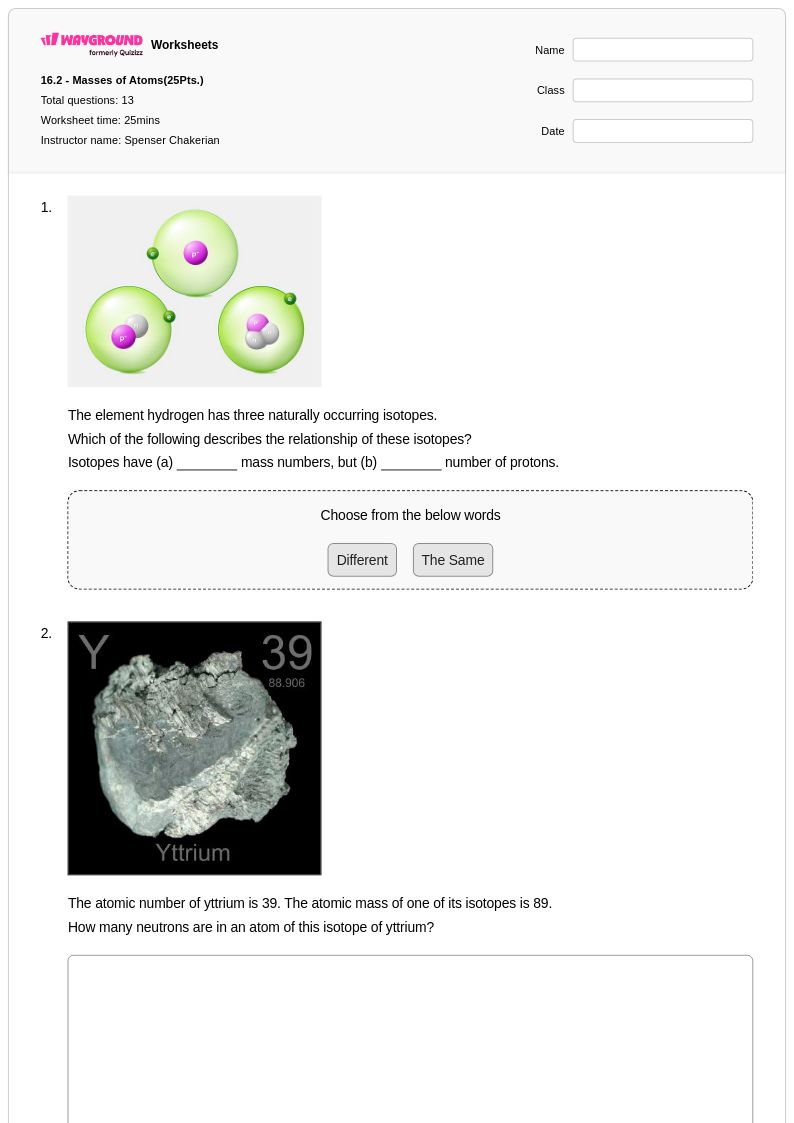
13 Q
9th - 12th
Explore Other Subject Worksheets for class 11
Explore printable Percent Abundance of Isotopes worksheets for Class 11
Percent abundance of isotopes worksheets for Class 11 chemistry students provide comprehensive practice with one of the most fundamental concepts in atomic theory and mass spectrometry. These educational resources through Wayground (formerly Quizizz) focus on developing students' ability to calculate the relative abundance of different isotopes of an element using atomic mass data, understand the relationship between isotopic composition and average atomic mass, and interpret mass spectrometer results. The worksheets strengthen critical analytical skills including mathematical problem-solving, data interpretation, and conceptual understanding of how isotopic variations affect elemental properties. Students work through practice problems that range from basic percentage calculations to complex multi-isotope scenarios, with each worksheet including a comprehensive answer key to support independent learning and self-assessment. These free printables are available in convenient pdf format, making them easily accessible for both classroom instruction and homework assignments.
Wayground (formerly Quizizz) empowers chemistry educators with an extensive collection of millions of teacher-created resources specifically designed for percent abundance of isotopes instruction at the Class 11 level. The platform's robust search and filtering capabilities allow teachers to quickly locate worksheets that align with specific curriculum standards and match their students' varying skill levels. Advanced differentiation tools enable educators to customize practice problems based on individual student needs, whether for remediation of struggling learners or enrichment challenges for advanced students. These versatile resources are available in both printable and digital formats, including downloadable pdf versions, providing maximum flexibility for diverse classroom environments and learning preferences. Teachers can seamlessly integrate these materials into their lesson planning workflow, using them for targeted skill practice, formative assessment, homework assignments, or test preparation, ultimately supporting more effective instruction in this essential chemistry concept.
FAQs
How do I teach percent abundance of isotopes to chemistry students?
What practice problems help students get better at isotope abundance calculations?
What mistakes do students commonly make when calculating percent abundance of isotopes?
How do I use these percent abundance of isotopes worksheets in my classroom?
How does percent abundance connect to average atomic mass on the periodic table?
How can I differentiate percent abundance worksheets for students at different skill levels?
