
10 Q
11th

16 Q
9th - 12th
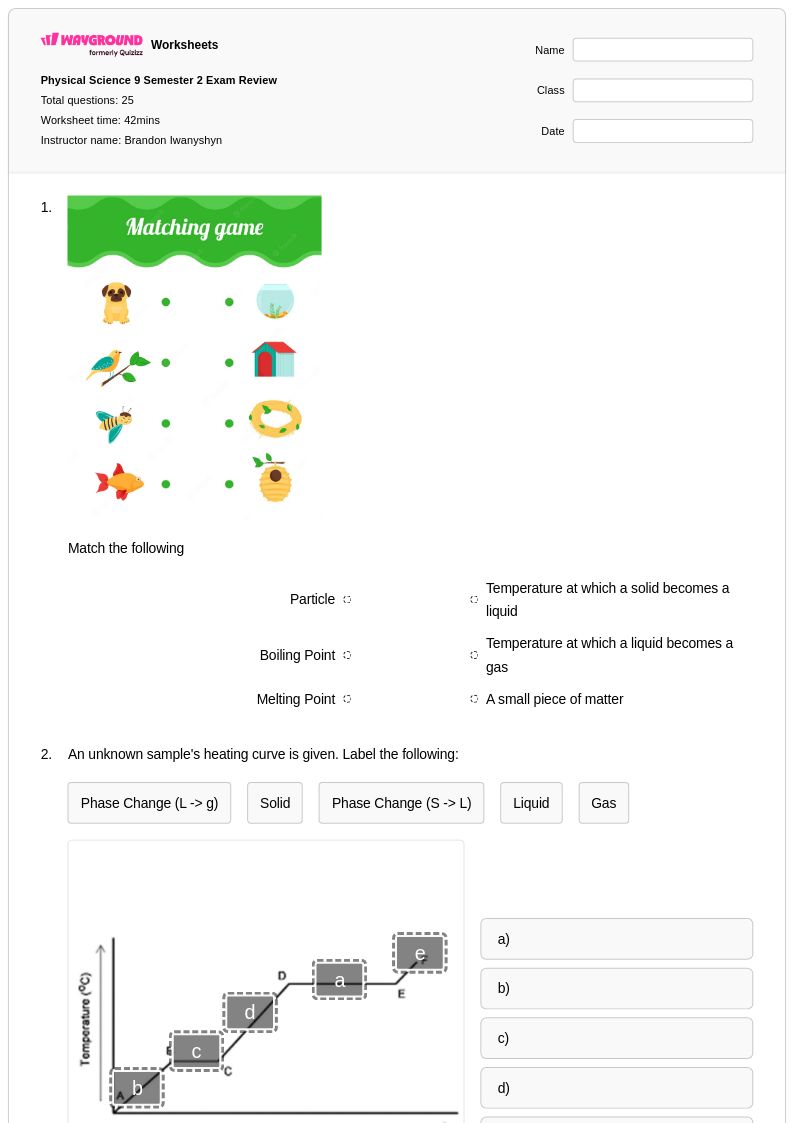
25 Q
9th - 12th
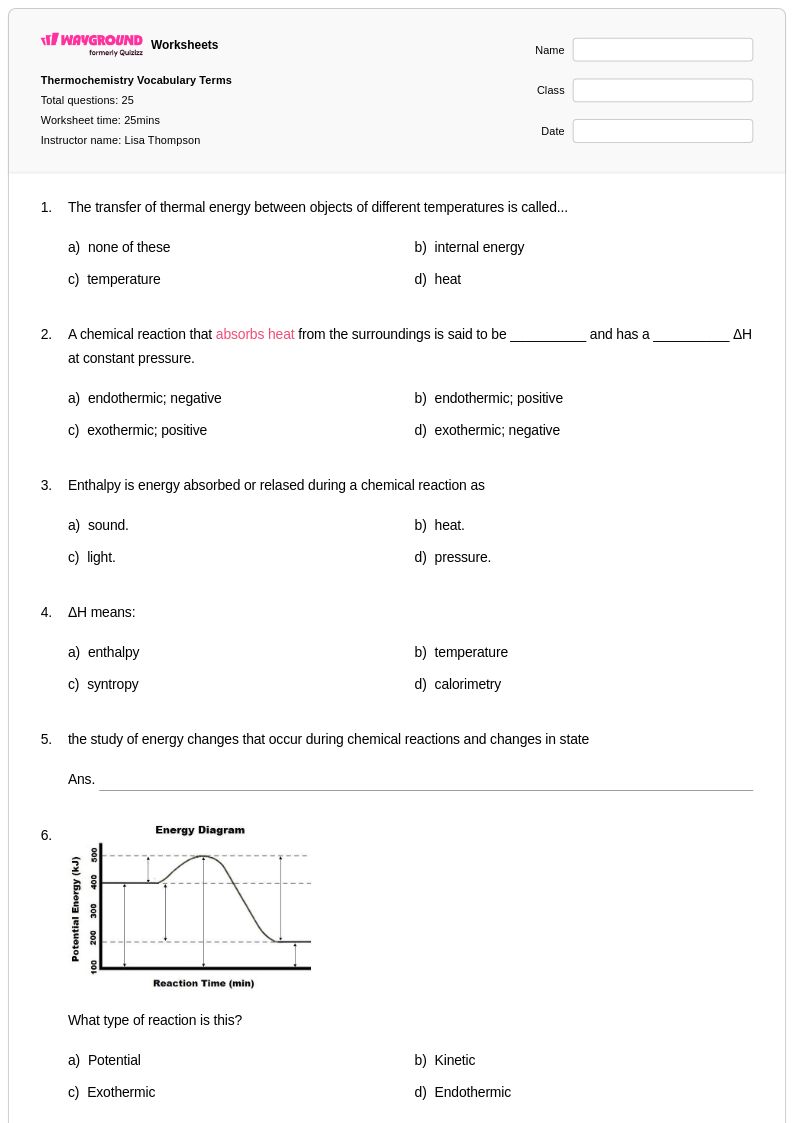
25 Q
10th - Uni
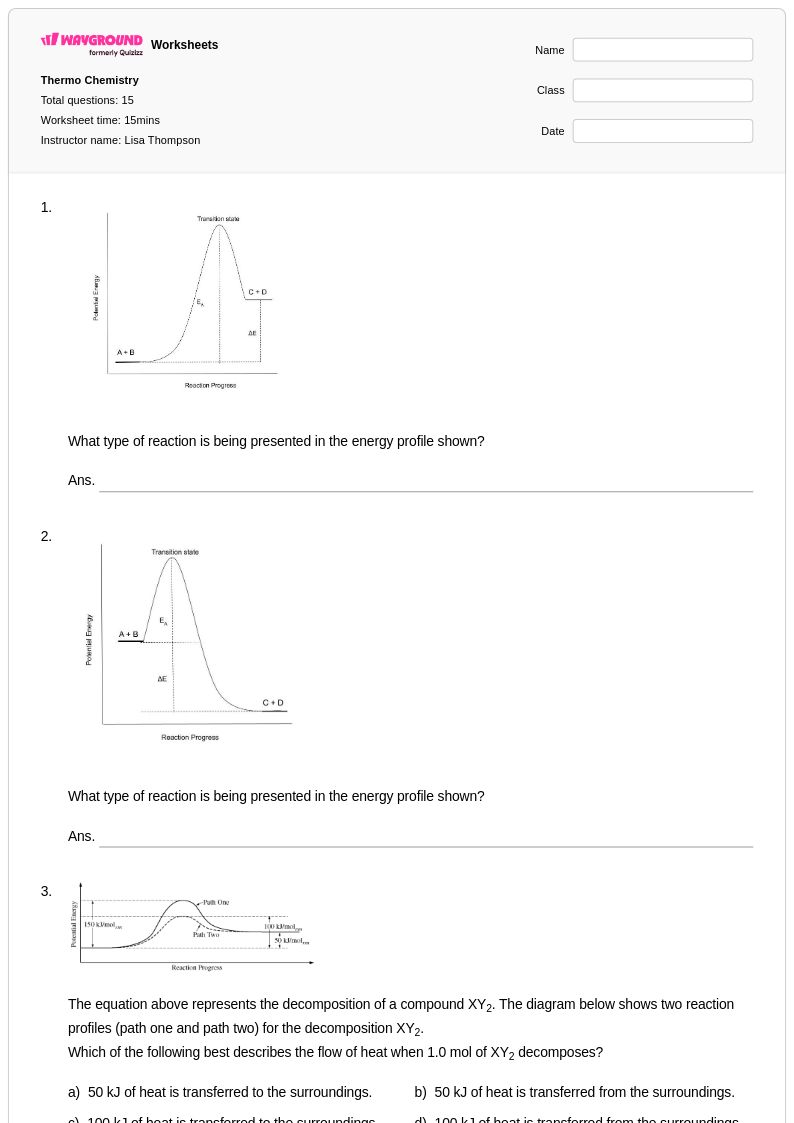
15 Q
11th - Uni

15 Q
10th - Uni
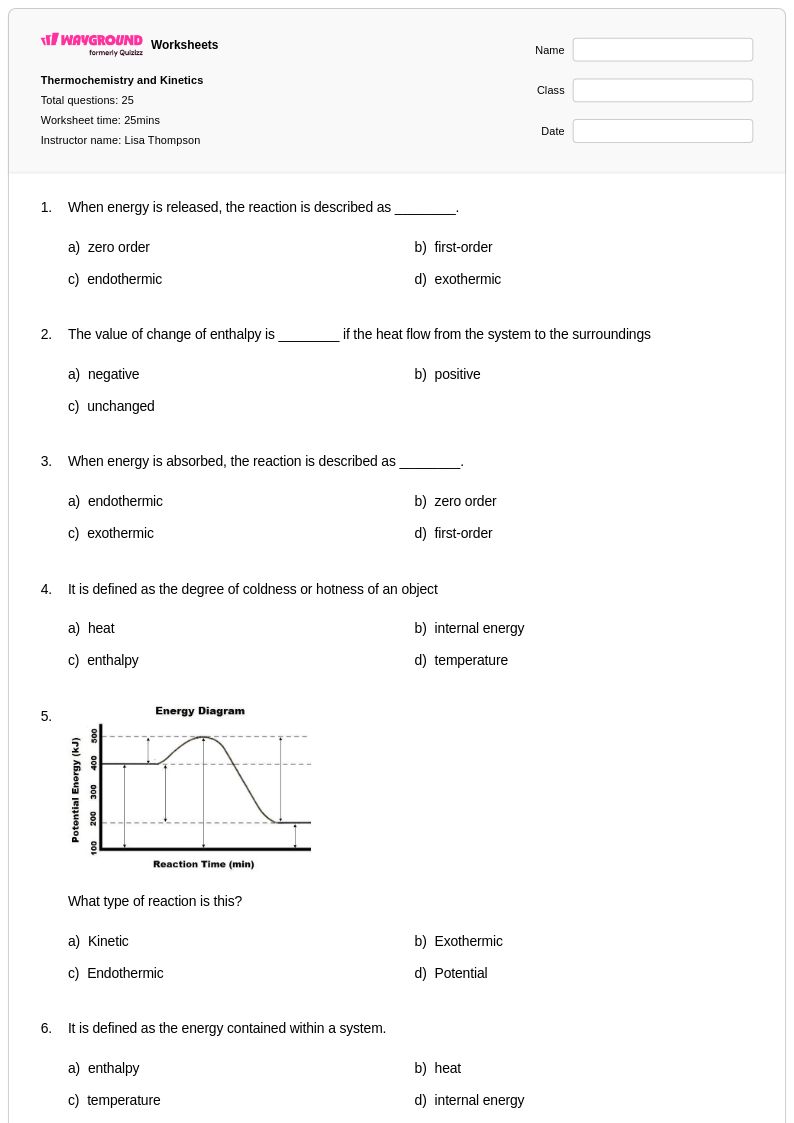
25 Q
10th - Uni

15 Q
11th - Uni
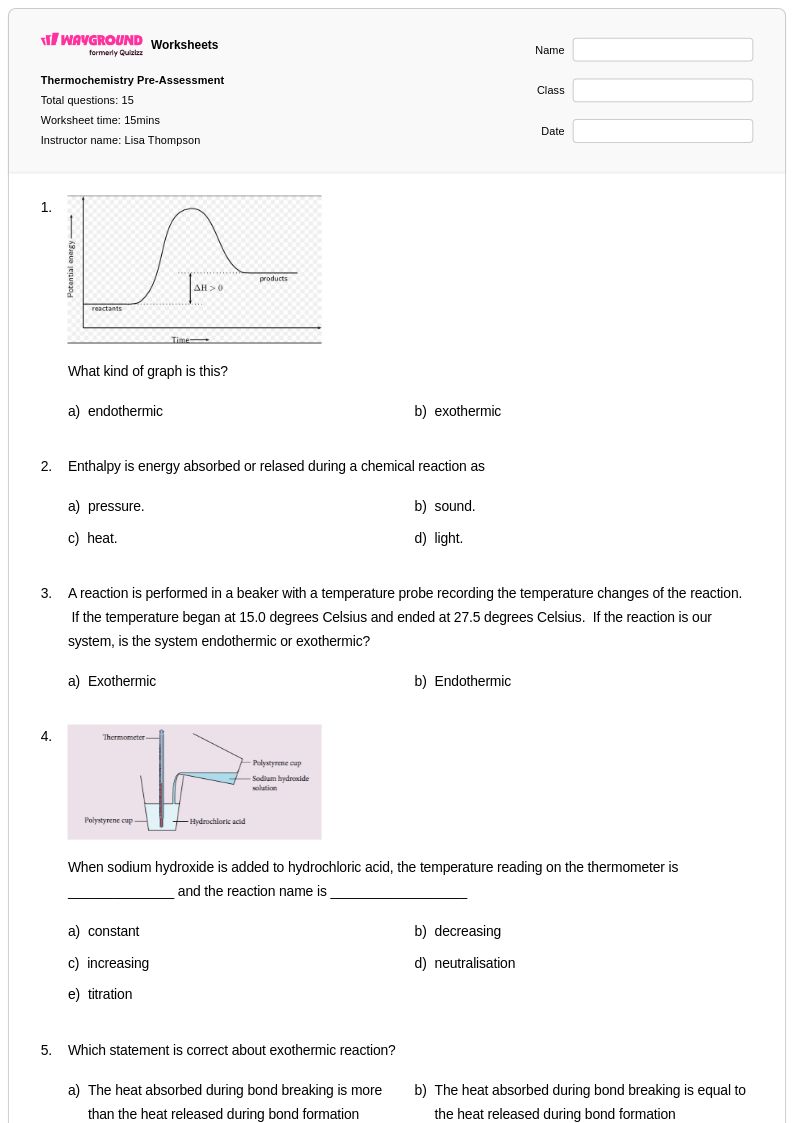
15 Q
10th - Uni

15 Q
10th - Uni
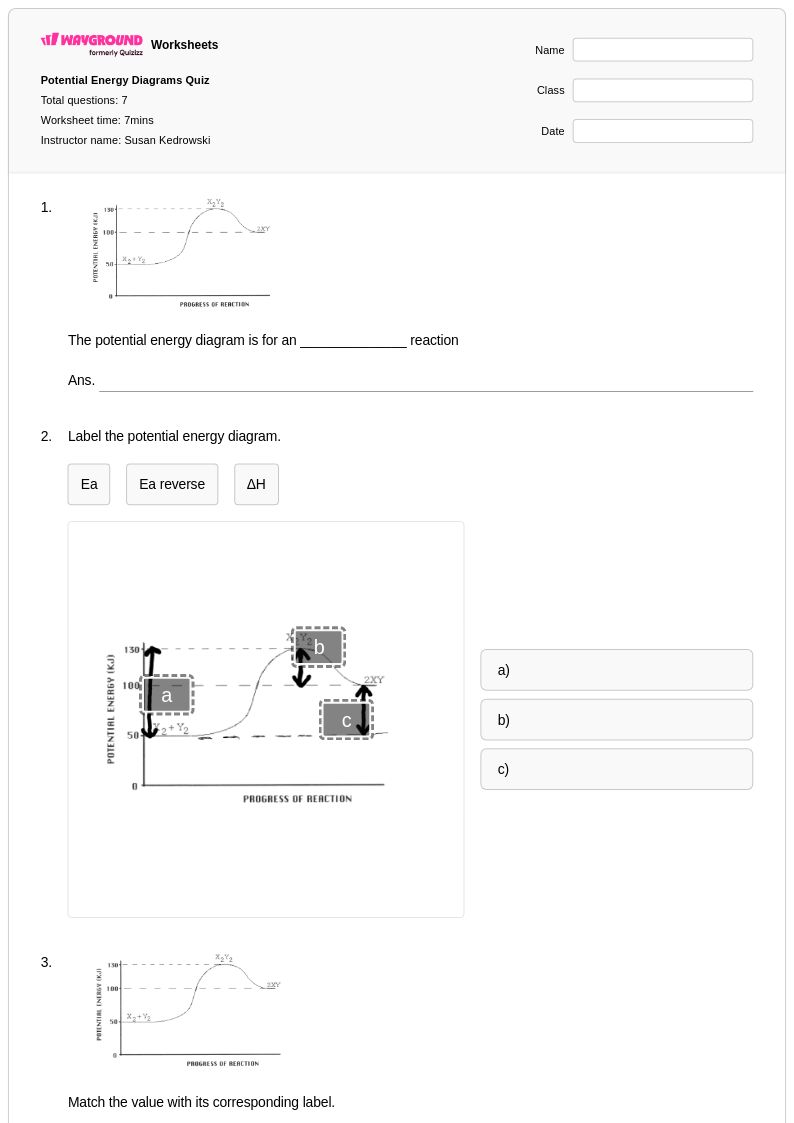
7 Q
9th - Uni
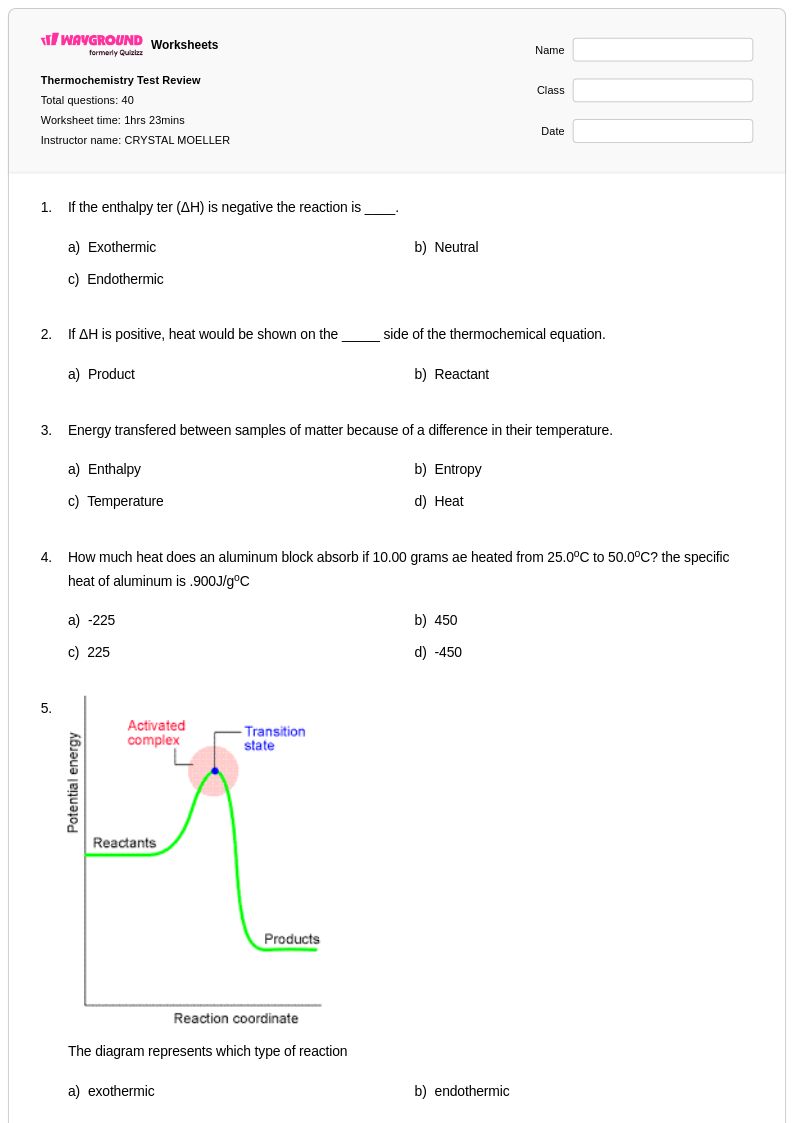
40 Q
10th - 12th

15 Q
11th - Uni

25 Q
11th - Uni

15 Q
9th - Uni

25 Q
11th - Uni

25 Q
8th - Uni
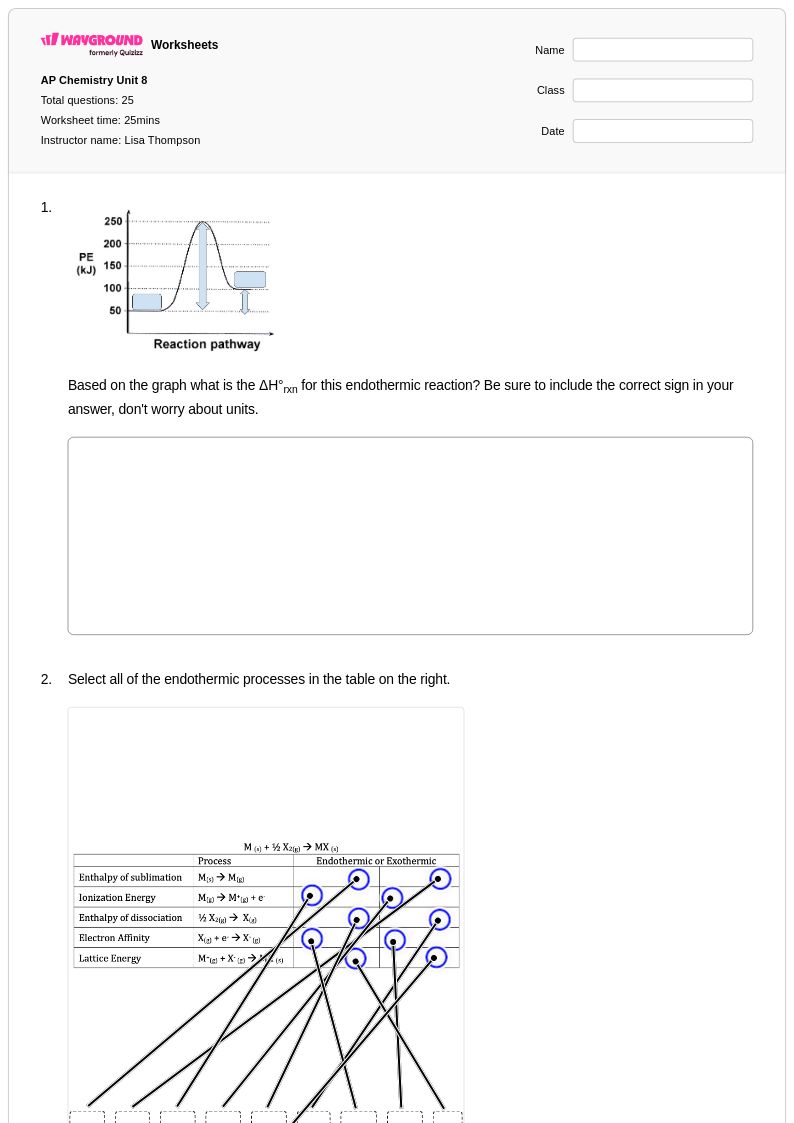
25 Q
11th - Uni
25 Q
11th - Uni

24 Q
9th - 12th

80 Q
9th - 12th

25 Q
8th - Uni

12 Q
11th - 12th

15 Q
9th - 12th
Explore Other Subject Worksheets for year 11
Explore printable Enthalpy worksheets for Year 11
Enthalpy worksheets for Year 11 students available through Wayground (formerly Quizizz) provide comprehensive practice with this fundamental thermodynamic concept that measures the total heat content of chemical systems. These expertly designed resources strengthen students' ability to calculate enthalpy changes during chemical reactions, interpret enthalpy diagrams, and apply Hess's Law to multi-step processes. The worksheet collections include detailed practice problems covering standard enthalpy of formation, combustion reactions, and bond energy calculations, with complete answer keys that support independent learning and self-assessment. Students work through scenarios involving endothermic and exothermic processes while developing proficiency in using calorimetry data and thermochemical equations. These free printable resources in pdf format ensure accessibility for diverse learning environments while building the quantitative reasoning skills essential for advanced chemistry coursework.
Wayground (formerly Quizizz) empowers educators with millions of teacher-created enthalpy worksheet resources that streamline instruction and assessment in Year 11 chemistry classrooms. The platform's robust search and filtering capabilities allow teachers to quickly locate materials aligned with specific curriculum standards and learning objectives, while differentiation tools enable customization for students with varying skill levels and learning needs. Teachers can access these comprehensive worksheet collections in both printable pdf format for traditional classroom use and digital formats for online learning environments, providing maximum flexibility in lesson delivery. These carefully curated resources support effective planning by offering ready-to-use materials for skill practice, targeted remediation for students struggling with thermodynamic concepts, and enrichment opportunities for advanced learners ready to explore complex enthalpy calculations and real-world applications in chemical engineering and environmental science.
FAQs
How do I teach enthalpy to high school chemistry students?
What exercises help students practice enthalpy calculations?
What mistakes do students commonly make when solving enthalpy problems?
How do I differentiate enthalpy worksheets for students at different skill levels?
How do I use Wayground's enthalpy worksheets in my chemistry class?
